Advanced Chiral Resolution of Dibenzobicyclo Diketones for High-Purity API Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access high-purity chiral intermediates, which are critical building blocks for active pharmaceutical ingredients (APIs). A significant technological breakthrough in this domain is documented in Chinese Patent CN103483170A, which details a novel method for resolving chiral diketone compounds. Specifically, this innovation focuses on the separation of 2,3:6,7-dibenzobicyclo[3.3.1]octa-2,6-diene-4,8-dione from its racemic mixture. Unlike conventional approaches that rely on labor-intensive precursor resolution, this patented technique utilizes a direct resolution strategy mediated by chiral binaphthol auxiliaries. By leveraging specific hydrogen bonding interactions, the process achieves exceptional optical purity with ee values reaching up to 99.9% after recrystallization. This represents a paradigm shift in how complex bicyclic diketones are manufactured, offering a robust solution for producing reliable pharmaceutical intermediate suppliers' inventory with superior quality metrics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of chiral 2,3:6,7-dibenzobicyclo[3.3.1]octa-2,6-diene-4,8-dione has been plagued by inefficiencies inherent in multi-step synthetic routes. According to prior art, such as literature cited in the patent background, the standard procedure necessitates the chiral resolution of the synthetic precursor, 2,4-diphenylglutaric acid. This legacy method involves multiple recrystallizations to remove meso-forms followed by resolution using quinine, a process that is notoriously inefficient. The yield of chiral 2,4-diphenylglutaric acid via this route is merely 17.6% with a modest ee value of 73%. Subsequently, this low-yield intermediate must undergo a Friedel-Crafts cyclization to form the target diketone, further compounding material losses. The cumulative total yield for obtaining chiral diketones through this traditional pathway is abysmal, reported at only 13.5% to 17%, with final ee values struggling to reach 97%. Such low throughput not only escalates production costs but also generates significant chemical waste, making it unsustainable for modern commercial scale-up of complex polymer additives or pharmaceutical intermediates.
The Novel Approach
In stark contrast, the method disclosed in CN103483170A bypasses the cumbersome precursor resolution entirely by targeting the diketone molecule directly. This innovative approach employs R-1,1'-bi-2-naphthol or S-1,1'-bi-2-naphthol as chiral resolving agents to interact directly with the racemic diketone substrate. ![Chemical structure of racemic 2,3:6,7-dibenzobicyclo[3.3.1]octa-2,6-diene-4,8-dione substrate](/insights/img/chiral-diketone-binol-resolution-pharma-supplier-20260309025115-02.png) . The process operates under mild conditions, typically in toluene or ethyl acetate solvents at temperatures ranging from 15°C to 35°C. By optimizing the molar ratio of the resolving agent to the diketone, preferably around 0.6:1, the system facilitates the selective precipitation of one enantiomer while leaving the other in the mother liquor. This direct resolution strategy dramatically improves the total yield of R and S-type chiral diketones with ee values exceeding 99% to a remarkable 55%. This tripling of efficiency compared to the literature's 17% fundamentally alters the economic viability of producing this high-purity OLED material or API intermediate.
. The process operates under mild conditions, typically in toluene or ethyl acetate solvents at temperatures ranging from 15°C to 35°C. By optimizing the molar ratio of the resolving agent to the diketone, preferably around 0.6:1, the system facilitates the selective precipitation of one enantiomer while leaving the other in the mother liquor. This direct resolution strategy dramatically improves the total yield of R and S-type chiral diketones with ee values exceeding 99% to a remarkable 55%. This tripling of efficiency compared to the literature's 17% fundamentally alters the economic viability of producing this high-purity OLED material or API intermediate.
Mechanistic Insights into Hydrogen-Bond Mediated Chiral Resolution
The core scientific principle driving this high-efficiency separation lies in the supramolecular recognition between the chiral auxiliary and the substrate. The resolution mechanism relies heavily on the formation of diastereomeric complexes stabilized by intermolecular hydrogen bonds. When a specific enantiomer of the resolving agent, such as R-1,1'-bi-2-naphthol shown below, is introduced to the racemic mixture, it exhibits a high degree of symmetry matching with one specific enantiomer of the diketone. 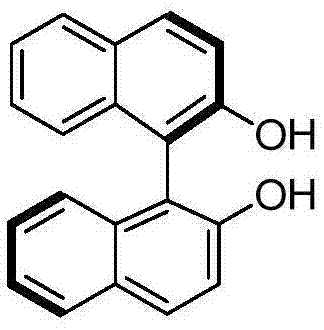 . For instance, R-BINOL matches symmetrically with the S-type diketone, allowing them to stack in an ordered manner through hydrogen bonding interactions within the solvent matrix. This ordered stacking reduces the solubility of the specific diastereomeric complex, causing it to precipitate out of the solution as a solid. Conversely, the mismatched pair (e.g., R-BINOL with R-diketone) cannot form these stable, ordered hydrogen-bonded networks effectively, remaining soluble in the organic phase. This differential solubility is the thermodynamic driver that enables the physical separation of the enantiomers through simple filtration.
. For instance, R-BINOL matches symmetrically with the S-type diketone, allowing them to stack in an ordered manner through hydrogen bonding interactions within the solvent matrix. This ordered stacking reduces the solubility of the specific diastereomeric complex, causing it to precipitate out of the solution as a solid. Conversely, the mismatched pair (e.g., R-BINOL with R-diketone) cannot form these stable, ordered hydrogen-bonded networks effectively, remaining soluble in the organic phase. This differential solubility is the thermodynamic driver that enables the physical separation of the enantiomers through simple filtration.
Furthermore, the process includes a sophisticated recovery and purification loop that ensures both high yield and high purity. The solid precipitate, enriched in one enantiomer, is washed with aqueous sodium hydroxide to remove residual resolving agent and then recrystallized from ethyl acetate. This recrystallization step is critical for pushing the optical purity from initial values of 73-95% up to the ultra-high standard of 99.9% ee. Simultaneously, the mother liquor, which contains the opposite enantiomer, is processed similarly to recover the second chiral product. The ability to recover the chiral resolving agent from the aqueous washes by acidification with hydrochloric acid is another key mechanistic feature. This recovery step ensures that the expensive chiral auxiliary retains its optical integrity (ee value remains unchanged) and can be recycled, thereby minimizing the consumption of chiral pool resources and enhancing the overall green chemistry profile of the manufacturing process.
How to Synthesize 2,3:6,7-Dibenzobicyclo Diketone Efficiently
Implementing this resolution technology requires precise control over reaction parameters to maximize the separation factor. The synthesis begins with the preparation of the racemic diketone, typically via cyclization of 2,4-diphenylglutaric acid using concentrated sulfuric acid, followed by the critical resolution step. The detailed standardized synthesis steps for achieving the optimal 55% total yield and 99.9% ee are outlined in the guide below. This protocol emphasizes the importance of solvent selection, where toluene is preferred over ethyl acetate for better yield trends, and strict temperature control between 15-35°C. Operators must ensure the molar concentration of the diketone is maintained around 0.2 mol/L to prevent saturation issues that could lead to co-precipitation of impurities. By adhering to these optimized conditions, manufacturers can consistently produce batch-after-batch of material that meets the stringent purity specifications required for downstream drug synthesis.
- Mix racemic diketone with R- or S-1,1'-bi-2-naphthol in toluene solvent at a molar ratio of approximately 0.6: 1.
- Maintain reaction temperature between 15-35°C for 1-24 hours to allow hydrogen-bonded complex formation and precipitation.
- Filter the solid precipitate and recrystallize from ethyl acetate to achieve optical purity exceeding 99% ee.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented resolution method translates into tangible strategic advantages beyond mere technical metrics. The most significant benefit is the drastic simplification of the supply chain for chiral intermediates. By eliminating the need to source and resolve the precursor 2,4-diphenylglutaric acid separately, companies can reduce the number of vendor qualifications and raw material inventories required. This consolidation of the synthetic route inherently reduces lead time for high-purity pharmaceutical intermediates, allowing for faster response to market demands. Furthermore, the ability to recover and reuse the chiral resolving agent with nearly quantitative efficiency means that the variable cost associated with chiral auxiliaries is significantly reduced. This creates a more stable cost structure that is less susceptible to fluctuations in the pricing of specialty chiral reagents.
- Cost Reduction in Manufacturing: The transition from a multi-step precursor resolution to a direct diketone resolution fundamentally lowers the cost of goods sold (COGS). By skipping the low-yield resolution of 2,4-diphenylglutaric acid, which historically wasted over 80% of the material, the new process captures value much earlier in the synthesis. The elimination of the Friedel-Crafts step post-resolution further reduces energy consumption and reagent usage. Qualitatively, this streamlining removes entire unit operations from the production schedule, leading to substantial cost savings in labor, utilities, and waste disposal without compromising on the quality of the final API intermediate.
- Enhanced Supply Chain Reliability: Reliability in the supply of chiral building blocks is often compromised by the complexity of legacy synthesis routes. This new method enhances reliability by utilizing robust, commodity-grade solvents like toluene and ethyl acetate, which are readily available globally, unlike specialized chiral catalysts that may have long lead times. The mild reaction conditions (15-35°C) also reduce the risk of thermal runaways or equipment failures, ensuring consistent production uptime. Additionally, the high recovery rate of the chiral auxiliary acts as a buffer against supply disruptions of the resolving agent, as the internal inventory of recovered material can sustain production for extended periods.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, but this resolution method is designed for scalability. The reliance on crystallization and filtration rather than complex chromatographic separations makes it highly amenable to large-scale batch processing. From an environmental perspective, the process aligns with green chemistry principles by maximizing atom economy through higher yields and enabling the recycling of the chiral selector. The reduction in solvent usage per kilogram of product, combined with the ability to treat aqueous waste streams effectively, ensures that the manufacturing process meets rigorous environmental compliance standards, facilitating smoother regulatory approvals for commercial scale-up of complex pharmaceutical additives.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this resolution technology is essential for stakeholders evaluating its integration into their supply chains. The following questions address common inquiries regarding the practical application and performance of this method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring that decision-makers have accurate information regarding yield expectations, purity levels, and operational parameters. This transparency helps mitigate risk during the technology transfer phase.
Q: What is the primary advantage of this resolution method over traditional precursor splitting?
A: Traditional methods require resolving the precursor 2,4-diphenylglutaric acid first, which yields only 17.6% with 73% ee, followed by a Friedel-Crafts reaction. This patented method resolves the diketone directly, achieving a total yield of up to 55% with ee values reaching 99.9%, significantly reducing process steps and material loss.
Q: Can the chiral resolving agent be recovered and reused?
A: Yes, the process allows for the efficient recovery of the chiral auxiliary. By acidifying the aqueous wash phase with hydrochloric acid, the 1,1'-bi-2-naphthol can be recovered with a yield of up to 97% and retained optical purity, ensuring minimal waste and lower raw material costs.
Q: What are the optimal reaction conditions for maximum enantiomeric excess?
A: Optimal results are achieved using toluene as the solvent with a diketone concentration of 0.2 mol/L. The preferred molar ratio of resolving agent to diketone is 0.6:1, reacting at 25°C for 6 hours, followed by recrystallization from ethyl acetate to reach 99.9% ee.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3:6,7-Dibenzobicyclo Diketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our technical team has extensively analyzed the potential of the resolution method described in CN103483170A and is fully equipped to translate this laboratory-scale innovation into industrial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this high-yield, high-ee process are realized at the volume your business requires. Our facilities are supported by rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2,3:6,7-dibenzobicyclo[3.3.1]octa-2,6-diene-4,8-dione meets the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to optimize your supply chain for chiral ketones. By leveraging our expertise in process development, we can provide a Customized Cost-Saving Analysis tailored to your specific production volumes. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Together, we can secure a sustainable and cost-effective supply of this vital intermediate, driving your drug development programs forward with confidence and efficiency.
