Scalable Synthesis of Saprorthoquinone: A Cost-Effective Route for Antitumor API Intermediates
Scalable Synthesis of Saprorthoquinone: A Cost-Effective Route for Antitumor API Intermediates
The pharmaceutical industry's relentless pursuit of potent antitumor agents has placed significant focus on naphthoquinone derivatives, particularly Saprorthoquinone (Formula I), a natural product isolated from Salvia prionitis Hance. As detailed in patent CN102070431B, a breakthrough synthetic methodology has been established that fundamentally alters the supply chain dynamics for this critical compound. Historically, reliance on plant extraction yielded negligible quantities, while early synthetic routes were plagued by excessive step counts and prohibitive costs. This novel approach leverages 7-methoxy-1-tetralone as a robust starting material, employing a strategic sequence of carbonyl alpha-site methylation, selective aromatic isopropylation, and Grignard addition to construct the complex scaffold efficiently. By integrating modern oxidation protocols using 2-iodoxybenzoic acid (IBX), this process delivers high-purity intermediates suitable for downstream drug development.
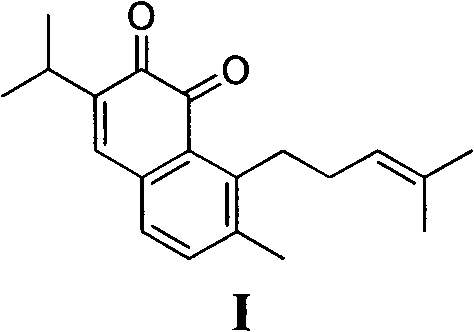
For R&D directors evaluating process feasibility, the structural integrity and purity profile of the final active pharmaceutical ingredient (API) intermediate are paramount. The described synthesis not only achieves the correct regiochemistry for the isopropyl and prenyl substituents but also ensures the formation of the sensitive ortho-quinone moiety without over-oxidation or degradation. This level of control is essential for maintaining the biological activity observed in preliminary studies, where the compound demonstrated significant inhibition against leukemia and lung cancer cell lines. Consequently, this method represents a viable pathway for generating the substantial quantities of material required for preclinical and clinical trials, positioning it as a cornerstone for reliable pharmaceutical intermediate supplier networks aiming to support oncology drug pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the acquisition of Saprorthoquinone was bottlenecked by two inefficient strategies: direct plant extraction and cumbersome total synthesis. Extraction from Salvia prionitis resulted in abysmal recovery rates of approximately 0.05%, rendering it commercially impossible to secure kilogram-scale quantities. On the synthetic front, early methodologies, such as the route developed by Matsumoto et al., relied on (+)-methyl dehydroabietic acid as a chiral pool starting material. While chemically elegant, this 15-step sequence involved expensive reagents and complex purification protocols that drove costs exponentially higher with each transformation.
Furthermore, alternative semi-synthetic approaches utilizing ferrustol or citric acid derivatives introduced severe safety and scalability hazards. For instance, certain legacy routes necessitated the use of Ni/Al alloys for reduction steps, a process known to generate uncontrollable volumes of carbon dioxide gas, posing significant engineering challenges in large reactors. Other methods required ultrasonic catalysis or high-temperature dehydration over Pd-C, conditions that are difficult to replicate consistently in a GMP manufacturing setting. These technical barriers created a fragile supply chain, where any disruption in the availability of niche starting materials or specialized equipment could halt production entirely, highlighting the urgent need for a more robust and accessible synthetic strategy.
The Novel Approach
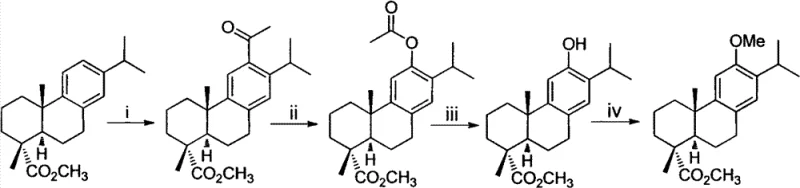
The methodology disclosed in CN102070431B circumvents these historical obstacles by initiating synthesis from 7-methoxy-1-tetralone, a commodity chemical that is both inexpensive and readily available in bulk quantities. The core innovation lies in the precise construction of the naphthalene skeleton through a concise eight-step sequence that maximizes atom economy and minimizes waste. Key transformations include a highly selective Friedel-Crafts alkylation using polyphosphoric acid (PPA) to install the isopropyl group, followed by a Grignard reaction to attach the essential prenyl side chain. This linear progression avoids the branching complexities and protecting group manipulations that characterized earlier attempts.
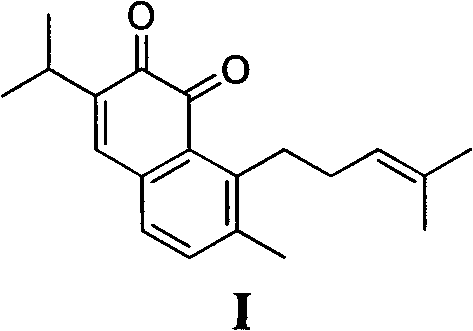
Crucially, the final stages of the synthesis employ 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) for aromatization and IBX for the final oxidation to the quinone. These reagents offer superior selectivity compared to traditional oxidants like Fremy's salt, reducing the formation of tarry byproducts and simplifying downstream purification. For procurement managers, this translates to a drastic simplification of the bill of materials, replacing exotic natural product derivatives with standard industrial chemicals. The result is a process that is not only chemically superior but also economically viable, enabling cost reduction in API manufacturing by eliminating the need for costly chiral starting materials and hazardous reaction conditions.
Mechanistic Insights into PPA-Catalyzed Alkylation and IBX Oxidation
From a mechanistic perspective, the success of this route hinges on the regioselective functionalization of the tetralone core. The introduction of the isopropyl group via PPA-catalyzed alkylation is a critical determinant of the final product's efficacy. Polyphosphoric acid acts as both a solvent and a catalyst, generating the electrophilic isopropyl cation in situ which attacks the electron-rich aromatic ring. The methoxy group at the 7-position directs the incoming electrophile to the 6-position due to strong ortho/para directing effects, ensuring the correct substitution pattern required for biological activity. This step demonstrates exceptional tolerance to the existing ketone functionality, avoiding unwanted side reactions that often plague Lewis acid-catalyzed alkylations on sensitive substrates.
Following the construction of the carbon skeleton, the conversion of the naphthol precursor to the target ortho-quinone requires careful control of oxidation potential. The use of IBX (2-iodoxybenzoic acid) in dimethylformamide provides a mild yet effective oxidation environment. Unlike harsher oxidants that might degrade the sensitive prenyl double bond or cause ring cleavage, IBX selectively targets the phenolic hydroxyl groups generated after demethylation. The mechanism involves the formation of an iodine-phenol intermediate which undergoes elimination to restore aromaticity and establish the quinone carbonyls. This specificity is vital for impurity control, ensuring that the final product meets stringent purity specifications without requiring extensive chromatographic purification, thereby facilitating the commercial scale-up of complex quinones.
How to Synthesize Saprorthoquinone Efficiently
The execution of this synthesis requires strict adherence to reaction parameters to maintain high yields and reproducibility. The process begins with the alpha-methylation of 7-methoxy-1-tetralone using sodium hydride and diethyl carbonate, followed by decarboxylation to install the alpha-methyl group. Subsequent steps involve the PPA-mediated isopropylation and the addition of the prenyl Grignard reagent, which must be performed under anhydrous conditions to prevent reagent decomposition. The final aromatization and oxidation steps are rapid and exothermic, necessitating precise temperature control. Detailed standardized operating procedures for each unit operation, including quench protocols and crystallization parameters, are essential for technology transfer.
- Perform carbonyl alpha-site methylation on 7-methoxy-1-tetralone using diethyl carbonate and a strong base to form the beta-keto ester intermediate.
- Execute selective isopropyl substitution on the aromatic ring using polyphosphoric acid (PPA) and isopropanol, followed by a Grignard reaction to introduce the prenyl side chain.
- Complete the synthesis via DDQ-mediated aromatization, ethanethiol demethylation, and final oxidation using IBX to yield the target o-quinone.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders, the transition to this synthetic route offers immediate relief from the volatility associated with natural product sourcing. By decoupling production from agricultural variables such as harvest seasons and geographic availability, manufacturers can guarantee consistent delivery schedules. The reliance on 7-methoxy-1-tetralone as a feedstock leverages an established global supply chain for bulk aromatics, mitigating the risk of raw material shortages. Furthermore, the reduction in total step count from over 15 in prior art to just 8 significantly compresses the manufacturing cycle time, allowing for faster response to market demand fluctuations and reducing lead time for high-purity intermediates.
- Cost Reduction in Manufacturing: The economic impact of this route is driven by the substitution of expensive chiral pool starting materials with commodity chemicals. Eliminating the need for (+)-methyl dehydroabietic acid or ferrustol removes a major cost driver from the bill of materials. Additionally, the avoidance of specialized equipment for ultrasonic catalysis or high-pressure hydrogenation reduces capital expenditure requirements. The use of standard solvents like ethanol, THF, and dichloromethane further aligns the process with existing infrastructure, resulting in substantial cost savings without compromising quality.
- Enhanced Supply Chain Reliability: The robustness of the reagents used, such as DDQ and IBX, ensures that production is not vulnerable to the supply disruptions often seen with niche catalysts. The process operates under atmospheric pressure and moderate temperatures, reducing the dependency on specialized high-pressure reactors or cryogenic cooling systems. This operational simplicity enhances the resilience of the supply chain, ensuring that production can be maintained across multiple manufacturing sites globally, thereby securing a stable supply of this critical antitumor intermediate.
- Scalability and Environmental Compliance: From an environmental standpoint, this method offers a cleaner profile by avoiding the generation of heavy metal waste associated with chromium-based oxidations or nickel reductions used in older methods. The waste streams are primarily organic and can be treated using standard incineration or solvent recovery protocols. The high atom efficiency of the Grignard and alkylation steps minimizes waste generation per kilogram of product, aligning with green chemistry principles and facilitating regulatory approval for commercial manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on process capabilities and limitations.
Q: What are the primary advantages of this synthesis route over plant extraction?
A: Plant extraction yields are extremely low (approximately 0.05%), making it unviable for commercial supply. This chemical synthesis offers a scalable, high-yield alternative using readily available starting materials like 7-methoxy-1-tetralone.
Q: How does this method improve safety compared to prior art syntheses?
A: Unlike previous methods requiring hazardous Ni/Al alloy reductions which generate large volumes of CO2 gas, or high-temperature Pd-C dehydration, this route utilizes milder conditions with standard reagents like PPA and IBX, significantly enhancing operational safety.
Q: Is this process suitable for large-scale GMP manufacturing?
A: Yes, the route avoids exotic catalysts and ultra-low temperature constraints. The use of robust reagents like DDQ for aromatization and IBX for oxidation allows for straightforward process control and scalability in a GMP environment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Saprorthoquinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of securing a stable supply of high-value antitumor intermediates like Saprorthoquinone. Our technical team has thoroughly analyzed this patented route and validated its potential for industrial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch delivered meets the exacting standards required for pharmaceutical development.
We invite you to collaborate with us to optimize your supply chain for this critical compound. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this synthetic route can lower your overall cost of goods. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring your project remains on schedule and within budget.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
