Scalable One-Pot Synthesis of Piperazinone-Hydroxypyridone Derivatives for Antiviral Drug Development
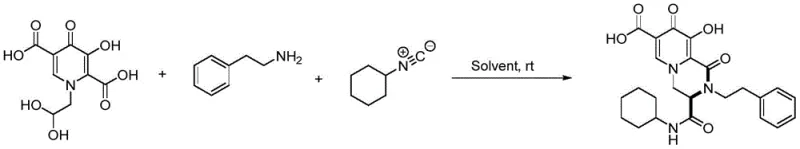
The pharmaceutical industry is constantly seeking more efficient pathways to synthesize complex heterocyclic scaffolds that serve as the backbone for next-generation antiviral therapeutics. A significant breakthrough in this domain is documented in patent CN110698473B, which discloses a novel class of piperazinone-hydroxypyridone-5-carboxyl compounds and their preparation methods. These compounds are critical intermediates in the development of RNA polymerase inhibitors, targeting viral replication mechanisms in pathogens such as HIV and influenza. The patent highlights a transformative shift from laborious, multi-step synthetic routes to a streamlined, one-pot multicomponent reaction. This technological advancement addresses long-standing challenges in medicinal chemistry, specifically the need for mild reaction conditions, high atom economy, and the elimination of toxic transition metal catalysts. By enabling the simultaneous introduction of multiple substituents under ambient conditions, this innovation offers a robust platform for generating diverse libraries of bioactive molecules, thereby accelerating the drug discovery pipeline for global pharmaceutical partners.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of piperazinone-fused hydroxypyridone cores, which are essential for chelating metal ions in viral enzyme active sites, has been fraught with inefficiencies. Traditional approaches, such as the de novo synthesis of representative inhibitors like S-033188, often necessitate upwards of thirteen distinct reaction steps starting from complex precursors like 3-benzyloxy-4-oxo-4H-pyran-2-carboxylic acid. These legacy methods are not only time-consuming but also rely heavily on expensive reagents and harsh reaction conditions that can compromise the integrity of sensitive functional groups. Furthermore, the cumulative yield across such a lengthy synthetic sequence is invariably low, leading to substantial material waste and inflated production costs. The reliance on transition metal catalysts in many conventional cyclization protocols introduces an additional burden, requiring rigorous and costly purification steps to remove trace metal residues to meet stringent pharmaceutical quality standards. These factors collectively create a bottleneck in the supply chain, limiting the availability of high-purity intermediates needed for clinical development and commercial manufacturing.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology described in the patent utilizes a highly efficient one-pot condensation reaction that dramatically simplifies the synthetic landscape. By reacting 1-(2,2-dihydroxyethyl)-3-hydroxy-4-oxo-1,4-dihydropyridone-2,5-dicarboxylic acid directly with various amines and isonitriles, the target piperazinone-hydroxypyridone scaffold is constructed in a single operational step. This approach operates effectively at room temperature, eliminating the need for energy-intensive heating or cooling cycles. The reaction demonstrates remarkable versatility, tolerating a wide array of aromatic and aliphatic substituents on both the amine and isonitrile components without significant loss in efficiency. Crucially, this metal-free protocol ensures that the final product is free from heavy metal contamination, thereby simplifying downstream processing and regulatory compliance. The simplicity of the workup, often requiring only suction filtration to isolate the solid product, underscores the practical utility of this method for industrial applications.
Mechanistic Insights into Metal-Free One-Pot Cyclization
The core of this synthetic innovation lies in the unique reactivity of the 1-(2,2-dihydroxyethyl) substituted pyridone precursor, which acts as a multifunctional building block capable of undergoing cascade cyclization. In the presence of an amine and an isonitrile, the reaction likely proceeds through an initial nucleophilic attack followed by an intramolecular cyclization that forms the piperazinone ring fused to the hydroxypyridone system. The absence of external metal catalysts suggests that the reaction is driven by the inherent electronic properties of the substrates and the specific activation provided by the solvent medium. Experimental optimization revealed that solvent polarity plays a pivotal role in facilitating this transformation. Protic solvents such as methanol and ethanol, as well as polar aprotic solvents like acetonitrile, were found to support conversion rates reaching 100%, indicating excellent solubility and stabilization of the transition states involved. Conversely, non-polar solvents like toluene resulted in significantly lower conversion, highlighting the importance of polar interactions in the reaction mechanism. This mechanistic understanding allows for precise tuning of reaction conditions to maximize yield and purity for diverse substrate combinations.
From an impurity control perspective, the direct nature of this one-pot synthesis inherently minimizes the formation of side products typically associated with multi-step sequences. Since the reaction does not involve isolation of unstable intermediates, the risk of degradation or side reactions between steps is virtually eliminated. The high selectivity of the cyclization ensures that the major product is the desired piperazinone-hydroxypyridone derivative, as evidenced by the clean NMR spectra and high melting points reported in the examples. For instance, Example 1 yielded a white solid with a purity profile suitable for direct biological testing, achieving a yield of 78.2% without the need for chromatographic purification. This inherent cleanliness of the reaction profile is a significant advantage for process chemists aiming to develop robust and scalable manufacturing processes that adhere to Green Chemistry principles. The ability to introduce diverse R1 and R2 groups while maintaining high fidelity in the core structure provides a powerful tool for structure-activity relationship (SAR) studies.
How to Synthesize Piperazinone-Hydroxypyridone Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to stoichiometry and solvent selection to ensure optimal results. The general procedure involves dissolving the pyridone dicarboxylic acid precursor, the chosen amine, and the isonitrile in a suitable solvent such as methanol. The molar ratio is typically maintained at 1:1.2:1.2 to drive the reaction to completion while minimizing excess reagent waste. The mixture is then stirred at room temperature for a period ranging from 0.5 to 2 hours, with reaction progress monitored via TLC. Upon completion, the product often precipitates directly from the reaction mixture, allowing for simple isolation via suction filtration. This straightforward protocol eliminates the need for complex extraction or distillation steps, making it accessible for rapid scale-up. For detailed standardized operating procedures and specific parameter adjustments for different substrates, please refer to the guide below.
- Dissolve 1-(2,2-dihydroxyethyl)-3-hydroxy-4-oxo-1,4-dihydropyridone-2,5-dicarboxylic acid, amine, and isonitrile in a protic or aprotic solvent such as methanol or ethanol.
- Maintain the reaction mixture at room temperature for 0.5 to 2 hours, ensuring a molar ratio of approximately 1: 1.2:1.2 for the acid, amine, and isonitrile respectively.
- Perform suction filtration on the reaction solution to isolate the solid target compound, eliminating the need for complex purification or metal scavenging steps.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond mere technical feasibility. The elimination of transition metal catalysts removes a significant cost center associated with both the purchase of expensive catalysts and the subsequent validation of their removal from the final API. This simplification translates directly into a more streamlined cost structure for the manufacturing of these critical pharmaceutical intermediates. Furthermore, the use of readily available and inexpensive starting materials, such as simple amines and isonitriles, enhances supply chain security by reducing dependency on specialized or scarce reagents. The mild reaction conditions also imply lower energy consumption, contributing to a reduced carbon footprint and alignment with corporate sustainability goals. These factors collectively position this technology as a highly attractive option for long-term sourcing strategies in the competitive antiviral drug market.
- Cost Reduction in Manufacturing: The metal-free nature of this synthesis fundamentally alters the cost dynamics of production. By avoiding the use of precious metal catalysts, manufacturers eliminate the need for specialized scavenging resins and extensive analytical testing for residual metals, which are often mandatory for regulatory approval. Additionally, the one-pot design reduces the number of unit operations, lowering labor costs and equipment usage time. The high yields observed across various substrates mean that less raw material is required to produce the same amount of product, further driving down the cost of goods sold (COGS). This economic efficiency allows for more competitive pricing models when supplying these intermediates to downstream drug developers.
- Enhanced Supply Chain Reliability: The robustness of this reaction against varying substrate structures ensures a stable supply of diverse intermediates. Since the reaction tolerates a wide range of functional groups, suppliers can maintain a flexible inventory of raw materials rather than relying on custom-synthesized precursors that may have long lead times. The simplicity of the workup, often involving just filtration, reduces the risk of batch failures due to complex purification issues. This reliability is crucial for maintaining continuous production schedules and meeting the tight deadlines often imposed by clinical trial timelines. Suppliers utilizing this method can offer greater assurance of delivery consistency, mitigating the risks of supply disruptions that can plague more fragile synthetic routes.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or ton scale is facilitated by the absence of exothermic hazards associated with strong reagents or high temperatures. The use of common solvents like methanol and ethanol simplifies solvent recovery and recycling systems, aligning with modern environmental, health, and safety (EHS) standards. The reduction in chemical waste generated per kilogram of product supports a greener manufacturing profile, which is increasingly becoming a key criterion for vendor selection by major pharmaceutical companies. This scalability ensures that the technology can grow with the demand, supporting everything from early-stage research quantities to full commercial production volumes without the need for process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these piperazinone-hydroxypyridone compounds. The answers are derived directly from the experimental data and technical specifications outlined in the patent literature. Understanding these details helps stakeholders make informed decisions regarding the integration of this technology into their existing workflows. Whether you are concerned about reaction scope, purification requirements, or regulatory implications, the following insights provide a clear overview of the method's capabilities and limitations in a commercial context.
Q: What are the primary advantages of this one-pot synthesis over traditional methods?
A: Unlike traditional multi-step syntheses that require harsh conditions and expensive reagents, this method operates at room temperature without metal catalysts, significantly simplifying purification and reducing production time.
Q: Which solvents provide the highest conversion rates for this reaction?
A: Experimental data indicates that polar protic solvents like methanol and ethanol, as well as acetonitrile, achieve conversion rates up to 100%, whereas non-polar solvents like toluene show significantly lower efficiency.
Q: Is this synthesis method suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes readily available raw materials, avoids toxic heavy metals, and involves a simple filtration workup, making it highly scalable and compliant with strict environmental regulations for pharmaceutical production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Piperazinone-Hydroxypyridone Supplier
As the demand for potent antiviral agents continues to rise, securing a reliable source of high-quality intermediates is paramount for pharmaceutical innovation. NINGBO INNO PHARMCHEM stands ready to support your development needs with our expertise in scaling complex synthetic pathways. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of piperazinone-hydroxypyridone derivatives we produce. We understand the critical nature of these intermediates in the drug development lifecycle and are committed to delivering materials that meet the highest international quality standards.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain partner dedicated to optimizing your manufacturing efficiency and accelerating your path to market with confidence and reliability.
