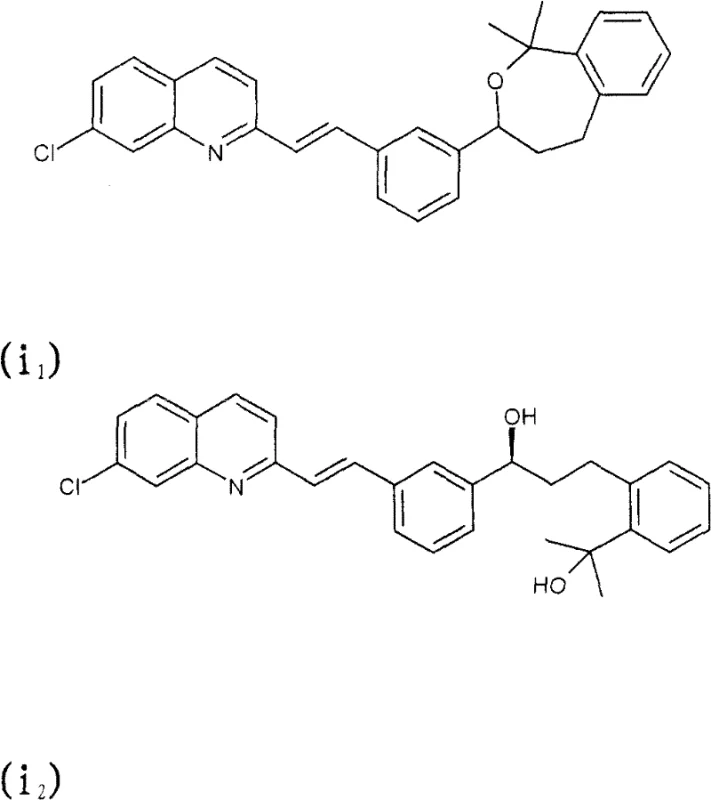
Advanced Purification of Montelukast Intermediates via Novel Chiral Amine Salt Crystallization Technology
Advanced Purification of Montelukast Intermediates via Novel Chiral Amine Salt Crystallization Technology
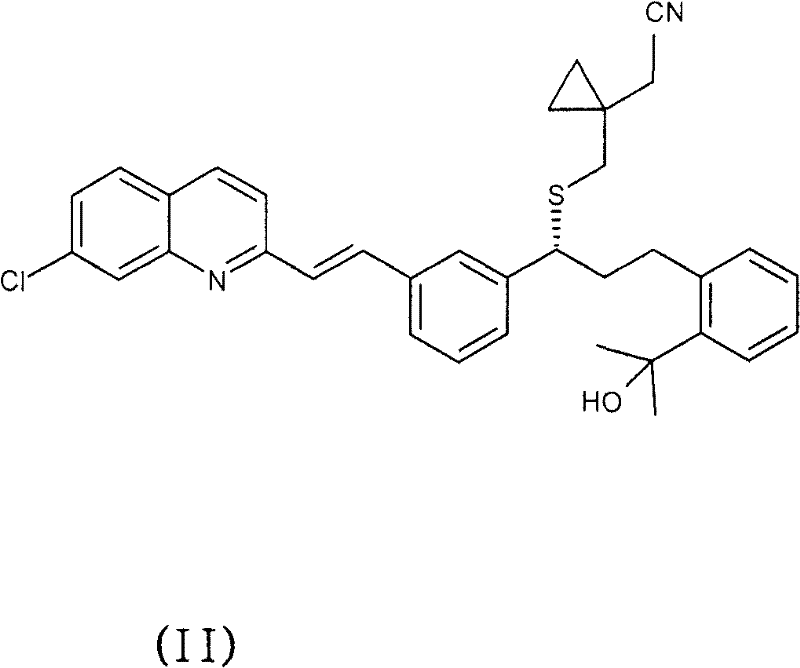
The pharmaceutical industry continuously seeks robust methodologies for the purification of complex active pharmaceutical ingredients (APIs) and their intermediates, particularly when dealing with chiral molecules prone to degradation. Patent CN101501000B introduces a groundbreaking purification process for Montelukast, a leukotriene receptor antagonist widely used in asthma management, which addresses the critical challenges of optical purity and chemical stability. This technology leverages the formation of specific novel amine salts—namely tris-(hydroxymethyl)aminomethane, L-(+)-threo-2-amino-1-phenyl-1,3-propanediol, and L-(+)-α-phenylglycinol salts—to achieve exceptional enantiomeric excess and chemical purity. Unlike traditional methods that rely on tedious chromatographic separation or yield-compromising recrystallizations, this approach offers a scalable, cost-effective route that transforms crude Montelukast acid into high-grade material suitable for final salt formation. For procurement and supply chain leaders, this represents a significant opportunity to secure a reliable pharmaceutical intermediates supplier capable of delivering consistent quality without the bottlenecks associated with conventional purification techniques.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Montelukast and its precursors has been fraught with difficulties stemming from the molecule's inherent instability towards oxygen and light, as well as the formation of stubborn by-products during synthesis. Conventional strategies, such as those described in earlier patents like EP480.717, often necessitate the generation of methyl ester intermediates followed by hydrolysis, a sequence that frequently results in low overall yields and requires extensive chromatographic purification to remove closely related impurities. Furthermore, existing salt formation methods using agents like dicyclohexylamine or tert-butylamine, while functional, often fail to provide sufficient upgrading of optical purity when starting from crude materials with lower enantiomeric excess. These legacy processes are not only time-consuming and labor-intensive but also environmentally burdensome due to the large volumes of solvents and silica gel required for column chromatography, making them less attractive for cost reduction in pharmaceutical intermediate manufacturing on a commercial scale.
The Novel Approach
The innovative methodology disclosed in CN101501000B circumvents these historical bottlenecks by utilizing a targeted crystallization strategy based on novel chiral amine salts. By converting Montelukast acid into specific salts such as the L-(+)-α-phenylglycinol salt, the process achieves a surprising degree of purification even when the starting material possesses relatively low optical purity. This approach eliminates the need for chromatographic columns entirely, relying instead on the differential solubility and crystallization kinetics of the diastereomeric salts to exclude impurities. The result is a streamlined workflow that delivers Montelukast with high chemical purity (often exceeding 98% HPLC area) and exceptional optical purity (e.e. >99.5%) directly from the crystallization mother liquor. This shift from separation-based purification to crystallization-based purification fundamentally alters the economic and operational landscape of producing high-purity pharmaceutical intermediates.
Mechanistic Insights into Chiral Amine Salt Resolution and pH-Controlled Extraction
The core of this purification technology lies in the precise stereochemical interaction between the Montelukast acid and the selected chiral amine resolving agents. When L-(+)-α-phenylglycinol or L-(+)-threo-2-amino-1-phenyl-1,3-propanediol is introduced, it forms a diastereomeric salt with the Montelukast acid. Due to the specific three-dimensional arrangement of the hydroxyl-substituted ethylene groups on these amines, the resulting salt lattice exhibits distinct solubility characteristics compared to the salts formed with unwanted enantiomers or structural impurities. This allows for the selective crystallization of the desired Montelukast-amine complex, effectively locking the target molecule into a solid phase while leaving impurities in the solution. The process is further enhanced by the stability of these salts, which can be isolated as solvates (e.g., ethanol or methanol solvates) to further refine the crystal habit and purity profile, ensuring that the optical integrity of the molecule is maintained throughout the isolation and subsequent regeneration steps.
Complementing the salt formation is a sophisticated multi-stage pH swing extraction protocol designed to remove specific process-related impurities prior to crystallization. As illustrated in the impurity profiles below, the process targets specific by-products that typically co-elute with Montelukast.
The purification workflow involves washing the aqueous phase containing the Montelukast salt at highly alkaline conditions (pH 12.0 to 13.5) to extract acidic impurities into an organic solvent, followed by a secondary wash at moderate alkalinity (pH 8.5 to 10.0) to remove other neutral or weakly acidic contaminants. Finally, the purified Montelukast is back-extracted into an organic phase at a controlled pH of 4.5 to 8.0. This granular control over the ionization state of the molecule and its impurities ensures that the crude acid entering the salt formation step is already significantly upgraded, maximizing the efficiency of the final crystallization.
Furthermore, the process is compatible with crude Montelukast derived from the hydrolysis of cyano intermediates, as shown in the reaction scheme above. Specific impurities arising from this pathway, such as unreacted cyano compounds and hydrolysis by-products, are effectively managed through the aforementioned extraction steps. This compatibility ensures that the purification method is not just a standalone technique but an integrated solution that can be seamlessly embedded into the broader synthetic route of Montelukast, providing a comprehensive strategy for quality control from intermediate to final API precursor.
How to Synthesize High-Purity Montelukast Efficiently
The implementation of this purification protocol requires careful attention to solvent selection, stoichiometry, and temperature control to maximize yield and purity. The general procedure involves suspending Montelukast acid in a suitable solvent such as toluene, ethyl acetate, or alcohols, and adding the chiral amine in a near-stoichiometric amount (0.95 to 1.05 equivalents). The mixture is typically heated to ensure dissolution and then cooled slowly to induce crystallization of the pure amine salt. Following filtration and drying, the salt can be regenerated to the free acid or directly converted to the sodium salt. The detailed standardized synthesis steps for implementing this purification route are outlined in the guide below.
- Prepare crude Montelukast acid and perform selective solvent extraction at high pH (12.0-13.5) to remove acidic impurities.
- Adjust pH to neutral range (4.5-8.0) to extract purified Montelukast acid into an organic solvent.
- React the purified acid with L-(+)-α-phenylglycinol or similar chiral amines to form a crystalline salt, followed by filtration and regeneration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this purification technology translates into tangible operational improvements and risk mitigation. By eliminating the reliance on chromatographic purification, manufacturers can drastically reduce the consumption of expensive silica gel and large volumes of elution solvents, leading to significant cost reduction in pharmaceutical intermediate manufacturing. The simplified workflow also shortens the production cycle time, as filtration and crystallization are inherently faster and more scalable than column chromatography. This efficiency gain enhances the overall throughput of the facility, allowing for better responsiveness to market demand and reducing the lead time for high-purity pharmaceutical intermediates. Additionally, the use of readily available and cost-effective chiral amines like L-(+)-α-phenylglycinol ensures that the raw material costs remain stable and predictable, avoiding the supply volatility associated with exotic or proprietary resolving agents.
- Cost Reduction in Manufacturing: The elimination of chromatographic purification steps removes a major cost center from the production budget. Chromatography is not only expensive in terms of consumables like silica and solvents but also requires specialized equipment and significant labor hours for operation and solvent recovery. By replacing this with a crystallization-based purification, the process achieves substantial cost savings through reduced material usage and lower energy consumption for solvent distillation. Furthermore, the high yields associated with the novel amine salts mean that less starting material is wasted, improving the overall atom economy and reducing the cost per kilogram of the final purified product.
- Enhanced Supply Chain Reliability: The robustness of this purification method contributes to a more resilient supply chain. Traditional purification methods that rely on complex separations are prone to variability and batch failures, which can disrupt supply schedules. In contrast, crystallization processes are well-understood, easily controllable, and highly reproducible on a large scale. The ability to upgrade optical purity from lower-grade crude materials also provides a safety net against variations in upstream synthesis, ensuring that the final product consistently meets stringent quality specifications regardless of minor fluctuations in the preceding steps. This reliability is crucial for maintaining continuous supply to downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process offers distinct advantages. The reduction in solvent usage and the avoidance of solid waste generated by silica gel columns align with green chemistry principles and simplify waste disposal compliance. The process uses common industrial solvents like toluene, ethyl acetate, and alcohols, which are easily recovered and recycled. The simplicity of the unit operations—mixing, heating, cooling, and filtering—makes the technology highly scalable from pilot plant to multi-ton commercial production without the need for complex engineering modifications, facilitating rapid commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel purification technology. These insights are derived directly from the experimental data and embodiments provided in the patent literature, offering clarity on the practical aspects of adopting this method for industrial production. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing lines.
Q: Why is L-(+)-α-phenylglycinol preferred for Montelukast purification?
A: L-(+)-α-phenylglycinol is preferred because it is significantly more cost-effective than other chiral resolving agents while delivering superior optical purity upgrades (e.e. >99.5%) and high yields during crystallization.
Q: How does this process remove specific impurities without chromatography?
A: The process utilizes a multi-stage pH swing extraction strategy. By washing the aqueous salt phase at specific alkaline levels (pH 12.0-13.5 and pH 8.5-10.0), ionizable by-products are selectively partitioned into the organic wash layer, leaving the Montelukast salt in the aqueous phase.
Q: Is chromatographic purification required for the final API?
A: No, chromatographic purification is not required. The novel amine salt formation acts as a powerful purification step that achieves high chemical and optical purity through crystallization alone, making it ideal for large-scale industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Montelukast Supplier
The technological advancements detailed in CN101501000B underscore the potential for producing Montelukast intermediates with unprecedented purity and efficiency. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet the most rigorous standards. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying optical purity and impurity profiles down to trace levels. We understand that the transition to a new purification strategy requires a partner who can guarantee both quality and quantity, and our track record in the commercial scale-up of complex pharmaceutical intermediates demonstrates our capability to deliver on these promises consistently.
We invite potential partners to engage with our technical procurement team to discuss how this advanced purification route can be tailored to your specific supply needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this chromatography-free process for your operations. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize your supply chain and enhance the quality of your final pharmaceutical products.
