Advanced Synthesis of 3-Cyclopentyloxy-4-Methoxybenzaldehyde for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic routes for key intermediates that balance high purity with operational efficiency. Patent CN1946665A introduces a transformative methodology for the preparation of 3-cyclopentyloxy-4-methoxybenzaldehyde, a critical building block for PDE4 inhibitors and anti-inflammatory agents. This technology addresses longstanding challenges in phenol alkylation by shifting from traditional polar aprotic solvents to ether-based systems, specifically tetrahydrofuran (THF). For R&D directors and process chemists, this represents a significant leap forward in designing telescoped syntheses where intermediate isolation is minimized. The core innovation lies in the compatibility of the reaction medium with downstream organometallic transformations, eliminating the need for solvent swaps that traditionally plague multi-step API manufacturing. By leveraging carbonate salts and phase transfer catalysis, this method achieves quantitative conversions while maintaining a clean impurity profile suitable for stringent regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the alkylation of isovanillin to form 3-cyclopentyloxy-4-methoxybenzaldehyde has relied heavily on solvents such as N,N-dimethylformamide (DMF), acetone, or acetonitrile. While these solvents effectively dissolve the reactants, they introduce severe bottlenecks in the broader synthetic sequence. The primary drawback is the incompatibility of these polar solvents with subsequent steps involving sensitive reagents like Grignard reagents, organolithiums, or carbanions, which demand strictly anhydrous and non-electrophilic environments. Consequently, manufacturers are forced to perform laborious aqueous work-ups, including water addition, extraction, separation, and rigorous drying, just to switch the solvent system. This not only increases the cycle time and operational costs but also leads to yield losses during the isolation and purification phases. Furthermore, the disposal of large volumes of high-boiling solvents like DMF poses significant environmental and waste management challenges, complicating the sustainability profile of the manufacturing process.
The Novel Approach
The methodology disclosed in CN1946665A circumvents these issues by utilizing tetrahydrofuran (THF) as the primary reaction medium for the coupling of phenols with cycloalkyl halides. This strategic solvent choice allows the resulting 3-cyclopentyloxy-4-methoxybenzaldehyde to remain in solution, ready for immediate use in subsequent reactions without isolation. The process employs a carbonate base, such as potassium carbonate, often enhanced by a phase transfer agent like tetrabutylammonium bromide, to drive the nucleophilic substitution efficiently. By avoiding the need for solvent exchange, the novel approach drastically simplifies the workflow, reducing the number of unit operations and minimizing material handling. This seamless integration is particularly valuable for the commercial scale-up of complex pharmaceutical intermediates, where throughput and reliability are paramount. The ability to filter off inorganic salts and proceed directly to the next step represents a paradigm shift towards leaner, more agile chemical manufacturing.
Mechanistic Insights into Carbonate-Mediated Etherification
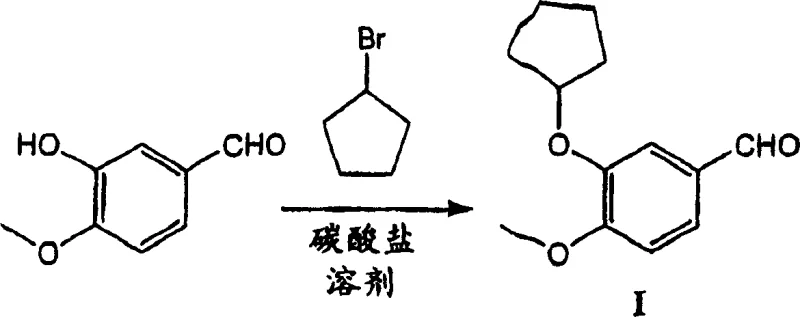
The core chemical transformation involves a nucleophilic substitution where the phenoxide anion, generated in situ by the deprotonation of the phenol by the carbonate base, attacks the cycloalkyl halide. In the context of this patent, 3-hydroxy-4-methoxybenzaldehyde reacts with cyclopentyl bromide to form the ether linkage. The use of finely ground potassium carbonate (particle size less than 536 μm or even 29 μm) increases the surface area available for reaction, thereby enhancing the kinetics of the deprotonation step. The addition of a quaternary ammonium salt acts as a phase transfer catalyst, shuttling the carbonate or phenoxide species into the organic phase where the alkyl halide resides. This mechanism ensures that the reaction proceeds homogeneously or via efficient interfacial contact, leading to high conversion rates often exceeding 97% within reasonable timeframes. The mild basicity of the carbonate also helps preserve the integrity of the aldehyde functionality, preventing unwanted side reactions such as aldol condensations that might occur with stronger bases.
Impurity control is intrinsically linked to the choice of solvent and reaction conditions. In traditional DMF-based processes, thermal degradation of the solvent or side reactions with the solvent can generate difficult-to-remove impurities. By contrast, the THF-based system described here is chemically inert under the reaction conditions, ensuring a cleaner crude profile. The patent data indicates that controlling the stoichiometry of the cycloalkyl halide, typically using a slight excess (1:1 to 1.5:1 ratio), drives the reaction to completion while minimizing the formation of dialkylated byproducts. Furthermore, the simplicity of the work-up—often just filtration of the inorganic carbonate salts—means that fewer opportunities exist for product degradation or contamination during isolation. For quality assurance teams, this translates to a more predictable impurity spectrum, facilitating easier validation and regulatory filing for the final drug substance.
How to Synthesize 3-Cyclopentyloxy-4-Methoxybenzaldehyde Efficiently
Implementing this synthesis requires careful attention to reagent quality and reaction parameters to maximize the benefits of the patented route. The process begins with the suspension of the phenolic substrate and base in anhydrous THF, followed by the controlled addition of the alkylating agent. Maintaining the reaction at reflux temperatures ensures sufficient energy for the substitution to proceed rapidly. Detailed standard operating procedures regarding the specific addition rates, temperature profiles, and filtration techniques are critical for reproducibility. For a comprehensive guide on the exact experimental conditions and stoichiometric ratios validated in the patent examples, please refer to the standardized synthesis protocol below.
- Combine 3-hydroxy-4-methoxybenzaldehyde, anhydrous potassium carbonate, and a phase transfer agent like tetrabutylammonium bromide in tetrahydrofuran solvent.
- Heat the mixture to reflux temperature (approximately 65-75°C) and add cyclopentyl bromide in portions to maintain reaction control.
- Monitor reaction completion via TLC or HPLC, then filter the mixture to remove inorganic salts, yielding a solution ready for direct downstream use.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this THF-based alkylation route offers substantial strategic advantages over legacy methods. The elimination of solvent swapping steps directly correlates to a reduction in processing time and energy consumption, which are key drivers of manufacturing costs. By streamlining the workflow, facilities can increase their batch turnover rate, effectively boosting capacity without the need for capital-intensive equipment upgrades. Moreover, the reliance on commodity chemicals like potassium carbonate and cyclopentyl bromide ensures a stable and cost-effective supply of raw materials, mitigating the risk of shortages associated with specialized reagents. This robustness is essential for maintaining continuity in the supply of high-purity pharmaceutical intermediates to global markets.
- Cost Reduction in Manufacturing: The most significant economic benefit arises from the ability to use the reaction mixture in situ for downstream transformations. Traditional methods require the isolation and drying of the intermediate, followed by redissolution in a different solvent compatible with the next step. This new approach eliminates these unit operations entirely, saving on solvent purchase, recovery, and disposal costs. Additionally, the reduced processing time lowers utility costs and labor overhead. The avoidance of high-boiling solvents like DMF also simplifies waste treatment, further contributing to overall cost optimization in pharma manufacturing.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis are widely available bulk chemicals, reducing dependency on single-source suppliers for exotic catalysts or solvents. Potassium carbonate and THF are produced at massive scales globally, ensuring consistent availability and price stability. This commoditization of the input materials enhances the resilience of the supply chain against market fluctuations. For supply chain heads, this means reduced lead time for high-purity intermediates and a lower risk of production stoppages due to material scarcity, ensuring reliable delivery to downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the simplicity of the operation, which primarily involves mixing, heating, and filtration. The absence of complex distillation steps for solvent removal reduces the safety risks associated with handling large volumes of hot solvents. Furthermore, the environmental footprint is significantly reduced by avoiding the use of reproductive toxins like DMF and minimizing waste generation. This aligns with modern green chemistry principles and helps manufacturers meet increasingly stringent environmental regulations, facilitating smoother regulatory approvals and community acceptance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is crucial for evaluating the feasibility of integrating this route into existing production lines.
Q: Why is THF preferred over DMF for this alkylation reaction?
A: THF is preferred because it is compatible with subsequent organometallic reactions, such as Grignard additions, which require anhydrous conditions. Unlike DMF or acetone, THF does not need to be removed before downstream processing, allowing for efficient in-situ usage.
Q: What is the role of the phase transfer agent in this synthesis?
A: The phase transfer agent, typically tetrabutylammonium bromide, facilitates the transport of the carbonate anion into the organic phase, significantly accelerating the nucleophilic substitution rate and improving overall conversion yields.
Q: Can the product be isolated as a solid immediately?
A: Yes, the product can be isolated via aqueous work-up and crystallization, but the patent highlights the advantage of using the crude THF solution directly for subsequent steps to avoid time-consuming drying and purification processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Cyclopentyloxy-4-Methoxybenzaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient intermediate synthesis plays in the broader drug development timeline. Our technical team has extensively analyzed the pathway described in CN1946665A and possesses the expertise to implement this THF-based coupling strategy at scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless. Our facility is equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-cyclopentyloxy-4-methoxybenzaldehyde meets the exacting standards required for pharmaceutical applications.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can benefit your specific project requirements. By leveraging our process development capabilities, we can provide a Customized Cost-Saving Analysis tailored to your volume needs. We encourage you to request specific COA data and route feasibility assessments to verify our capability to deliver high-quality intermediates consistently. Let us collaborate to accelerate your pipeline with reliable, cost-effective chemical solutions.
