Scalable Production of Optically Active Imidazole Aromatic Alcohol Derivatives for Advanced Antifungal Therapeutics
The pharmaceutical industry is witnessing a paradigm shift towards single-enantiomer drugs, driven by the need for higher efficacy and reduced toxicity profiles. Patent CN1765887A introduces a groundbreaking preparation method for optically active imidazole aromatic alcohol derivatives, specifically targeting the synthesis of high-purity antifungal intermediates. This technology leverages a sophisticated combination of classical organic synthesis and modern biocatalysis to produce (R)- and (S)-α-alkoxy-β-(1-imidazolyl)-2,4-difluoroethylbenzene nitrates. Unlike traditional racemic syntheses that require cumbersome separation processes, this patented route integrates an enzymatic kinetic resolution step that significantly enhances stereochemical control. For R&D directors and process chemists, this represents a critical advancement in accessing chiral building blocks that serve as precursors to next-generation azole antifungals, potentially surpassing the activity of established drugs like econazole and miconazole.
The demand for reliable pharmaceutical intermediates supplier partnerships has never been higher, especially for complex chiral molecules where impurity profiles can dictate regulatory approval. The methodology outlined in this patent provides a robust framework for manufacturing these critical substances. By utilizing readily available starting materials such as m-difluorobenzene and employing efficient catalytic systems, the process addresses key pain points in cost reduction in antifungal drug manufacturing. The ability to selectively produce the biologically active (R)-isomer not only improves the therapeutic index but also streamlines the downstream purification requirements, offering substantial value to procurement teams looking to optimize their supply chains for high-value active pharmaceutical ingredients (APIs).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral imidazole derivatives has relied heavily on non-selective chemical reactions followed by resolution via diastereomeric salt formation or preparative chiral chromatography. These conventional approaches are fraught with inefficiencies; chemical resolution often suffers from a maximum theoretical yield of 50% for the desired enantiomer, with the remaining material requiring costly recycling or disposal. Furthermore, the use of stoichiometric amounts of chiral resolving agents increases raw material costs and generates significant chemical waste, complicating environmental compliance. Traditional methods may also involve harsh reaction conditions or heavy metal catalysts that leave difficult-to-remove residues, posing risks to product purity and patient safety. For supply chain managers, these factors translate into longer lead times, higher production costs, and increased vulnerability to raw material price fluctuations associated with specialized chiral reagents.
The Novel Approach
The novel approach detailed in the patent revolutionizes this landscape by introducing an organic phase lipase-catalyzed transesterification for kinetic resolution. This biocatalytic step operates under mild conditions, typically between 20°C and 65°C, preserving the integrity of sensitive functional groups while achieving high enantiomeric excess (ee) values. By coupling this biological specificity with efficient phase transfer catalysis (PTC) for subsequent alkylation steps, the process eliminates the need for expensive chiral columns or stoichiometric resolving agents. The integration of PTC using quaternary ammonium salts or polyethylene glycols facilitates reactions in biphasic systems, enhancing mass transfer and reaction rates. This streamlined workflow not only improves overall yield but also simplifies the isolation of the final nitrate salts, directly contributing to commercial scale-up of complex pharmaceutical intermediates with greater economic viability and environmental sustainability.
Mechanistic Insights into Lipase-Catalyzed Kinetic Resolution and Alkylation
The core of this synthetic strategy lies in the precise orchestration of stereoselective transformations. The process begins with a Friedel-Crafts acylation of m-difluorobenzene using chloroacetyl chloride and anhydrous aluminum trichloride, yielding 2-chloro-2',4'-difluoroacetophenone. This ketone is subsequently reduced using sodium borohydride (NaBH4) in alcoholic solvents to generate the racemic alcohol intermediate. The pivotal step occurs when this racemic mixture is subjected to lipase-catalyzed transesterification with vinyl acetate. The enzyme, such as Alcaligenes sp. or Pseudomonas stutzeri lipase, selectively acetylates one enantiomer (typically the (S)-form) while leaving the other ((R)-form) unreacted. This differential reactivity allows for the physical separation of the acetate ester and the free alcohol via column chromatography, effectively resolving the racemate. The separated acetate can then be hydrolyzed back to the alcohol, providing access to both optical isomers with high optical purity.
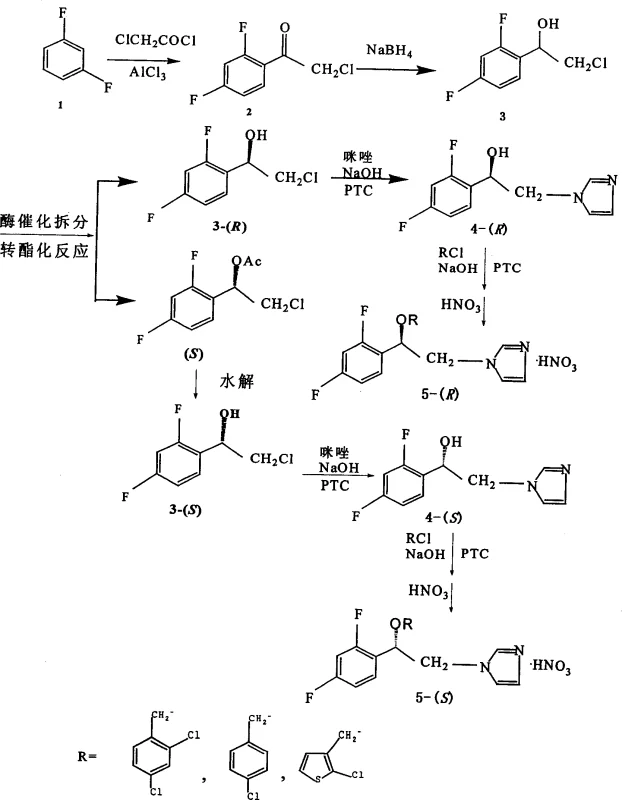
Following resolution, the optically active alcohol undergoes N-alkylation with imidazole. This step is crucial for establishing the pharmacophore responsible for antifungal activity. Conducted in aprotic solvents like tetrahydrofuran (THF) or DMF with sodium hydroxide and a phase transfer catalyst, this reaction ensures efficient nucleophilic substitution. The final O-alkylation introduces the substituted benzyl group (e.g., 2,4-dichlorobenzyl), completing the molecular architecture. The use of phase transfer catalysts here is mechanistically significant as they shuttle hydroxide ions into the organic phase, accelerating the deprotonation of the alcohol and facilitating the SN2 attack on the benzyl chloride. Finally, treatment with concentrated nitric acid converts the free base into the stable nitrate salt, which is the preferred form for pharmaceutical formulation due to its improved solubility and stability characteristics.
How to Synthesize Optically Active Imidazole Derivatives Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly during the enzymatic resolution and alkylation phases. The process is designed to be modular, allowing for the optimization of each step independently before integration into a continuous flow or batch process. Maintaining strict control over temperature and moisture levels is essential, especially during the Friedel-Crafts acylation and the enzymatic steps, to prevent side reactions and ensure high catalyst turnover. The following guide outlines the standardized operational sequence derived from the patent examples, providing a roadmap for technical teams aiming to replicate this high-efficiency pathway.
- Perform Friedel-Crafts acylation of m-difluorobenzene with chloroacetyl chloride using anhydrous aluminum trichloride to form 2-chloro-2',4'-difluoroacetophenone.
- Reduce the ketone intermediate using sodium borohydride in methanol or ethanol to obtain racemic 2-chloro-1-(2,4-difluorophenyl)-ethanol.
- Execute enzymatic kinetic resolution using lipase (e.g., Alcaligenes sp.) and vinyl acetate in an organic solvent to separate (R)- and (S)-isomers via transesterification.
- Conduct N-alkylation of the resolved chiral alcohol with imidazole under phase transfer catalysis conditions using quaternary ammonium salts.
- Complete the synthesis via O-alkylation with substituted benzyl chlorides followed by salt formation with nitric acid to yield the final nitrate derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic resolution technology offers distinct strategic advantages beyond mere technical feasibility. The shift from stoichiometric chemical resolution to catalytic enzymatic processes fundamentally alters the cost structure of production. By eliminating the need for large quantities of chiral resolving agents and reducing solvent consumption through more efficient work-ups, the overall cost of goods sold (COGS) is significantly lowered. Furthermore, the reliance on commodity chemicals like m-difluorobenzene and chloroacetyl chloride ensures a stable and predictable supply base, mitigating the risks associated with sourcing exotic or proprietary reagents. This stability is crucial for maintaining consistent production schedules and meeting the rigorous delivery timelines demanded by global pharmaceutical clients.
- Cost Reduction in Manufacturing: The implementation of lipase-catalyzed resolution drastically reduces the dependency on expensive chiral auxiliaries and heavy metal catalysts. Since enzymes are used in catalytic amounts and can often be immobilized for reuse, the recurring cost of the chiral induction agent is minimized. Additionally, the high selectivity of the enzyme reduces the formation of unwanted by-products, thereby lowering the costs associated with waste treatment and purification. This leaner manufacturing approach translates into substantial cost savings that can be passed down the supply chain, enhancing the competitiveness of the final antifungal product in the marketplace.
- Enhanced Supply Chain Reliability: The synthetic route utilizes robust, widely available starting materials that are not subject to the same supply constraints as specialized chiral pool resources. The reaction conditions are moderate and do not require extreme cryogenic temperatures or high-pressure equipment, reducing the risk of unplanned downtime due to equipment failure. This operational resilience ensures a continuous and reliable supply of high-purity chiral intermediates, allowing manufacturers to build inventory buffers and respond swiftly to fluctuations in market demand without compromising on quality or delivery commitments.
- Scalability and Environmental Compliance: The process is inherently scalable, with reaction parameters that translate well from kilogram to multi-ton scales. The use of aqueous work-ups and the avoidance of toxic heavy metals simplify the effluent treatment process, aligning with increasingly stringent environmental regulations. The ability to recycle solvents and the reduced generation of hazardous waste contribute to a greener manufacturing footprint. This environmental compliance not only avoids regulatory penalties but also enhances the brand reputation of the manufacturer as a sustainable partner, a factor that is becoming increasingly important in vendor selection criteria for major pharmaceutical companies.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its potential for integration into their existing portfolios. The following questions address common concerns regarding the practical application, purity standards, and scalability of the described method. These insights are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: How does the enzymatic resolution method improve purity compared to traditional chemical resolution?
A: The patented enzymatic kinetic resolution utilizes specific lipases (such as Alcaligenes sp. or Pseudomonas stutzeri) which exhibit high stereoselectivity. This biological specificity allows for the precise differentiation of enantiomers under mild conditions, achieving ee values exceeding 90% without the need for complex diastereomeric salt crystallization or heavy metal chiral auxiliaries often required in conventional chemical methods.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for scalability. Key steps such as the Friedel-Crafts acylation and the subsequent alkylation reactions utilize standard industrial reagents and operate at moderate temperatures (50°C-80°C). The use of phase transfer catalysts enhances reaction efficiency in biphasic systems, simplifying work-up procedures and making the transition from laboratory to multi-ton production feasible.
Q: What are the primary advantages of the (R)-isomer over the (S)-isomer in antifungal applications?
A: According to pharmacological data associated with this class of compounds, the (R)-isomers generally demonstrate superior antifungal activity against a broad spectrum of pathogens, including Candida albicans and Aspergillus fumigatus. Producing the single optically active (R)-form eliminates the metabolic burden and potential toxicity associated with the inactive (S)-enantiomer, leading to a safer and more efficacious therapeutic profile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Imidazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of precision and reliability in the production of chiral pharmaceutical intermediates. Our state-of-the-art facilities are equipped to handle the complex requirements of enzymatic resolutions and phase transfer catalysis, ensuring that every batch meets the highest standards of quality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our deep expertise in process chemistry to optimize yields and minimize impurities. Our rigorous QC labs employ advanced analytical techniques to verify stereochemical purity and identity, guaranteeing that our stringent purity specifications are consistently met for every shipment.
We invite global partners to collaborate with us to leverage this advanced synthesis technology for their antifungal drug development programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
