Advanced Cobalt-Catalyzed Oxidative Coupling Technology for Scalable Pharmaceutical Intermediate Production
Advanced Cobalt-Catalyzed Oxidative Coupling Technology for Scalable Pharmaceutical Intermediate Production
The landscape of fine chemical synthesis is undergoing a paradigm shift towards greener, more atom-economical processes, a transition vividly exemplified by the technology disclosed in patent CN110759814B. This pivotal intellectual property introduces a robust method for the oxidative coupling of 2,4-di-tert-butylphenol to produce 3,3',5,5'-tetra-tert-butyl biphenyl-2,2'-diphenol, a critical intermediate in the value chain of hydroformylation catalysts. By leveraging a cobalt-based catalytic system activated by sodium picolinate, this innovation replaces hazardous stoichiometric oxidants with benign air or oxygen, operating efficiently in an aqueous medium. For R&D directors and procurement strategists alike, this represents a significant leap forward in process intensification, offering a pathway to high-purity intermediates while simultaneously addressing the escalating regulatory pressures regarding industrial waste discharge and solvent usage in pharmaceutical and agrochemical manufacturing sectors.
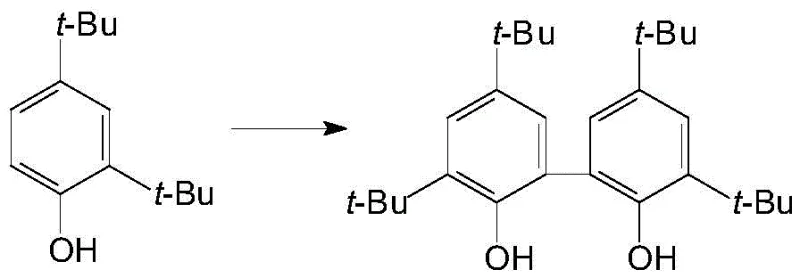
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sterically hindered biphenyl diphenols has been plagued by significant environmental and operational inefficiencies inherent to legacy technologies. One prominent prior art method, such as that described in U.S. Pat. No. 4,438,067, relies on the use of hydrogen peroxide as an oxidant within a highly alkaline aqueous solution of sodium hydroxide. While this route can achieve reasonable conversion, it generates copious amounts of alkali-containing waste liquid and requires extensive washing procedures to remove inorganic salts from the final product, creating a substantial burden on wastewater treatment facilities. Another conventional approach utilizes copper complexes in organic solvents like isopropanol, which necessitates complex post-reaction extraction protocols to separate the product from the solvent and catalyst residues. These traditional pathways not only inflate the cost of goods sold through excessive solvent consumption and energy-intensive purification steps but also introduce variability in product quality due to the difficulty in completely removing trace metal contaminants and organic impurities.
The Novel Approach
In stark contrast, the novel cobalt-catalyzed oxidative coupling method delineated in the patent data offers a streamlined, environmentally benign alternative that fundamentally restructures the reaction engineering profile. By employing a catalytic amount of cobalt salt combined with sodium picolinate as a ligand, the system activates molecular oxygen from air to drive the coupling reaction under mild thermal conditions ranging from 70°C to 100°C. This approach eliminates the need for stoichiometric quantities of hazardous oxidants and avoids the use of volatile organic compounds as reaction media, utilizing deionized water instead. The result is a process that not only achieves exceptional yields, documented in the examples to range from 93% to 99%, but also simplifies the isolation procedure to a mere filtration and water wash. This drastic reduction in unit operations translates directly into lower capital expenditure for equipment and reduced operational complexity, making it an attractive option for reliable pharmaceutical intermediate supplier networks seeking to optimize their manufacturing footprints.
Mechanistic Insights into Cobalt-Picolinate Catalyzed Oxidative Coupling
The efficacy of this transformation hinges on the precise coordination chemistry between the cobalt center and the picolinate ligand, which creates a highly active catalytic species capable of mediating single-electron transfer processes. Upon mixing the cobalt salt, such as cobalt acetate or cobalt chloride, with sodium picolinate in the aqueous phase, an in situ complex is formed that possesses the appropriate redox potential to abstract a hydrogen atom from the phenolic hydroxyl group of the 2,4-di-tert-butylphenol substrate. This abstraction generates a resonance-stabilized phenoxy radical, which, due to the steric bulk of the tert-butyl groups at the ortho and para positions, is directed specifically towards coupling at the unsubstituted ortho position relative to the hydroxyl group. The presence of the picolinate ligand is critical as it prevents the aggregation of cobalt species into inactive oxides and modulates the electron density at the metal center, ensuring that the radical generation rate is balanced with the coupling rate to minimize the formation of polymeric byproducts or quinone methide derivatives.
Furthermore, the mechanism inherently supports superior impurity control, a factor of paramount importance for R&D teams focused on the purity profiles of downstream catalyst ligands. The use of water as a solvent plays a dual role: it acts as a heat sink to manage the exothermic nature of the oxidation and facilitates the precipitation of the highly hydrophobic biphenyl product as it forms. This spontaneous phase separation drives the equilibrium towards completion according to Le Chatelier's principle and physically isolates the product from the aqueous catalytic phase, thereby preventing over-oxidation to quinones. The selectivity observed, where 3,3',5,5'-tetra-tert-butyl biphenyl-2,2'-diphenol is obtained with such high fidelity, suggests that the steric constraints imposed by the four tert-butyl groups, combined with the specific geometry of the cobalt-picolinate active site, effectively block alternative coupling modes. This mechanistic elegance ensures that the crude product requires minimal purification, directly addressing the stringent purity specifications required for high-performance ligand applications in homogeneous catalysis.
How to Synthesize 3,3',5,5'-tetra-tert-butyl biphenyl-2,2'-diphenol Efficiently
The implementation of this synthetic route is designed for operational simplicity, allowing for seamless integration into existing batch reactor infrastructure without the need for specialized high-pressure equipment. The process begins with the suspension of the phenolic starting material in water, followed by heating to ensure melting and dispersion, after which the catalytic system is introduced. The reaction is sustained by the continuous sparging of air or oxygen, maintaining a steady state of oxidant availability that matches the consumption rate of the substrate.
- Mix deionized water and 2,4-di-tert-butylphenol, heat to 70-100°C, then add cobalt salt and sodium picolinate.
- Introduce air or oxygen under normal pressure stirring and maintain temperature at 70-100°C for 3-12 hours.
- Filter the reaction mixture, wash the filter cake with deionized water until neutral, and dry to obtain the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this cobalt-catalyzed technology presents a compelling value proposition centered on cost structure optimization and risk mitigation. The transition from organic solvents to water as the primary reaction medium eliminates the volatility risks associated with flammable solvents and removes the recurring cost of solvent purchase, recovery, and disposal. Moreover, the reliance on air as the terminal oxidant decouples the process from the supply chain fluctuations of expensive chemical oxidants like hydrogen peroxide or hypochlorites, ensuring a stable and virtually limitless supply of the key reagent. This resilience is critical for maintaining continuous production schedules and avoiding downtime caused by raw material shortages, thereby enhancing the overall reliability of the supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the downstream processing workflow. By eliminating the need for extensive organic extractions and the neutralization of large volumes of alkaline waste, the operational expenditure associated with labor, energy, and waste treatment is significantly curtailed. The high yield profile, consistently reaching near-quantitative levels in optimized examples, ensures maximum atom economy, meaning that less raw material is wasted per unit of product produced. Additionally, the avoidance of precious metal catalysts or complex ligand systems in favor of abundant cobalt salts further depresses the direct material costs, allowing for a more competitive pricing structure in the final commercial offering of these specialized intermediates.
- Enhanced Supply Chain Reliability: From a logistics perspective, the use of non-hazardous, non-flammable materials simplifies the storage and handling requirements within the manufacturing facility. Water and air do not require the same level of regulatory compliance and safety infrastructure as organic solvents or strong oxidizers, reducing the administrative burden and insurance costs associated with chemical inventory. The robustness of the reaction conditions, which tolerate a range of temperatures and catalyst loadings without significant loss of performance, provides a buffer against minor process deviations, ensuring consistent batch-to-batch quality. This stability is essential for long-term supply agreements where consistency is valued as highly as price, fostering stronger partnerships between manufacturers and their downstream clients in the fine chemical sector.
- Scalability and Environmental Compliance: The environmental footprint of this manufacturing route is markedly smaller than that of conventional methods, aligning with the increasingly strict global regulations on industrial emissions and effluent quality. The aqueous waste stream generated is relatively clean compared to the salt-laden brines of alkaline processes or the solvent-contaminated streams of organic syntheses, simplifying wastewater treatment and reducing the risk of regulatory non-compliance penalties. Furthermore, the scalability of the process is enhanced by the absence of mass transfer limitations often encountered in multiphase organic reactions; the simple gas-liquid-solid interaction in water allows for straightforward scale-up from pilot plant to full commercial production volumes. This ease of scale-up ensures that supply can be rapidly ramped up to meet market demand without the need for prolonged process re-engineering or validation cycles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this oxidative coupling technology. These insights are derived directly from the experimental data and technical disclosures within the patent literature, providing a factual basis for decision-making.
Q: What are the primary advantages of the cobalt-catalyzed method over traditional alkaline oxidation?
A: Unlike traditional methods using hydrogen peroxide in alkaline solutions which generate massive amounts of salt-containing wastewater, this cobalt-catalyzed process utilizes water as the sole solvent and air as the oxidant. This drastically simplifies post-treatment by eliminating the need for extensive washing to remove alkali salts, thereby reducing environmental burden and processing time.
Q: How does the sodium picolinate ligand influence the reaction selectivity?
A: Sodium picolinate acts as a crucial auxiliary ligand that coordinates with the cobalt center to form an active catalytic species in situ. This coordination environment modulates the redox potential of the cobalt, facilitating the selective generation of phenoxy radicals while suppressing non-selective polymerization or over-oxidation side reactions, leading to yields exceeding 90%.
Q: Is this process suitable for large-scale industrial production of ligand precursors?
A: Yes, the process is highly scalable due to its use of normal pressure conditions and inexpensive oxidants like air. The absence of hazardous organic solvents and the simplicity of the work-up procedure (filtration and water washing) make it ideal for commercial scale-up, ensuring consistent supply continuity for downstream hydroformylation catalyst manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3',5,5'-tetra-tert-butyl biphenyl-2,2'-diphenol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is not merely a regulatory requirement but a strategic imperative for maintaining competitiveness in the global fine chemicals market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the cobalt-catalyzed oxidative coupling process are fully realized in a commercial setting. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 3,3',5,5'-tetra-tert-butyl biphenyl-2,2'-diphenol performs reliably in your downstream catalytic applications. Our infrastructure is designed to handle the specific nuances of aqueous-phase oxidations, providing a secure and compliant manufacturing environment for high-value intermediates.
We invite you to collaborate with us to leverage this advanced technology for your specific supply chain needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this greener route for your specific volume requirements. We encourage you to reach out today to discuss your project specifics,索取 specific COA data, and review our comprehensive route feasibility assessments. Let us partner with you to build a more sustainable and cost-effective supply chain for your critical pharmaceutical and agrochemical intermediates.
