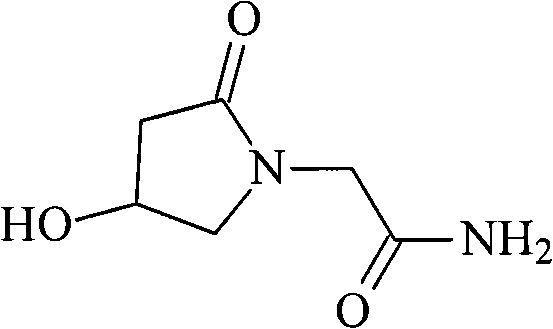
Advanced Purification Technology for High-Purity Oxiracetam Pharmaceutical Intermediates
The pharmaceutical landscape for neuroprotective agents demands uncompromising standards of purity, particularly for compounds targeting central nervous system disorders like senile dementia. Patent CN102050774A introduces a groundbreaking purification methodology for Oxiracetam, a potent nootropic agent known chemically as 2-(4-hydroxy-2-oxopyrrolidin-1-yl)acetamide. This innovation addresses the critical limitations of existing manufacturing processes which often struggle to eliminate trace impurities that compromise clinical safety. The disclosed technique leverages a synergistic combination of activated carbon adsorption, precise organic solvent precipitation, and a specialized neutral alumina column chromatography system. By optimizing parameters such as mobile phase composition and column temperature, this method achieves a dramatic enhancement in product quality, ensuring that the final active pharmaceutical ingredient meets the rigorous specifications required for treating cognitive decline and organic brain syndromes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis and purification of Oxiracetam have been plagued by significant technical hurdles that hinder large-scale commercial viability. Prior art methods, such as those disclosed in WO2005/115978 and various Japanese patents, often rely on harsh alkaline conditions that inadvertently degrade the sensitive Oxiracetam molecule, leading to substantially reduced yields. Furthermore, traditional routes frequently necessitate the use of protecting groups for hydroxyl functionalities, which introduces additional synthetic steps, increases raw material consumption, and prolongs production cycles to over twenty hours. The reliance on silica gel or macroporous resins for purification in these older methods presents further challenges; silica gel can exhibit inconsistent separation efficiency, while macroporous resins are prone to physical fragmentation and the leaching of pore-creating agents, causing secondary pollution that is difficult to remove. These inefficiencies result in a final product with purity levels insufficient for direct clinical application, necessitating costly and time-consuming reprocessing.
The Novel Approach
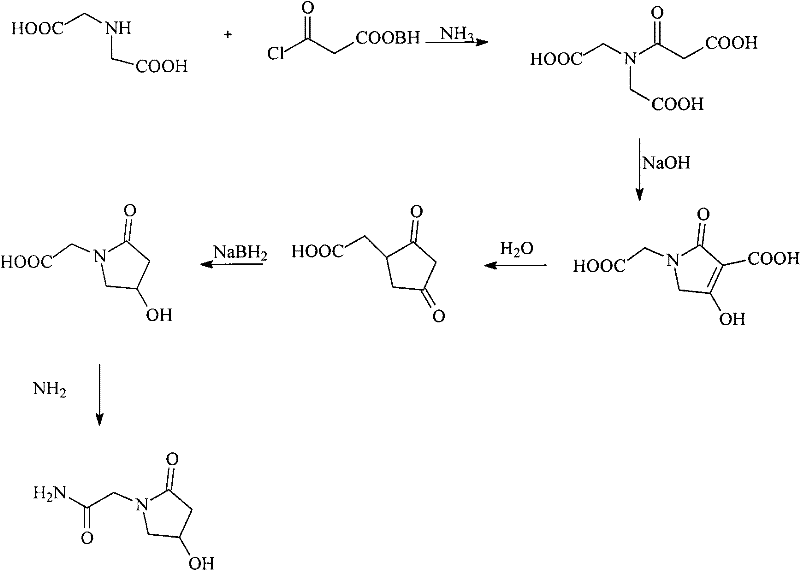
In stark contrast, the novel approach detailed in the patent data streamlines the purification workflow into a robust, four-stage process that eliminates the need for complex protecting group chemistry. The method initiates with a controlled activated carbon adsorption step at moderate temperatures (50-60°C), effectively removing colored impurities without degrading the active compound. This is followed by a strategic precipitation using a specific ratio of acetonitrile and isopropyl ether, which isolates the bulk solid before the critical chromatographic separation. The core innovation lies in the utilization of a neutral alumina column rather than traditional silica or ion-exchange resins. This choice of stationary phase, combined with a mobile phase of acetonitrile and water in a 1:3 volume ratio, provides exceptional selectivity for Oxiracetam, allowing for the collection of fractions with content greater than 90 percent. This streamlined pathway not only simplifies the operational complexity but also drastically improves the overall yield and purity profile of the final drug substance.
Mechanistic Insights into Alumina Column Chromatography Purification
The efficacy of this purification strategy is rooted in the specific physicochemical interactions between the Oxiracetam molecule and the neutral alumina stationary phase. Unlike acidic silica gel, which can potentially catalyze the hydrolysis of the lactam ring or the amide bond under certain conditions, neutral alumina offers a chemically inert environment that preserves the structural integrity of the gamma-aminobutyric acid derivative. The patent data highlights that the particle size of the alumina (100-200 mesh or 200-300 mesh) and the column dimensions are critically optimized to maximize surface area contact while maintaining an optimal flow rate of 2.5 to 5.0 ml/min. This balance ensures that the residence time of the solute is sufficient for the differential adsorption of impurities versus the target molecule, facilitating a sharp separation boundary. The use of an acetonitrile-water mobile phase further enhances this selectivity by modulating the polarity of the system, ensuring that polar impurities are retained or eluted separately from the highly polar Oxiracetam.
Impurity control is further reinforced by the initial activated carbon treatment, which acts as a coarse filter for high molecular weight byproducts and colored tars that could otherwise foul the chromatography column. The subsequent precipitation step serves as a secondary purification barrier, removing bulk salts and highly soluble contaminants before the fine separation occurs. By merging only those eluent fractions where the medicament content exceeds 90 percent, the process effectively cuts off the 'tails' of the chromatographic peak where impurities typically concentrate. This rigorous fractionation protocol, combined with a final recrystallization step using the same organic solvent system, ensures that the resulting crystal lattice is free from solvent inclusions and lattice defects, thereby guaranteeing the high stability and bioavailability required for a central nervous system therapeutic agent.
How to Synthesize Oxiracetam Efficiently
The synthesis and purification protocol outlined in the patent represents a significant leap forward in process chemistry for nootropic drugs. It transforms a previously cumbersome multi-step refinement into a scalable, continuous operation suitable for industrial manufacturing. The following guide summarizes the critical operational parameters derived from the patent examples, focusing on the precise control of temperature, solvent ratios, and flow rates necessary to replicate the high-yield results. For a comprehensive understanding of the laboratory-to-plant scale-up, operators must adhere strictly to the specified mass ratios of crude drug to chromatographic filler, as this directly influences the resolution of the separation.
- Dissolve crude Oxiracetam in purified water and perform activated carbon adsorption at 50-60°C to remove color and impurities.
- Add an organic solvent mixture (Acetonitrile: Isopropyl ether) to the filtrate to induce precipitation of the solid product.
- Perform chromatographic separation using a neutral alumina column with an Acetonitrile: Water mobile phase to isolate high-purity fractions.
- Evaporate the eluent, add organic solvent for final crystallization, and dry under reduced pressure to obtain the pure compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this purification technology translates into tangible strategic benefits that extend beyond mere technical specifications. The elimination of expensive ion-exchange resins and the reduction in processing steps directly correlate to a significant reduction in manufacturing overheads. By avoiding the regeneration and activation protocols required for ion-exchange columns, the facility can operate with lower utility consumption and reduced chemical waste disposal costs. Furthermore, the robustness of the neutral alumina column, which does not suffer from the fragmentation issues common to macroporous resins, ensures longer column life and reduced downtime for column packing and maintenance. This operational stability is crucial for maintaining consistent supply schedules in a high-demand pharmaceutical market.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by replacing high-cost ion-exchange resins with economically viable neutral alumina, which is readily available and requires less complex handling. The simplified workflow removes the need for multiple protection and deprotection steps found in older synthetic routes, thereby reducing raw material consumption and labor hours per batch. Additionally, the high recovery rate of the active pharmaceutical ingredient minimizes waste generation, ensuring that a greater proportion of the input material is converted into saleable high-purity product, effectively lowering the cost of goods sold.
- Enhanced Supply Chain Reliability: The use of common, non-hazardous solvents like acetonitrile and isopropyl ether, combined with stable stationary phases, mitigates the risk of supply disruptions associated with specialized or regulated reagents. The method's tolerance for variation in crude feedstock quality, due to the powerful cleaning capacity of the activated carbon and alumina steps, ensures that production can continue even if upstream synthesis batches vary slightly in impurity profiles. This resilience creates a more predictable production timeline, allowing for better inventory planning and reduced lead times for fulfilling large-scale orders from global pharmaceutical partners.
- Scalability and Environmental Compliance: The chromatographic conditions described are inherently scalable, with column dimensions and flow rates that can be linearly adjusted from pilot plant to full commercial production without losing separation efficiency. The process generates significantly less hazardous waste compared to methods requiring strong bases or chlorinated solvents for extraction, aligning with modern green chemistry principles and stringent environmental regulations. This compliance reduces the regulatory burden on the manufacturing site and lowers the costs associated with waste treatment and environmental monitoring, making the entire supply chain more sustainable and future-proof.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on how this method outperforms traditional techniques in terms of purity, yield, and operational safety. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing lines.
Q: Why is neutral alumina preferred over silica gel for Oxiracetam purification?
A: Neutral alumina provides superior isolation effects for this specific compound compared to silica gel or macroporous resins. It avoids the rigidity and fragmentation issues associated with resins and prevents the secondary pollution often caused by pore-creating agents, while maintaining a lower cost profile than ion exchange columns.
Q: What purity levels can be achieved with this patented purification method?
A: The described process consistently achieves purity levels exceeding 99.5 percent, with specific embodiments demonstrating purity up to 99.9 percent. This significant improvement over prior art ensures the removal of related substances that could cause toxic side effects in clinical applications.
Q: How does this method address the stability issues of Oxiracetam?
A: By utilizing mild reaction conditions (50-60°C) and avoiding strong alkaline environments that typically degrade Oxiracetam, this method preserves the structural integrity of the molecule. The efficient removal of impurities also prevents the discoloration and content reduction often seen during long-term storage of improperly purified batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxiracetam Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high-purity standards defined in CN102050774A are met consistently at every batch size. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the detection of trace related substances that could impact patient safety. We are committed to delivering Oxiracetam intermediates that not only meet but exceed the quality benchmarks required for the formulation of advanced nootropic therapies.
We invite global pharmaceutical partners to engage with our technical procurement team to discuss how this advanced purification technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of confidence and technical support.
