Advanced Manufacturing of Phenserine Analogs via Streamlined Physostigmine Hydrolysis
Advanced Manufacturing of Phenserine Analogs via Streamlined Physostigmine Hydrolysis
The pharmaceutical industry continuously seeks robust synthetic routes for acetylcholinesterase inhibitors, particularly for treating neurodegenerative conditions such as Alzheimer's disease. Patent CN1612880A introduces a transformative methodology for preparing phenserine and its analogs, addressing critical inefficiencies found in legacy manufacturing protocols. This innovation centers on the direct hydrolysis of physostigmine salts to form eseroline intermediates, bypassing the traditionally cumbersome conversion to free bases. By optimizing reaction conditions and solvent systems, this process achieves exceptional purity levels and yields, positioning it as a vital asset for reliable pharmaceutical intermediates supplier networks aiming to enhance production efficiency. The strategic elimination of redundant processing steps not only simplifies the workflow but also mitigates the risk of compound degradation, ensuring a consistent supply of high-quality active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phenserine from physostigmine has been plagued by operational complexities that severely impact overall process economics and product quality. Traditional methods necessitate the initial conversion of physostigmine salts into their corresponding free base forms before any hydrolysis can occur, introducing unnecessary unit operations that increase both time and material costs. Furthermore, the resulting oxyphysostigmine (eseroline) is highly susceptible to oxidative degradation, requiring stringent inert atmosphere controls and vacuum applications to maintain stability during isolation. These legacy protocols often involve multiple extraction and purification stages using water-immiscible solvents, which complicates waste management and reduces the final recovery rate of the desired intermediate. Consequently, manufacturers face significant challenges in scaling these processes while maintaining the rigorous purity standards required for clinical applications.
The Novel Approach
In stark contrast, the methodology disclosed in CN1612880A revolutionizes the production landscape by enabling the direct hydrolysis of physostigmine salts within an aqueous reaction medium. This approach obviates the need for free base conversion, thereby streamlining the initial reaction phase and reducing the potential for side reactions. The process leverages alkali metal hydroxides for hydrolysis followed by a novel pH adjustment strategy using alkali metal bisulfites, which effectively preserves the integrity of the sensitive eseroline intermediate. By operating within a specific pH range of 8.0 to 9.5, the method maximizes extraction efficiency while minimizing product loss, a critical improvement over previous techniques that suffered from poor yield retention. This streamlined pathway facilitates the isolation of high-purity intermediates ready for subsequent condensation, marking a significant leap forward in process chemistry optimization.
Mechanistic Insights into Alkaline Hydrolysis and Carbamate Formation
The core chemical transformation involves the nucleophilic attack facilitated by alkali metal hydroxides on the carbamate moiety of the physostigmine salt, leading to the cleavage of the carbamoyl group and the formation of the phenolic hydroxyl group in eseroline. Unlike conventional routes that struggle with solubility and stability issues in organic media, this reaction thrives in a hydrated environment where the salt form of the starting material is directly accessible. The use of water-immiscible ethers like tert-butyl methyl ether during the extraction phase allows for efficient phase separation without compromising the stability of the product. Crucially, the substitution of mineral acids with alkali metal bisulfites for pH neutralization prevents the formation of undesirable acid addition salts that could complicate downstream processing, ensuring that the intermediate remains in a chemically stable state suitable for immediate further reaction or storage.
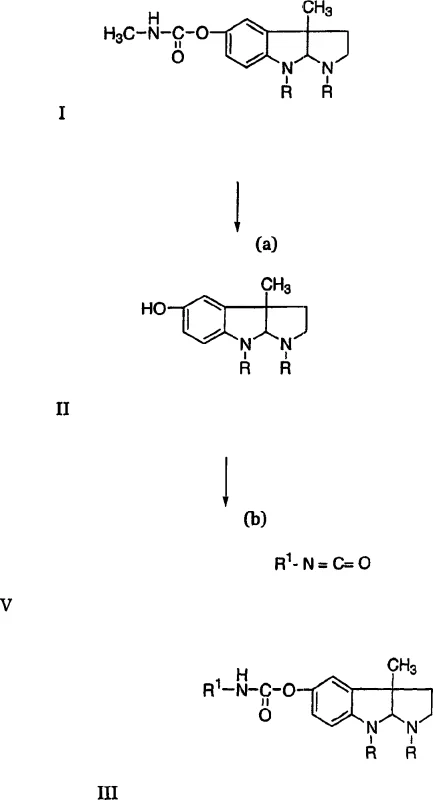
Following the isolation of the purified eseroline intermediate, the synthesis proceeds through a deprotonation-condensation sequence to generate the final phenserine structure. In an anhydrous aprotic solvent system, a strong organic base such as n-butyllithium is employed to deprotonate the phenolic hydroxyl group, generating a highly reactive nucleophile. This activated species then undergoes a rapid addition reaction with the chosen isocyanate derivative, forming the new carbamate linkage characteristic of phenserine analogs. The reaction conditions are meticulously controlled to maintain temperatures between 15°C and 30°C, preventing thermal degradation while ensuring complete conversion. The final product precipitates upon the addition of water, allowing for simple filtration and drying without the need for energy-intensive distillation or chromatographic purification, thus preserving the stereochemical integrity of the chiral centers throughout the synthesis.
How to Synthesize Phenserine Efficiently
The synthesis of phenserine via this patented route offers a clear pathway for laboratory and pilot-scale operations to achieve high reproducibility and yield. The process begins with the careful preparation of the aqueous hydrolysis mixture, ensuring that the stoichiometry of the alkali metal hydroxide is sufficient to drive the reaction to completion without excessive degradation. Operators must pay close attention to the pH adjustment phase, utilizing sodium metabisulfite or similar agents to precisely hit the target pH window for optimal extraction.
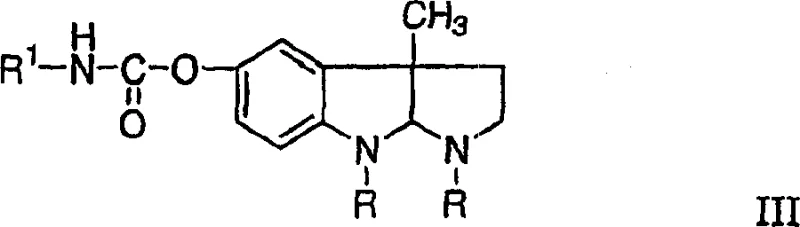
- Hydrolyze physostigmine salts (Formula I) directly in an aqueous medium using alkali metal hydroxide at controlled pH to generate eseroline (Formula II).
- Adjust the pH to 8.0-9.5 using alkali metal bisulfite to minimize product loss and extract the purified eseroline intermediate.
- React the purified eseroline with an isocyanate (Formula V) in an anhydrous aprotic solvent using a strong organic base catalyst to yield phenserine (Formula III).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain managers, the adoption of this synthesis route presents substantial opportunities for cost optimization and risk mitigation. By reducing the number of unit operations and eliminating the need for specialized inert atmosphere equipment during the hydrolysis step, the overall capital expenditure and operational overhead are significantly lowered. The ability to utilize common, commercially available solvents such as water, ethers, and alcohols reduces dependency on exotic reagents, thereby enhancing supply chain resilience against market volatility. Furthermore, the high purity of the intermediate obtained directly from the reaction mixture minimizes the need for extensive recrystallization or purification steps, leading to faster batch turnover times and improved throughput capabilities for manufacturing facilities.
- Cost Reduction in Manufacturing: The elimination of the free base conversion step and the simplification of workup procedures directly translate to reduced labor and utility costs per kilogram of product. By avoiding the use of mineral acids and complex neutralization protocols, the process reduces the consumption of auxiliary chemicals and the associated costs of waste disposal. The high yield reported in the examples demonstrates efficient atom economy, ensuring that raw material costs are optimized and that less starting material is required to produce the same amount of final API. This efficiency is critical for maintaining competitive pricing in the global market for Alzheimer's therapeutics.
- Enhanced Supply Chain Reliability: The robustness of the aqueous hydrolysis step ensures consistent production outcomes regardless of minor fluctuations in raw material quality, providing a stable supply of key intermediates. Since the process does not rely on highly sensitive reagents that require strict cold chain logistics or specialized handling, sourcing becomes more flexible and less prone to disruption. The scalability of the method allows manufacturers to ramp up production volumes rapidly in response to market demand without the need for extensive process re-validation, ensuring continuity of supply for downstream drug formulation partners.
- Scalability and Environmental Compliance: The use of water as a primary reaction medium aligns with green chemistry principles, reducing the environmental footprint associated with volatile organic compound emissions. Simplified isolation techniques that rely on precipitation and filtration rather than distillation lower energy consumption, contributing to more sustainable manufacturing practices. The reduction in hazardous waste generation, particularly from avoiding heavy metal catalysts or toxic solvents, simplifies regulatory compliance and lowers the costs associated with environmental health and safety management, making the process attractive for long-term commercial investment.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this patented synthesis method. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: What is the primary advantage of using physostigmine salts directly in this synthesis?
A: The process eliminates the need to convert physostigmine salts to the free base prior to hydrolysis, significantly reducing processing steps and preventing degradation associated with free base handling.
Q: How does the new method improve the purity of the eseroline intermediate?
A: By utilizing alkali metal bisulfite for pH adjustment instead of mineral acids, the method prevents adverse effects on yield and allows for extraction at a pH range of 8.0-9.5, achieving purities up to 99.9%.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the method is designed for economic large-scale production, utilizing common solvents like water and ethers, and avoiding complex isolation steps that typically hinder scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenserine Supplier
At NINGBO INNO PHARMCHEM, we leverage our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring this advanced synthesis technology to life. Our commitment to stringent purity specifications and rigorous QC labs ensures that every batch of phenserine intermediate meets the highest global standards for pharmaceutical applications. We understand the critical nature of supply consistency for life-saving medications and have built our infrastructure to support the complex requirements of modern drug manufacturing.
We invite potential partners to engage with our technical procurement team to discuss how this optimized process can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this streamlined method. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver high-quality phenserine analogs efficiently and reliably.
