Advanced Synthesis of Dihydropyrazole Thiazole Derivatives for Oncology Drug Development
Advanced Synthesis of Dihydropyrazole Thiazole Derivatives for Oncology Drug Development
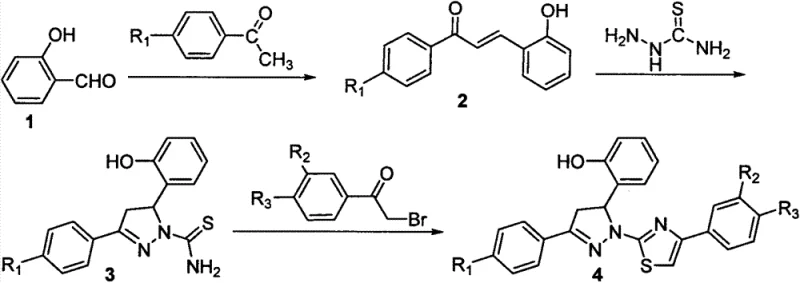
The pharmaceutical industry is constantly seeking robust, scalable, and high-yielding synthetic routes for novel heterocyclic compounds that exhibit potent biological activity. Patent CN103664926A introduces a significant breakthrough in the synthesis of dihydropyrazole thiazole derivatives, a class of compounds demonstrating remarkable inhibitory effects against human breast cancer cells (MCF-7), cervical cancer cells (Hela), and liver cancer cells (Hepg2). This technology represents a critical advancement for reliable pharmaceutical intermediate suppliers aiming to support the development of next-generation antitumor agents. By integrating a pyrazoline core with a thiazole moiety, the patented method creates a structurally diverse library of molecules with enhanced pharmacological potential. The process is characterized by its operational simplicity, utilizing readily available starting materials such as salicylaldehyde and substituted acetophenones, which ensures a stable supply chain foundation for downstream drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for constructing complex heterocyclic systems often suffer from significant drawbacks that hinder their commercial viability. Many conventional methods rely on harsh reaction conditions, including extreme temperatures or the use of toxic, expensive transition metal catalysts that require rigorous removal steps to meet stringent purity specifications for high-purity pharmaceutical intermediates. Furthermore, older methodologies frequently exhibit poor atom economy and low regioselectivity, leading to complex impurity profiles that are difficult to separate. These inefficiencies result in prolonged production cycles and inflated costs, creating bottlenecks for cost reduction in API manufacturing. Additionally, the multi-step nature of traditional routes often involves unstable intermediates that degrade during isolation, further reducing overall throughput and complicating the commercial scale-up of complex polymer additives or drug candidates.
The Novel Approach
The methodology disclosed in CN103664926A overcomes these historical challenges through a streamlined, three-step sequence that maximizes efficiency and minimizes waste. This novel approach utilizes a base-catalyzed Claisen-Schmidt condensation followed by a cyclization with thiosemicarbazide and a final ring-closing reaction with alpha-bromoacetophenones. This strategy avoids the use of heavy metal catalysts entirely, relying instead on mild alkaline conditions and thermal energy to drive the reactions forward. The result is a process that delivers white crystalline products with exceptional purity and yields consistently exceeding 87%, with some derivatives reaching up to 94.6%. This high level of efficiency directly translates to substantial cost savings by reducing raw material consumption and simplifying downstream processing. The robustness of this chemistry ensures that reducing lead time for high-purity intermediates becomes a tangible reality for procurement teams managing tight development schedules.
Mechanistic Insights into Base-Catalyzed Heterocyclic Construction
The core of this synthetic innovation lies in the precise orchestration of nucleophilic attacks and cyclization events that build the molecular architecture. The first step involves the formation of a chalcone derivative via the condensation of salicylaldehyde and a substituted acetophenone in an alkaline medium. This reaction proceeds through an enolate intermediate that attacks the carbonyl carbon of the aldehyde, followed by dehydration to establish the conjugated enone system. In the second stage, the chalcone undergoes a cyclocondensation with thiosemicarbazide. The nucleophilic nitrogen of the thiosemicarbazide attacks the beta-carbon of the enone system, initiating a cascade that closes the five-membered pyrazoline ring. This step is critical for establishing the stereochemical integrity and biological activity of the final molecule.
The final transformation involves the reaction of the dihydropyrazoline intermediate with an alpha-bromoacetophenone derivative. This step constructs the thiazole ring through a nucleophilic substitution followed by intramolecular cyclization. The sulfur atom of the thioamide group attacks the alpha-carbon of the bromoketone, displacing the bromide ion. Subsequent cyclization and dehydration finalize the fused heterocyclic system. This mechanism is highly advantageous because it tolerates a wide range of substituents (R1, R2, R3 groups such as H, F, CH3O, Br, CF3) without compromising yield. The absence of sensitive functional group incompatibilities allows for the rapid generation of diverse analog libraries, accelerating the structure-activity relationship (SAR) studies essential for modern drug discovery.
How to Synthesize Dihydropyrazole Thiazole Derivatives Efficiently
Implementing this synthesis requires careful control of reaction parameters to maintain the high yields reported in the patent data. The process begins with the preparation of the chalcone intermediate, where stoichiometry and temperature must be optimized to prevent polymerization side reactions. Following isolation, the pyrazoline cyclization is performed under reflux conditions, monitored closely by TLC to ensure complete conversion of the starting chalcone. The final thiazole formation is conducted in polar aprotic solvents like DMF to solubilize the intermediates effectively. While the general principles are straightforward, precise execution is key to achieving the reported 90%+ yields. For detailed operational parameters, stoichiometry ratios, and specific workup protocols, please refer to the standardized guide below.
- Perform Claisen-Schmidt condensation between salicylaldehyde and substituted acetophenone in alkaline solution to form chalcone intermediate.
- React the chalcone derivative with thiosemicarbazide in organic solvent under heating to cyclize into the dihydropyrazoline core.
- Condense the dihydropyrazoline intermediate with alpha-bromoacetophenone derivatives to finalize the thiazole ring structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers transformative benefits that extend beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the purification process. Since the reactions proceed with high selectivity and yield, the crude products often require only simple filtration and recrystallization from ethanol to achieve analytical purity. This eliminates the need for resource-intensive column chromatography on a large scale, significantly lowering the operational expenditure (OpEx) associated with solvent consumption and silica gel disposal. Furthermore, the starting materials—salicylaldehydes and acetophenones—are commodity chemicals available from multiple global vendors, mitigating the risk of single-source supply disruptions and ensuring enhanced supply chain reliability.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the costly and time-consuming step of heavy metal scavenging, which is often a regulatory requirement for pharmaceutical ingredients. Additionally, the high atom economy of the condensation reactions means that a greater proportion of raw materials are incorporated into the final product, reducing waste disposal costs. The ability to perform reactions at moderate temperatures also lowers energy consumption compared to high-pressure or cryogenic alternatives. These factors combine to create a leaner, more cost-effective manufacturing profile that improves margin potential for the final API.
- Enhanced Supply Chain Reliability: The robustness of the chemistry allows for flexible production scheduling. Because the intermediates are stable and the reaction conditions are not overly sensitive to minor fluctuations in temperature or moisture, the process is forgiving and reproducible across different batches. This consistency is vital for maintaining continuous supply to R&D partners and clinical trial manufacturers. The use of common solvents like DMF and ethanol further simplifies logistics, as these do not require specialized handling or storage infrastructure, facilitating smoother commercial scale-up of complex pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental perspective, this route is superior to many traditional methods. The absence of toxic heavy metals reduces the burden on wastewater treatment facilities and lowers the environmental footprint of the manufacturing site. The high yields mean less chemical waste is generated per kilogram of product, aligning with green chemistry principles. Scalability is inherently supported by the homogeneous nature of the reactions, which can be easily transferred from laboratory glassware to industrial stainless steel reactors without significant re-engineering. This ensures that reducing lead time for high-purity intermediates is achievable even when demand scales rapidly during late-stage clinical development.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its potential for their specific drug pipelines. The following questions address common concerns regarding purity, scalability, and regulatory compliance. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a transparent view of the technology's capabilities. By addressing these points early, we aim to facilitate faster decision-making and smoother collaboration between chemical suppliers and pharmaceutical developers.
Q: What is the typical yield for this dihydropyrazole thiazole synthesis?
A: According to patent CN103664926A, the process achieves exceptionally high yields ranging from 87.8% to 94.6% across various substituted derivatives, ensuring efficient material utilization.
Q: Are heavy metal catalysts required for this reaction pathway?
A: No, the described methodology relies on base-catalyzed condensation and thermal cyclization, eliminating the need for expensive transition metal catalysts and simplifying purification.
Q: Can this process be scaled for commercial API production?
A: Yes, the use of common organic solvents like DMF and ethanol, combined with simple workup procedures like filtration and recrystallization, makes the process highly amenable to kilogram-to-ton scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dihydropyrazole Thiazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of oncology drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project never faces supply bottlenecks. We are committed to delivering stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our expertise in heterocyclic chemistry allows us to optimize this specific patent route further, tailoring the process to meet your unique cost and timeline requirements while maintaining full regulatory compliance.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis can benefit your specific pipeline. Contact us today to request a Customized Cost-Saving Analysis tailored to your volume needs. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how we can become your strategic partner in bringing life-saving antitumor therapies to market efficiently and reliably.
