Advanced Metal-Free Synthesis of Polysubstituted Thiophenes for Commercial Scale Production
The chemical industry is currently witnessing a paradigm shift towards greener, more sustainable synthetic methodologies, particularly in the realm of heterocyclic chemistry where thiophene derivatives play a pivotal role. Patent CN111560006A introduces a groundbreaking approach for the preparation of polysubstituted thiophenes, addressing long-standing challenges related to metal contamination and operational complexity. This innovation leverages the reactivity of sulfur-containing ylides and electron-deficient alkynes to construct the thiophene core through a thermally driven cyclization process. By eliminating the reliance on expensive transition metal catalysts, this technology not only simplifies the purification workflow but also significantly enhances the environmental profile of the synthesis. For pharmaceutical and fine chemical manufacturers, this represents a critical advancement in securing reliable supply chains for high-value intermediates while adhering to stringent regulatory standards regarding residual metals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional strategies for synthesizing polysubstituted thiophenes often rely heavily on transition metal catalysis, such as palladium or copper-mediated cross-coupling reactions, which introduce significant downstream processing burdens. These conventional routes frequently necessitate the use of malodorous sulfur sources like hydrogen sulfide or sodium sulfide, posing severe safety hazards and environmental disposal challenges for production facilities. Furthermore, the presence of metal residues in the final product is a persistent issue that requires additional, costly purification steps such as scavenging or recrystallization to meet pharmaceutical grade specifications. The harsh reaction conditions often associated with these legacy methods, including extreme temperatures or sensitive inert atmospheres, further limit their practicality for large-scale commercial manufacturing. Consequently, there is an urgent industrial demand for alternative synthetic pathways that can bypass these inefficiencies while maintaining high structural diversity.
The Novel Approach
The methodology disclosed in the patent data offers a transformative solution by utilizing a metal-free thermal cyclization strategy that operates under remarkably mild and straightforward conditions. By employing sulfur ylides as key building blocks, the reaction proceeds efficiently in 1,2-dichloroethane (DCE) at a moderate temperature of 85°C, removing the need for complex catalytic systems. This approach allows for the direct assembly of tetra- and tri-substituted thiophenes with excellent functional group tolerance, including the incorporation of sensitive fluorinated moieties. The simplicity of the workup procedure, which involves basic concentration and chromatography, drastically reduces the operational time and resource consumption compared to multi-step metal-catalyzed sequences. This novel route effectively bridges the gap between academic innovation and industrial applicability, providing a robust platform for the scalable production of complex thiophene architectures.
Mechanistic Insights into Thermal Ylide-Alkyne Cyclization
The core of this synthetic breakthrough lies in the unique reactivity profile of the sulfur ylide intermediate, which acts as a 1,3-dipole in a formal [3+2] cycloaddition with the electron-deficient alkyne dipolarophile. Upon heating, the nucleophilic carbon of the ylide attacks the electrophilic triple bond of the alkyne, initiating a cascade of electronic rearrangements that ultimately lead to the formation of the thiophene ring system. This mechanism is driven purely by thermal energy, negating the requirement for external Lewis acids or transition metal complexes to activate the substrates. The electron-withdrawing groups (EWG) present on both the ylide and the alkyne play a crucial role in stabilizing the transition states and directing the regioselectivity of the cyclization. Understanding this mechanistic pathway is essential for process chemists aiming to optimize reaction parameters for specific substrate classes.
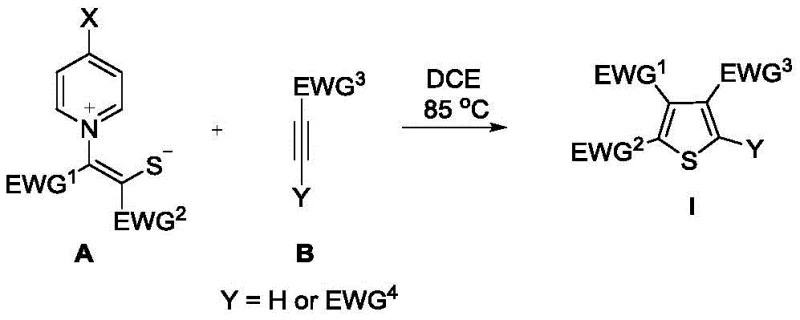
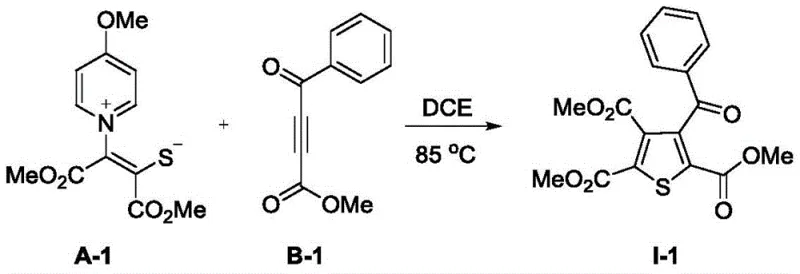
Furthermore, the absence of metal catalysts inherently minimizes the generation of inorganic impurities, resulting in a cleaner crude reaction profile that simplifies isolation. The versatility of this mechanism is evidenced by its compatibility with a wide array of substituents, ranging from esters and ketones to various fluorinated groups like trifluoromethyl and trifluoromethylthio. This broad scope ensures that the method can be adapted for the synthesis of diverse thiophene libraries required for drug discovery and material science applications. The robustness of the cyclization step underpins the high yields observed across different examples, demonstrating the reliability of the underlying chemical transformation. For R&D teams, this mechanistic clarity provides a solid foundation for further derivative exploration and process optimization.
How to Synthesize Polysubstituted Thiophenes Efficiently
Implementing this synthesis protocol in a laboratory or pilot plant setting requires precise control over stoichiometry and reaction monitoring to ensure maximum conversion and yield. The standard procedure involves dissolving the sulfur ylide precursor and the electron-deficient alkyne in dichloroethane at a specific concentration, typically around 0.1M, to maintain optimal reaction kinetics. A molar excess of the ylide (1.5 equivalents) relative to the alkyne (1.0 equivalent) is employed to drive the reaction to completion and compensate for any potential side reactions or decomposition. The mixture is then heated to 85°C, and the progress is closely monitored, often via TLC or HPLC, until the starting alkyne is fully consumed. Following the reaction, the solvent is removed under reduced pressure, and the crude product is purified using standard silica gel chromatography techniques.
- Mix sulfur-containing ylide A and electron-deficient alkyne B in dichloroethane (DCE) with a molar ratio of 1.5: 1.
- Heat the reaction mixture at 85°C under monitored conditions until the alkyne reactant is completely consumed.
- Concentrate the reaction mixture under reduced pressure and purify the residue via silica gel column chromatography to isolate the target thiophene.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this metal-free synthesis route offers substantial strategic benefits that directly impact the bottom line and operational resilience. The elimination of precious metal catalysts such as palladium or rhodium removes a significant variable cost component and mitigates the risk associated with the volatility of metal prices in the global market. Additionally, the simplified purification process reduces the consumption of specialized scavenging resins and solvents, leading to a more streamlined and cost-effective manufacturing workflow. The use of readily available starting materials and common solvents like DCE ensures that the supply chain remains robust and less susceptible to disruptions caused by the scarcity of exotic reagents. These factors collectively contribute to a more predictable and economical production model for high-value thiophene intermediates.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts fundamentally alters the cost structure of thiophene production by eliminating expensive catalytic loads and the associated downstream metal removal processes. Without the need for specialized ligands or inert gas protection, the operational expenditure on utilities and consumables is significantly lowered, enhancing overall process efficiency. The high yields reported in the patent data suggest that raw material utilization is optimized, minimizing waste generation and maximizing the output per batch. This economic efficiency makes the process highly attractive for commercial scale-up, allowing manufacturers to offer competitive pricing for complex heterocyclic building blocks.
- Enhanced Supply Chain Reliability: Relying on stable, non-hazardous reagents like sulfur ylides and electron-deficient alkynes ensures a consistent and reliable supply of raw materials without the logistical challenges of handling toxic gases. The moderate reaction conditions reduce the dependency on specialized high-pressure reactors or cryogenic cooling systems, making the process adaptable to a wider range of manufacturing facilities. This flexibility allows for diversified sourcing and production strategies, reducing the risk of single-point failures in the supply network. Consequently, customers can expect more stable lead times and uninterrupted availability of critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The green chemistry attributes of this method, characterized by the absence of heavy metals and the use of standard organic solvents, align perfectly with increasingly stringent environmental regulations. Scaling this process from gram to kilogram quantities does not introduce new safety hazards or waste treatment complexities, facilitating a smoother technology transfer to production plants. The reduced environmental footprint supports corporate sustainability goals and simplifies the regulatory approval process for new drug applications. This compliance advantage is a key differentiator for suppliers aiming to partner with top-tier pharmaceutical companies focused on responsible sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel thiophene synthesis technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing production pipelines or new drug development projects.
Q: What are the primary advantages of this thiophene synthesis method over traditional metal-catalyzed routes?
A: This method eliminates the need for transition metal catalysts, thereby avoiding costly metal removal steps and preventing heavy metal contamination in the final API intermediates, which is critical for pharmaceutical compliance.
Q: Is this protocol compatible with fluorinated substituents commonly used in drug design?
A: Yes, the protocol is specifically designed to accommodate various electron-withdrawing groups, including trifluoromethyl (CF3) and trifluoromethylthio (SCF3) groups, enabling the efficient construction of fluorinated thiophene scaffolds.
Q: How does the reaction condition impact the scalability of this process?
A: The reaction operates at a moderate temperature of 85°C in a common solvent like DCE without requiring inert atmosphere or specialized high-pressure equipment, making it highly amenable to kilogram-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Thiophenes Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging advanced technologies like the metal-free thiophene cyclization to deliver superior quality intermediates for the global pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale discoveries are successfully translated into robust manufacturing processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of polysubstituted thiophenes meets the highest international standards for API intermediates. Our commitment to technical excellence allows us to navigate complex synthetic challenges and provide tailored solutions that accelerate our clients' drug development timelines.
We invite you to engage with our technical procurement team to discuss how this cutting-edge synthesis route can optimize your supply chain and reduce costs for your specific projects. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic benefits of switching to this metal-free protocol for your thiophene requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that demonstrate our capability to support your long-term growth. Partnering with us ensures access to reliable, high-quality chemicals backed by deep scientific expertise and a dedication to customer success.
