Advanced Synthesis of Afatinib Cis-Isomer for Pharmaceutical Quality Control and Supply Chain Stability
Advanced Synthesis of Afatinib Cis-Isomer for Pharmaceutical Quality Control and Supply Chain Stability
The pharmaceutical industry relies heavily on the rigorous characterization of active pharmaceutical ingredients (APIs) to ensure patient safety and regulatory compliance. A critical aspect of this quality assurance framework involves the availability of high-purity impurity reference standards. Patent CN106831733B introduces a significant advancement in this domain by detailing a robust preparation method for the afatinib cis-isomer, a crucial impurity associated with the synthesis of afatinib maleate. Afatinib, an irreversible inhibitor of EGFR and HER2 tyrosine kinases, is a vital treatment for non-small cell lung cancer, and its complex molecular architecture necessitates precise control over stereoisomeric purity. The ability to synthesize the cis-isomer independently allows manufacturers to establish accurate detection limits and validate their purification processes effectively.
This technical insight explores the novel synthetic pathway disclosed in the patent, which utilizes a phosphonate intermediate and a bisulfite adduct under mild alkaline conditions. Unlike traditional methods that struggle with low conversion rates and difficult separations, this approach offers a controllable and scalable solution. For global procurement and R&D teams, understanding this methodology is essential for securing a reliable supply of reference materials and optimizing the main API production line. By decoupling the impurity formation from the main process, companies can better manage their impurity profiles and meet stringent pharmacopoeial standards without compromising the yield of the therapeutic agent.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
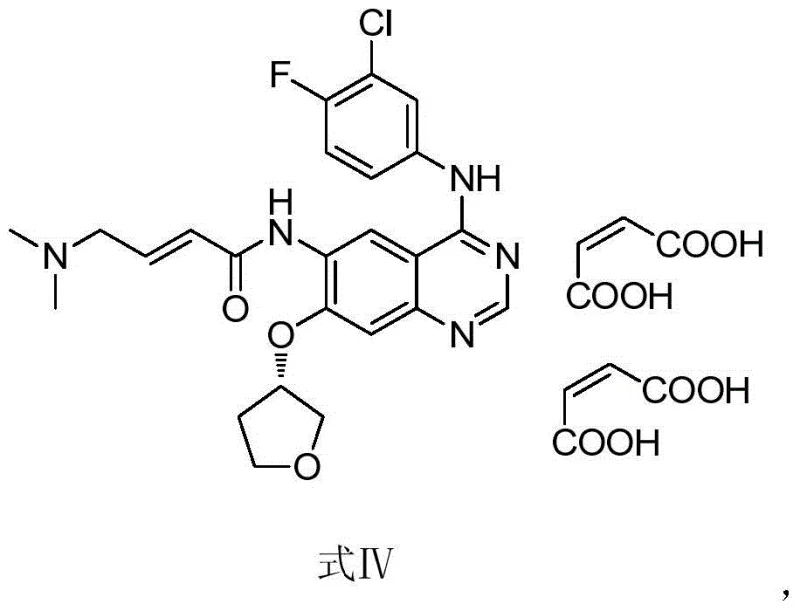
In the established manufacturing routes for afatinib maleate, such as those described in prior art like CN1867564B, the formation of geometric isomers is an inherent challenge. The synthetic pathway typically involves an acylation step where the side chain is introduced to the quinazoline core. However, this reaction does not exclusively produce the desired trans-alkene configuration; it inevitably generates a small amount of the cis-isomer impurity, designated as Formula II. The structural similarity between the cis and trans isomers makes their separation via standard crystallization techniques exceptionally difficult and inefficient. Consequently, the cis-isomer often persists as a trace contaminant, complicating the quality control landscape.
Furthermore, relying on the main production batch to source this impurity for analytical reference purposes is economically and operationally unsound. The conversion rate to the cis-isomer in the standard process is low, and isolating it requires extensive and costly chromatographic resources that disrupt the primary manufacturing flow. Without a dedicated synthesis route, pharmaceutical companies face bottlenecks in obtaining sufficient quantities of the cis-isomer for method validation and stability testing. This dependency creates a vulnerability in the supply chain, where the availability of critical quality control materials is tied to the variability of the main API synthesis.
The Novel Approach
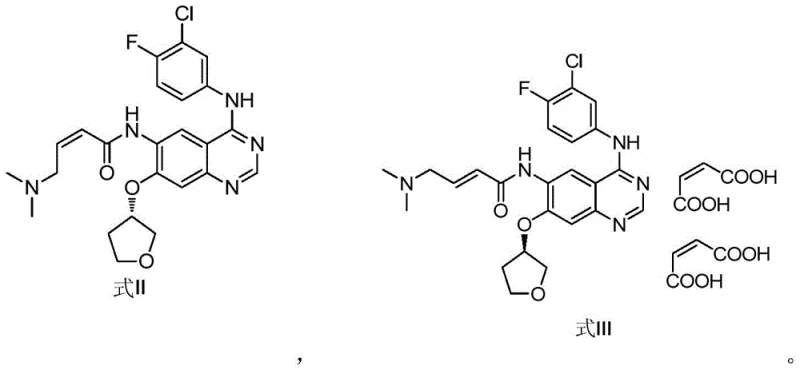
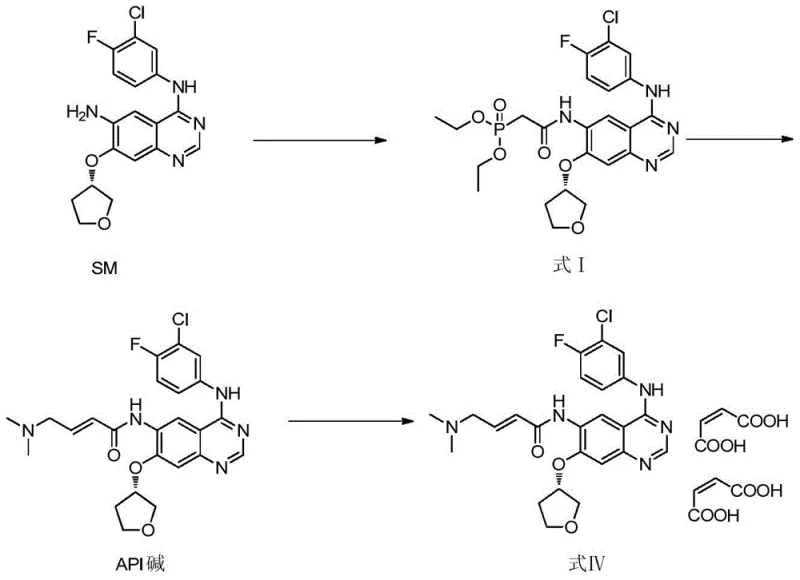
The methodology presented in CN106831733B circumvents these limitations by employing a dedicated synthetic route starting from a specific phosphonate intermediate, Formula I. Instead of relying on the stochastic formation of the cis-isomer during the final coupling steps of the API, this method constructs the Z-alkene geometry directly through a controlled condensation reaction. By utilizing dimethylaminoacetaldehyde sodium bisulfite as the aldehyde equivalent and reacting it with the phosphonate precursor under specific alkaline conditions, the process selectively favors the formation of the cis-configuration. This targeted approach eliminates the need to scavenge the impurity from large batches of the main product.
Moreover, the operational parameters of this novel route are significantly milder and more manageable than traditional high-temperature or harsh acidic conditions often found in older olefination protocols. The reaction proceeds efficiently at temperatures ranging from 20°C to 30°C, reducing energy consumption and thermal stress on the sensitive quinazoline scaffold. The workup procedure is equally streamlined, involving a simple aqueous quench that precipitates the crude product, thereby minimizing solvent usage and waste generation. This shift from a passive byproduct recovery model to an active, dedicated synthesis model represents a paradigm shift in how pharmaceutical impurities are managed and sourced.
Mechanistic Insights into Phosphonate-Mediated Olefination
The core chemical transformation in this patent involves a variation of the Horner-Wadsworth-Emmons (HWE) reaction, tailored to achieve Z-selectivity. The starting material, Formula I, contains a diethyl phosphonate group attached to the acetamide side chain. When treated with a base such as lithium hydroxide or potassium carbonate in a polar aprotic solvent like DMF or ethanol, the phosphonate is deprotonated to form a reactive carbanion species. This nucleophile then attacks the carbonyl carbon of the dimethylaminoacetaldehyde, which is released in situ from its stable sodium bisulfite adduct. The stability of the bisulfite salt allows for controlled release of the aldehyde, preventing polymerization or side reactions that could occur with free aldehyde handling.
The stereochemical outcome of the reaction, favoring the cis (Z) isomer, is influenced by the nature of the phosphonate ester and the specific reaction conditions employed. While classical HWE reactions typically yield E-alkenes, modifications involving specific bases and semi-stabilized ylides can alter the transition state energetics to favor the Z-product. In this specific application, the steric environment of the quinazoline ring and the choice of lithium or potassium cations likely play a pivotal role in directing the geometry of the newly formed double bond. Following the elimination of the phosphate byproduct, the resulting molecule possesses the exact stereochemistry required for the afatinib cis-isomer reference standard.
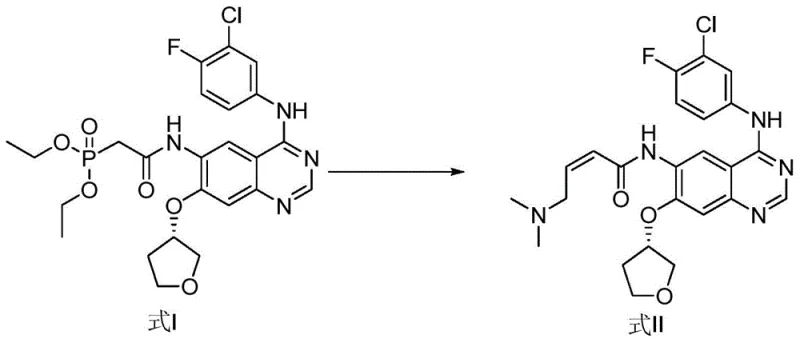
Impurity control in this synthesis is achieved through a combination of selective reaction conditions and advanced purification techniques. The patent highlights the use of preparative liquid chromatography with octadecylsilane chemically bonded silica as the stationary phase. The mobile phase system, comprising an aqueous phosphate buffer and acetonitrile, is optimized to resolve the cis-isomer from any remaining trans-isomer or unreacted starting materials. The gradient elution profile is carefully designed to exploit the subtle polarity differences between the geometric isomers. This level of chromatographic resolution ensures that the final isolated product meets the rigorous purity specifications required for a reference substance, typically exceeding 98% purity as determined by HPLC analysis.
How to Synthesize Afatinib Cis-Isomer Efficiently
The synthesis of this critical impurity standard is designed to be accessible for laboratory-scale production while maintaining the potential for scale-up. The process begins with the dissolution of the phosphonate intermediate in a suitable solvent, followed by the sequential addition of the base and the aldehyde source. Temperature control is paramount throughout the reaction and the subsequent quenching phase to ensure consistent crystal formation and prevent oiling out. The detailed procedural steps involve precise stoichiometric ratios and timing to maximize the conversion efficiency.
- React intermediate Formula I with dimethylaminoacetaldehyde sodium bisulfite in DMF or ethanol at 20-30°C using an alkaline reagent like lithium hydroxide.
- Quench the reaction by adding water while maintaining temperature below 30°C to precipitate the crude cis-isomer product.
- Purify the crude solid via column chromatography using octadecylsilane bonded silica and a gradient elution of phosphate buffer and acetonitrile.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this dedicated synthesis route offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the decoupling of impurity production from the main API manufacturing line. By sourcing the cis-isomer through a specialized, independent pathway, companies eliminate the risk of supply disruptions caused by fluctuations in the main afatinib production schedule. This independence ensures a steady availability of reference standards, which is critical for ongoing quality control testing and regulatory audits. Furthermore, the simplified workup procedure, which relies on water precipitation rather than complex extractions, reduces the operational burden on manufacturing facilities.
- Cost Reduction in Manufacturing: The novel method significantly lowers the cost barrier associated with impurity isolation. Traditional methods require processing massive quantities of API reaction mixtures to recover trace amounts of the cis-isomer, a process that is both solvent-intensive and laborious. In contrast, this dedicated synthesis starts with readily available intermediates and proceeds with high selectivity, drastically reducing the volume of raw materials and solvents required per gram of product. The elimination of expensive transition metal catalysts, which are sometimes used in alternative cross-coupling strategies, further contributes to cost optimization by removing the need for costly metal scavenging steps.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials, such as dimethylaminoacetaldehyde sodium bisulfite and the phosphonate intermediate, strengthens the supply chain resilience. These reagents are stable and easy to source from multiple vendors, mitigating the risk of single-source bottlenecks. Additionally, the mild reaction conditions (20-30°C) mean that the synthesis does not require specialized high-pressure or cryogenic equipment, allowing for production in a wider range of contract manufacturing organizations (CMOs). This flexibility enhances the overall agility of the supply network, enabling faster response times to increased demand for quality control materials.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process aligns well with green chemistry principles. The use of ethanol or DMF as solvents, combined with an aqueous quench, simplifies waste stream management. The high purity achieved directly from the reaction and chromatography reduces the need for repetitive recrystallizations, which often generate significant mother liquor waste. As regulatory pressure mounts for pharmaceutical manufacturers to reduce their environmental footprint, adopting synthetic routes that minimize solvent usage and energy consumption becomes a key competitive advantage. The scalability of the chromatographic purification step also ensures that the method can be adapted from milligram-scale lab synthesis to kilogram-scale production without fundamental changes to the chemistry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of the afatinib cis-isomer. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the method's capabilities and limitations for industry stakeholders.
Q: Why is the afatinib cis-isomer difficult to separate during standard manufacturing?
A: In the standard synthesis of afatinib maleate, the cis-isomer (Formula II) is generated as a minor byproduct alongside the desired trans-isomer. Due to structural similarities, conventional crystallization often fails to separate them effectively, requiring specialized chromatographic conditions.
Q: What are the critical reaction conditions for maximizing cis-isomer yield?
A: The patent specifies mild temperatures between 20-30°C and the use of specific alkaline reagents such as lithium hydroxide or potassium carbonate. Maintaining strict temperature control during the aqueous quench is also vital to prevent degradation.
Q: How is the purity of the synthesized impurity standard verified?
A: High-performance liquid chromatography (HPLC) is used with a C18 column and gradient elution. The method achieves purity levels exceeding 98%, confirmed by mass spectrometry and NMR analysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Afatinib Cis-Isomer Supplier
At NINGBO INNO PHARMCHEM, we understand the critical role that high-quality impurity standards play in the drug development lifecycle. Our technical team has extensively analyzed the synthetic pathways described in CN106831733B and is fully equipped to execute this chemistry with precision. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you need milligrams for analytical method validation or grams for stability studies, our output remains consistent. Our facility is supported by rigorous QC labs and stringent purity specifications, guaranteeing that every batch of afatinib cis-isomer we deliver meets the highest international standards for reference materials.
We invite pharmaceutical partners to leverage our expertise for their afatinib projects. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that evaluates how our optimized synthesis can reduce your overall quality control expenditures. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project timelines. Let us handle the complexity of impurity synthesis so you can focus on delivering safe and effective therapies to patients worldwide.
