Advanced Negative Liquid Crystal Compounds with Terminal Alkenyl for High-Performance Displays
Advanced Negative Liquid Crystal Compounds with Terminal Alkenyl for High-Performance Displays
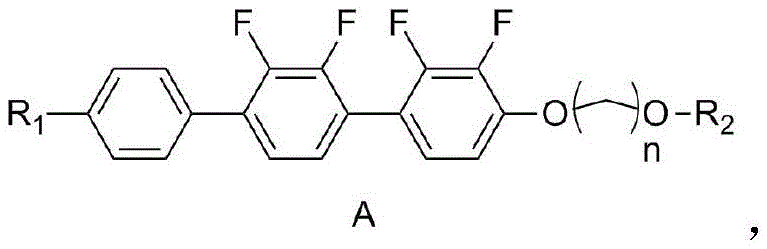
The rapid evolution of liquid crystal display (LCD) technology demands materials that can support lower voltage driving and faster response times without compromising thermal stability. Patent CN111378456B addresses these critical industry needs by disclosing a novel class of negative liquid crystal compounds containing terminal alkenyl groups. These compounds are specifically engineered to enhance the performance of liquid crystal compositions, offering a high clearing point and significant negative dielectric anisotropy. For R&D directors and procurement specialists in the electronic chemicals sector, this patent represents a significant leap forward in material science, providing a robust synthetic pathway that relies on common reagents rather than exotic, costly catalysts. The technical breakthrough lies not just in the final molecular structure, but in the efficiency and safety of the six-step synthesis method described, which ensures high purity and yield suitable for mass production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art in the field of negative liquid crystals, such as the technologies disclosed in WO2018107911A1 and CN108728116A, has historically struggled with inherent performance bottlenecks. These conventional compounds often exhibit insufficient clearing points, meaning they lose their liquid crystalline phase at temperatures that are too low for reliable operation in diverse environmental conditions. Furthermore, they frequently suffer from low optical anisotropy and poor low-temperature stability, which directly impacts the contrast ratio and response speed of the final display panel. From a manufacturing perspective, older synthetic routes often involve complex purification steps or hazardous reagents that drive up production costs and introduce supply chain risks. The inability to simultaneously achieve high dielectric anisotropy and thermal stability has forced formulators to use complex mixtures, complicating the quality control process and increasing the overall cost of goods sold for display manufacturers.
The Novel Approach
The synthesis method detailed in CN111378456B offers a transformative solution by utilizing a modular approach centered on a 2,2',3'-tetrafluoro[1,1'-biphenyl]-4-phenol core. This novel route replaces problematic steps with safer, more efficient reactions such as standard etherification and palladium-catalyzed coupling. By introducing a terminal alkenyl group through a controlled ylide reaction, the method significantly enhances the fluidity and response time of the resulting liquid crystal mixture. The use of readily available starting materials like 1-chloro-4-methoxybutane and simple oxidants ensures that the process is not only chemically elegant but also economically viable. This approach effectively decouples the trade-off between thermal stability and electro-optical performance, allowing for the creation of high-purity intermediates that can be scaled without the exponential cost increases seen in legacy processes.
Mechanistic Insights into the Multi-Step Synthetic Route
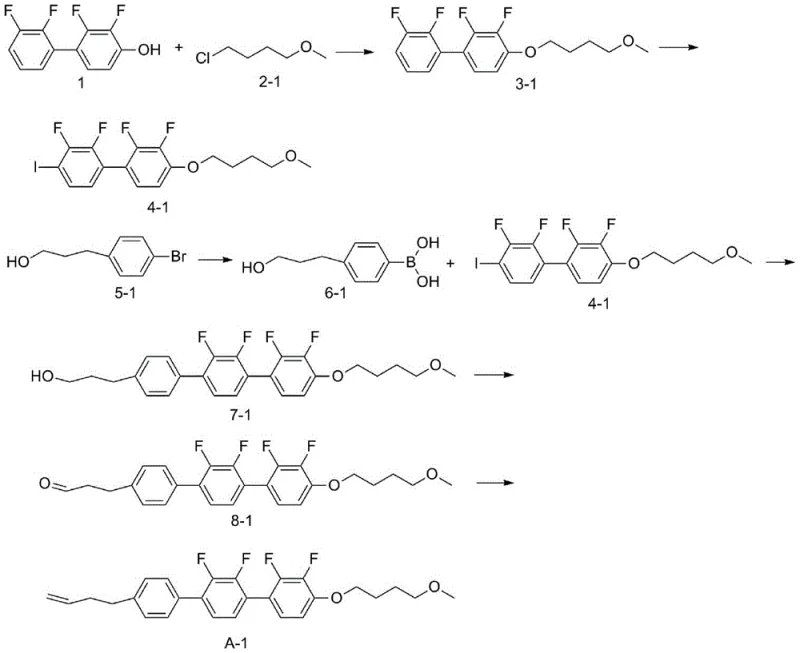
The core of this technology lies in a meticulously designed six-step sequence that builds molecular complexity while maintaining strict control over impurities. The process begins with the nucleophilic substitution of the phenolic hydroxyl group, followed by a regioselective halogenation using lithium reagents at cryogenic temperatures (-50°C to -80°C) to ensure precise positioning of the iodine or bromine atom. This halogenated intermediate is then coupled with a boronic acid derivative via a Suzuki-Miyaura cross-coupling reaction. This step is critical for forming the terphenyl backbone, which is essential for the rigid rod-like structure required for liquid crystallinity. The choice of palladium catalysts, such as tetrakis(triphenylphosphine)palladium, combined with mild bases like potassium carbonate, minimizes the formation of homocoupling byproducts, thereby simplifying downstream purification.
Following the coupling, the synthesis employs a TEMPO-mediated oxidation to convert a primary alcohol into an aldehyde with high selectivity, avoiding over-oxidation to carboxylic acids which would terminate the chain prematurely. The final step involves a Wittig-type olefination using a phosphonium ylide salt to install the terminal alkene. This specific functionalization is crucial for tuning the viscosity and dielectric properties of the final material. Throughout the process, the patent emphasizes the use of standard workup procedures like silica gel column chromatography and recrystallization, which are well-understood unit operations in fine chemical manufacturing. This mechanistic clarity allows process chemists to predict impurity profiles accurately and implement effective control strategies, ensuring the final API-grade or electronic-grade purity required for display applications.
How to Synthesize Negative Liquid Crystal Compound Efficiently
Implementing this synthesis requires careful attention to temperature control during the lithiation and ylide reaction steps, as well as rigorous exclusion of moisture during the coupling phases. The patent provides specific embodiments, such as Example 1, which details the conversion of 2,2',3'-tetrafluoro[1,1'-biphenyl]-4-phenol into the target compound A-1 with an overall high yield. The process is designed to be linear and convergent, allowing different fragments of the molecule to be prepared in parallel before the final assembly. For technical teams looking to replicate or adapt this chemistry, the following standardized guide outlines the critical operational parameters derived directly from the patent data.
- Perform etherification of 2,2',3'-tetrafluoro[1,1'-biphenyl]-4-phenol with a chloroalkane using strong base to form the biphenyl ether intermediate.
- Conduct lithiation followed by halogenation (iodination/bromination) to introduce a halogen handle for cross-coupling.
- Prepare the boronic acid derivative from a bromophenyl alcohol precursor via lithiation and borate ester hydrolysis.
- Execute a palladium-catalyzed Suzuki coupling between the halogenated biphenyl and the boronic acid to form the terphenyl core.
- Oxidize the terminal alcohol group to an aldehyde using TEMPO and an oxidant like trichloroisocyanuric acid.
- Finalize the synthesis via a Wittig-type ylide reaction to install the terminal alkenyl group, yielding the target liquid crystal compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of this patent are profound. The shift towards using common reagents like potassium carbonate, sodium hydroxide, and standard organic solvents (THF, toluene, ethanol) drastically reduces the raw material cost baseline. Unlike processes that rely on precious metal catalysts in stoichiometric amounts or highly specialized ligands that require long lead times, this method utilizes catalytic amounts of widely available palladium complexes. This transition significantly lowers the barrier to entry for production and mitigates the risk of supply disruptions caused by geopolitical factors affecting rare metal availability. Furthermore, the simplified purification steps reduce solvent consumption and waste generation, aligning with modern environmental compliance standards and reducing disposal costs.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents in favor of commodity chemicals leads to substantial cost savings in the bill of materials. By avoiding complex protection-deprotection sequences and utilizing high-yield coupling reactions, the overall process mass intensity is improved, meaning less waste is generated per kilogram of product. This efficiency translates directly into a lower cost of goods sold, allowing buyers to negotiate more competitive pricing for high-performance display materials without sacrificing quality specifications.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials ensures a stable and continuous supply chain. Since the precursors such as chloroalkanes and bromophenyl alcohols are produced by multiple global suppliers, there is no single point of failure in the raw material sourcing strategy. This diversification allows manufacturers to maintain consistent production schedules even during market fluctuations, ensuring that downstream display panel producers receive their materials on time and without interruption.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-ton industrial production. The reaction conditions, typically ranging from ambient to moderate heating (up to 120°C), do not require specialized high-pressure or cryogenic infrastructure beyond standard chillers, reducing capital expenditure for plant upgrades. Additionally, the reduced use of toxic heavy metals and the ability to recycle solvents like ethyl acetate and petroleum ether support greener manufacturing initiatives, helping companies meet increasingly stringent regulatory requirements for electronic chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this liquid crystal technology. These answers are derived from the specific experimental data and beneficial effects reported in the patent documentation, providing clarity for stakeholders evaluating this material for integration into their supply chains.
Q: What are the key performance advantages of the liquid crystal compound disclosed in CN111378456B?
A: The compound exhibits a high clearing point, a high absolute value of negative dielectric anisotropy, high optical anisotropy, and superior low-temperature stability, making it ideal for low-voltage driving and quick response LCD applications.
Q: How does this synthesis method improve upon prior art like WO2018107911A1?
A: Unlike prior art which suffers from low clearing points and poor stability, this method uses common, low-cost reagents and avoids dangerous conditions, resulting in higher yields and fewer byproducts suitable for industrial scale-up.
Q: Is the synthetic route scalable for commercial production?
A: Yes, the process utilizes standard organic solvents like THF and toluene, and avoids exotic catalysts, ensuring that the route is robust, safe, and easily adaptable for large-scale manufacturing from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Negative Liquid Crystal Compound Supplier
As the demand for high-performance display materials continues to surge, partnering with an experienced chemical manufacturer is essential for securing a competitive edge. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move from pilot scale to full industrialization without technical bottlenecks. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of negative liquid crystal compound meets the exacting standards required for next-generation LCD panels. We understand the critical nature of electronic chemicals and are committed to delivering consistency and reliability in every shipment.
We invite you to collaborate with our technical team to explore how this patented synthesis route can be optimized for your specific application needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis, where we can evaluate your current material costs against this new efficient methodology. We are ready to provide specific COA data and route feasibility assessments to demonstrate how we can support your R&D and production goals with high-quality, cost-effective solutions.
