Advanced Catalytic Synthesis of Triazole Pyrimidine Compounds for Commercial Scale-Up
Advanced Catalytic Synthesis of Triazole Pyrimidine Compounds for Commercial Scale-Up
The global agrochemical sector continuously demands more efficient and environmentally sustainable pathways for producing critical herbicide antidotes, particularly for high-volume products like Paraquat. Patent CN100554269C introduces a transformative methodology for synthesizing triazole pyrimidine compounds, specifically targeting the production of 2-amino-6-methyl-4-propyl-4H-[1,2,4]triazolo[1,5-a]pyrimidin-5-one. This specific compound serves as a vital safener, mitigating the high toxicity associated with Paraquat usage, thereby addressing stringent international safety regulations imposed by organizations such as the WHO. The core innovation lies in replacing traditional, harsh acid hydrolysis with a mild Lewis acid-catalyzed amination process, which fundamentally alters the economic and safety profile of the manufacturing workflow. By shifting the reaction paradigm, this technology offers a robust solution for manufacturers seeking to enhance process safety while maintaining rigorous quality standards required for high-purity agrochemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those described in US Patent 6570014B1, rely heavily on the use of stoichiometric quantities of strong mineral acids, typically hydrochloric acid, to effect the final deprotection and amination steps. This reliance creates a profoundly corrosive environment that necessitates the use of specialized glass-lined reactors, significantly inflating capital expenditure and limiting the choice of construction materials for large-scale vessels. Furthermore, the neutralization of vast volumes of acidic waste streams post-reaction generates substantial amounts of saline wastewater, imposing a heavy burden on effluent treatment facilities and increasing the overall environmental footprint of the production site. The exothermic nature of acid hydrolysis also presents significant process control challenges, where localized hot spots can lead to the degradation of the sensitive triazole ring system, resulting in lower yields and complex impurity profiles that require costly downstream purification. These operational hazards and inefficiencies render conventional acid-based routes increasingly untenable for modern, sustainability-focused chemical manufacturing.
The Novel Approach
The novel approach detailed in CN100554269C circumvents these historical bottlenecks by employing catalytic amounts of Lewis acids, such as aluminum trichloride or tin tetrachloride, to drive the conversion of iminopyrimidinone intermediates to the final amine product. This catalytic strategy operates under much milder thermal conditions, typically ranging from 80°C to the reflux temperature of the organic solvent, which drastically reduces energy consumption compared to high-temperature acid digestion. The elimination of bulk mineral acid not only preserves reactor integrity, allowing for the use of standard stainless steel equipment, but also simplifies the work-up procedure by avoiding the need for extensive neutralization and salt removal steps. Moreover, the reaction demonstrates exceptional selectivity, minimizing side reactions and ensuring that the structural integrity of the heterocyclic core is maintained throughout the transformation. This methodological shift represents a significant leap forward in cost reduction in agrochemical intermediates manufacturing by streamlining the process flow and enhancing overall operational safety.
Mechanistic Insights into Lewis Acid-Catalyzed Amination
The mechanistic elegance of this process relies on the ability of the Lewis acid catalyst to coordinate with the nitrogen atoms of the imine functionality within the iminopyrimidinone intermediate, thereby activating the bond for nucleophilic attack or hydrolysis without requiring a highly acidic proton source. In the presence of catalysts like AlCl3, the electron density distribution within the heterocyclic ring is modulated, facilitating the cleavage of the protecting benzylidene group under温和 conditions that would otherwise be insufficient for non-catalyzed hydrolysis. This activation allows the reaction to proceed efficiently in organic solvents such as toluene, where the solubility of the organic intermediates is optimized, ensuring homogeneous reaction kinetics and consistent heat transfer throughout the reactor volume. The catalytic cycle is highly efficient, requiring only 0.01% to 5% molar loading of the catalyst relative to the substrate, which minimizes the introduction of metal contaminants into the final product stream. Such precise control over the reaction mechanism is critical for R&D teams focused on commercial scale-up of complex agrochemical intermediates, as it ensures reproducibility and batch-to-b一致性.
Impurity control is inherently superior in this catalytic regime because the mild conditions prevent the thermal degradation of the triazole ring, a common failure mode in aggressive acid hydrolysis. The specific selection of solvents like toluene allows for the azeotropic removal of water or low-boiling byproducts, driving the equilibrium towards the desired product while simultaneously purifying the reaction mixture in situ. Additionally, the byproduct benzaldehyde generated during the deprotection step remains in the organic phase and can be easily separated and recycled back to the beginning of the synthesis line, creating a closed-loop system that maximizes atom economy. This level of process integration reduces the formation of tarry byproducts and polymeric residues that often plague acid-catalyzed reactions, leading to a cleaner crude product that requires minimal recrystallization. For procurement specialists, this translates to a more reliable supply chain with fewer batch rejections and a consistent quality profile that meets stringent regulatory specifications for reliable agrochemical intermediate supplier partnerships.
How to Synthesize 2-Amino-6-methyl-4-propyl-4H-[1,2,4]triazolo[1,5-a]pyrimidin-5-one Efficiently
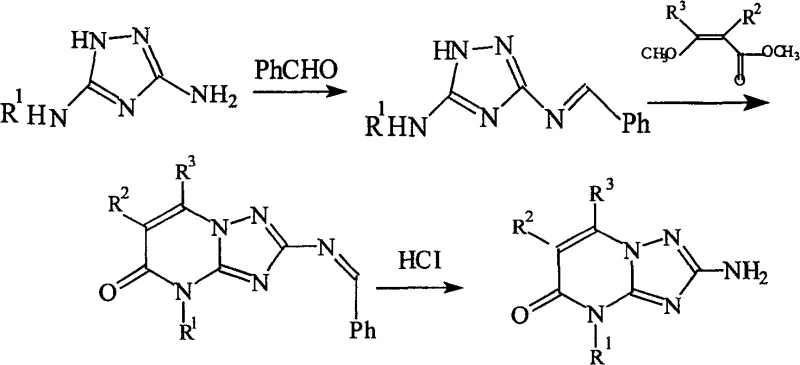
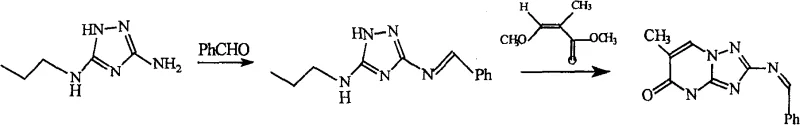
The synthesis protocol outlined in the patent provides a clear, scalable pathway that begins with the condensation of 1,2,4-triazole-3,5-diamine with benzaldehyde to form a protected imine intermediate, followed by cyclization with a substituted acrylate. The critical final step involves treating the resulting iminopyrimidinone with a Lewis acid catalyst in refluxing toluene, a process that has been demonstrated to achieve yields exceeding 95% with product content greater than 98%. This high efficiency eliminates the need for extensive chromatographic purification, making the route ideally suited for multi-ton production campaigns where throughput and cost-efficiency are paramount. The detailed standardized synthetic steps below illustrate the precise operational parameters required to replicate these results in a pilot or commercial plant setting.
- Condense 1,2,4-triazole-3,5-diamine with benzaldehyde in toluene under reflux to form the imine intermediate.
- React the imine intermediate with methyl methoxyacrylate in the presence of a base to effect cyclization into the iminopyrimidinone structure.
- Perform the final amination using a Lewis acid catalyst (e.g., AlCl3) in organic solvent at reflux to yield the target triazole pyrimidine with >95% yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Lewis acid-catalyzed technology offers profound strategic advantages that extend far beyond simple yield improvements. The primary value driver is the drastic simplification of the waste management infrastructure, as the elimination of bulk hydrochloric acid removes the need for expensive corrosion-resistant alloys and complex neutralization tanks, directly lowering both capital and operational expenditures. Furthermore, the ability to recover and reuse both the solvent and the benzaldehyde byproduct creates a circular material flow that significantly reduces raw material consumption and insulates the production cost from volatility in feedstock pricing. The mild reaction conditions also enhance worker safety by reducing exposure to hazardous corrosive vapors, thereby lowering insurance premiums and regulatory compliance risks associated with handling strong acids. These factors combine to create a manufacturing process that is not only economically superior but also resilient to tightening environmental regulations, ensuring long-term supply chain reliability for critical agrochemical inputs.
- Cost Reduction in Manufacturing: The transition from stoichiometric acid usage to catalytic Lewis acid systems fundamentally alters the cost structure by removing the expense associated with acid procurement, storage, and neutralization chemicals. By avoiding the corrosion of reactor vessels, the lifespan of production equipment is significantly extended, deferring capital replacement costs and reducing maintenance downtime. The high selectivity of the reaction minimizes the loss of valuable starting materials to side products, ensuring that the theoretical yield is closely approached in practice, which directly improves the cost-per-kilogram of the final active ingredient. Additionally, the energy demand is optimized due to the lower operating temperatures and the ability to utilize solvent reflux for process control, contributing to a leaner and more competitive manufacturing cost base.
- Enhanced Supply Chain Reliability: The robustness of this catalytic process ensures consistent batch quality, reducing the risk of supply disruptions caused by failed batches or off-specification material that requires reprocessing. The use of common, commercially available solvents like toluene and catalysts like aluminum trichloride ensures that the supply of critical reagents is stable and not subject to the geopolitical constraints that might affect more exotic reagents. The simplified work-up procedure shortens the overall cycle time per batch, allowing for increased production capacity within existing facility footprints and enabling faster response times to market demand fluctuations. This operational agility is crucial for maintaining continuity of supply for downstream formulators who depend on timely delivery of high-quality intermediates for herbicide production.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids the heat transfer limitations and safety hazards associated with large-scale acid hydrolysis, making the transition from pilot plant to commercial production seamless and predictable. The reduction in saline wastewater generation aligns with global trends towards greener chemistry, facilitating easier permitting and reducing the liability associated with environmental discharge limits. By implementing a closed-loop solvent recovery system, the facility minimizes its volatile organic compound (VOC) emissions, further enhancing its environmental standing and community relations. This alignment with sustainability goals makes the technology attractive for multinational corporations seeking to reduce the carbon footprint of their supply chains while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route, providing clarity for stakeholders evaluating its adoption. These insights are derived directly from the experimental data and process descriptions within CN100554269C, ensuring that the information is grounded in verified chemical principles and practical outcomes. Understanding these details is essential for making informed decisions about process integration and vendor qualification.
Q: What is the primary advantage of using Lewis acid catalysts over hydrochloric acid in this synthesis?
A: The use of Lewis acid catalysts such as aluminum trichloride eliminates the need for large volumes of corrosive hydrochloric acid, thereby preventing severe equipment corrosion and reducing the generation of acidic wastewater, which significantly lowers environmental compliance costs.
Q: Can the solvents and byproducts be recovered in this process?
A: Yes, the process allows for the efficient recovery and recycling of the organic solvent (toluene) and the byproduct benzaldehyde generated during the final amination step, which can be reused in the initial condensation reaction to maximize atom economy.
Q: What purity levels can be achieved with this catalytic method?
A: The patented method consistently achieves product content exceeding 98% and synthesis yields greater than 95%, often eliminating the need for further purification steps before the material is suitable for downstream agrochemical formulation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-6-methyl-4-propyl-4H-[1,2,4]triazolo[1,5-a]pyrimidin-5-one Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, leveraging advanced catalytic technologies like the one described in CN100554269C to deliver superior value to our global partners. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of the agrochemical industry without compromising on quality. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch of triazole pyrimidine intermediate meets the exacting standards required for herbicide safener applications. Our commitment to process excellence means that we can offer a stable, high-quality supply of this critical intermediate, supporting your production schedules with reliability and precision.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to our catalytically produced materials. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to validate our capabilities and ensure that our solutions align perfectly with your manufacturing goals and quality expectations.
