Advanced Manufacturing of Trans-4-Acetoxy-2-Methyl-2-Butene Aldehyde via Novel Isoprene Chlorination
Introduction to High-Purity Vitamin A Intermediate Synthesis
The global demand for high-purity Vitamin A and its derivatives necessitates robust, scalable, and environmentally compliant synthetic routes for key precursors. Patent CN101092355A introduces a transformative methodology for the preparation of trans-4-acetoxy-2-methyl-2-butene aldehyde, a critical five-carbon unsaturated skeleton compound. This technology represents a significant departure from legacy oxidation processes by leveraging a direct chlorination-esterification strategy followed by a mild hydrolytic cleavage. By utilizing isoprene as the foundational feedstock and tert-butyl hypochlorite as the functionalizing agent within an acetic anhydride medium, the process achieves a streamlined operational profile that minimizes waste generation. The structural integrity of the target molecule, characterized by its specific trans-configuration and acetoxy functionality, is preserved through careful temperature control and catalyst selection, ensuring the production of a reliable pharmaceutical intermediate suitable for downstream vitamin synthesis.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trans-4-acetoxy-2-methyl-2-butene aldehyde has been plagued by inefficiencies and environmental hazards inherent to older oxidation technologies. Traditional domestic manufacturing often relied on acetone dimethyl acetal as a starting material, a pathway characterized by excessive reaction steps, low overall yields, and prohibitive raw material costs that erode profit margins. Furthermore, established international methods, such as those disclosed in United States Patent 4175204, depend heavily on the reaction of isoprene with sodium hypochlorite followed by oxidation using dimethyl sulfoxide (DMSO). While these methods can achieve moderate yields, they generate dimethyl sulfide as a unavoidable byproduct, which possesses an extremely offensive odor and requires complex, energy-intensive removal protocols to prevent contamination of the final product. Additionally, alternative literature methods employing TEMPO catalysis and periodic acid-loaded ion exchange resins introduce expensive reagents and multi-step addition protocols that drastically increase the complexity of the supply chain and the difficulty of waste treatment.
The Novel Approach
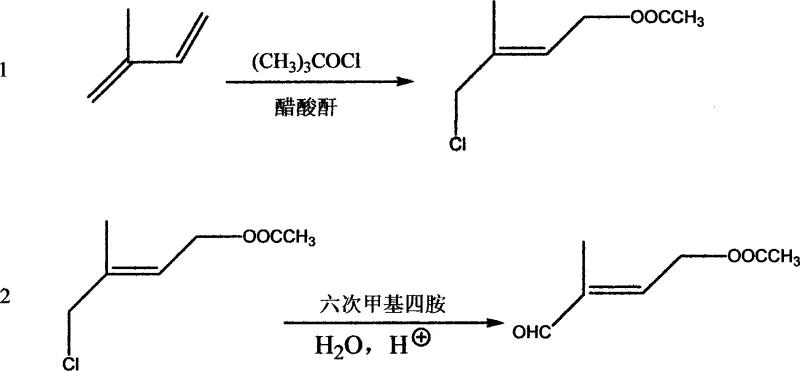
In stark contrast to these cumbersome legacy techniques, the novel approach detailed in the patent data utilizes a concise two-step sequence that maximizes atom economy and operational simplicity. The process initiates with the direct reaction of isoprene and tert-butyl hypochlorite in an acetic anhydride solution, effectively combining chlorination and esterification into a single pot operation to generate a chlorine-containing ester intermediate. This intermediate is subsequently treated with hexamethylenetetramine (urotropine) and subjected to acidic hydrolysis to yield the target aldehyde. This strategic shift eliminates the need for DMSO entirely, thereby removing the risk of sulfide byproduct formation and the associated purification burdens. The reaction conditions are remarkably mild, proceeding at temperatures ranging from -10°C to 90°C depending on the stage, and utilize readily available catalysts such as tosic acid or sulfuric acid. The result is a robust synthetic route that delivers yields between 40% and 56% based on isoprene conversion, with significantly reduced environmental impact and lower financial overhead.
Mechanistic Insights into Isoprene Chlorination and Urotropine Hydrolysis
The core of this technological advancement lies in the precise mechanistic control of the electrophilic addition and subsequent nucleophilic substitution. In the first stage, tert-butyl hypochlorite serves as a source of electrophilic chlorine, which adds across the double bond of isoprene in the presence of acetic anhydride. The acetic anhydride acts not merely as a solvent but as a reactive trapping agent, immediately esterifying the transient alcohol species formed during the addition to prevent rearrangement or polymerization. The addition of a catalytic amount of mineral or organic acid, such as p-toluenesulfonic acid, facilitates the ionization of the hypochlorite and stabilizes the carbocation intermediate, ensuring high regioselectivity for the desired chloro-ester isomer. This one-pot transformation is critical for maintaining high throughput, as it avoids the isolation of unstable intermediates that typically degrade under standard workup conditions.
Following the formation of the chloro-ester, the second stage employs hexamethylenetetramine to effect a Sommelet-type reaction variant. The nitrogen atoms of the urotropine act as strong nucleophiles, displacing the chloride ion to form a quaternary ammonium salt. This step is conducted in organic solvents like toluene or acetonitrile at moderate temperatures (30-40°C) to ensure complete conversion without degrading the sensitive ester linkage. The subsequent hydrolysis, triggered by the addition of water and glacial acetic acid at elevated temperatures (60-90°C), cleaves the quaternary salt to release the aldehyde functionality while regenerating the amine components. This mechanism bypasses the need for harsh oxidizing agents, thereby preserving the stereochemistry of the double bond and ensuring the exclusive formation of the trans-isomer, which is essential for the biological activity of the final Vitamin A product.
How to Synthesize Trans-4-Acetoxy-2-Methyl-2-Butene Aldehyde Efficiently
To implement this synthesis effectively, operators must adhere to strict temperature gradients and stoichiometric ratios as defined in the patent examples. The process begins by cooling the acetic anhydride and isoprene mixture to sub-zero temperatures before the controlled addition of tert-butyl hypochlorite, a step crucial for managing the exotherm and preventing side reactions. Following the initial addition, the reaction mass is warmed to facilitate the catalytic cycle, after which the organic phase is separated and dried. The second phase involves the dissolution of the chloro-ester in a suitable solvent, followed by the addition of urotropine and a controlled hydrolysis sequence. Detailed standardized synthetic steps, including specific molar ratios, stirring times, and distillation pressures for solvent recovery, are outlined in the comprehensive guide below to ensure reproducibility and safety.
- React isoprene with tert-butyl hypochlorite in an acetic anhydride solution at -10 to 0°C, then warm to 60-90°C with an acid catalyst to form the chloro-ester intermediate.
- Treat the resulting chloro-ester with hexamethylenetetramine (urotropine) in an organic solvent at 30-40°C.
- Hydrolyze the mixture by adding water and glacial acetic acid, warming to 60-90°C to yield the final trans-4-acetoxy-2-methyl-2-butene aldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this isoprene-based route offers substantial strategic benefits centered around cost stability and operational reliability. By shifting away from complex multi-step oxidations that require specialized catalysts like TEMPO or periodic acid resins, manufacturers can significantly reduce the dependency on volatile high-value reagent markets. The elimination of DMSO not only simplifies the chemical inventory but also removes the logistical and regulatory challenges associated with handling and disposing of sulfur-containing waste streams, which are increasingly subject to stringent environmental regulations. This streamlining of the chemical bill of materials translates directly into a more predictable cost structure and reduced exposure to supply chain disruptions caused by the scarcity of niche oxidizing agents.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the utilization of isoprene, a commodity petrochemical feedstock that is abundantly available and cost-effective compared to acetone dimethyl acetals or other specialized precursors. Furthermore, the dual role of acetic anhydride as both solvent and reactant reduces the total volume of materials required, while the ability to recover solvents like toluene and acetonitrile through simple distillation lowers the recurring operational expenditure. The avoidance of expensive transition metal catalysts and the reduction in reaction steps from three or more down to two fundamentally lowers the processing time and energy consumption per kilogram of product, resulting in substantial cost savings without compromising quality.
- Enhanced Supply Chain Reliability: From a sourcing perspective, the reliance on bulk chemicals such as isoprene, acetic anhydride, and hexamethylenetetramine ensures a robust supply chain that is less susceptible to the bottlenecks often seen with fine chemical reagents. These raw materials are produced on a massive industrial scale globally, guaranteeing consistent availability and reducing the lead time for high-purity pharmaceutical intermediates. The simplified workflow also means that production campaigns can be turned around more quickly, allowing for greater flexibility in meeting fluctuating market demands for Vitamin A precursors without the need for extended campaign planning or complex logistics coordination.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, featuring straightforward unit operations such as liquid-liquid separation and vacuum distillation that are easily replicated in large-scale reactors. The absence of malodorous dimethyl sulfide byproducts significantly improves the working environment and reduces the burden on scrubber systems and waste treatment facilities, facilitating easier compliance with environmental protection standards. This clean profile makes the technology highly attractive for manufacturing sites facing increasing pressure to reduce their carbon footprint and volatile organic compound (VOC) emissions, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, impurity profiles, and scalability factors. Understanding these nuances is essential for R&D teams evaluating the feasibility of technology transfer and for quality assurance personnel establishing specification limits for the final intermediate.
Q: What are the primary advantages of this isoprene-based route over traditional DMSO oxidation methods?
A: The primary advantage is the complete elimination of dimethyl sulfoxide (DMSO) and the associated generation of malodorous dimethyl sulfide byproducts. This new method utilizes tert-butyl hypochlorite and acetic anhydride to directly form the chloro-ester, followed by a clean hydrolysis using urotropine, resulting in a yield of 40-56% without the complex purification steps required to remove sulfur contaminants.
Q: Why is hexamethylenetetramine (urotropine) used in the second step of the synthesis?
A: Hexamethylenetetramine acts as a nitrogen nucleophile that reacts with the chloro-ester intermediate to form a quaternary ammonium salt. Upon subsequent acidic hydrolysis, this salt decomposes to release the desired aldehyde functionality. This avoids the need for expensive transition metal oxidants like TEMPO or periodic acid found in other literature methods.
Q: Can this process be scaled for industrial production of Vitamin A intermediates?
A: Yes, the patent explicitly states the method is suitable for industrial production due to its few reaction procedures, easy operation, and the use of low-cost raw materials like isoprene and acetic anhydride. The solvents used, such as toluene or acetonitrile, are easily recoverable, further supporting large-scale commercial viability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-4-Acetoxy-2-Methyl-2-Butene Aldehyde Supplier
As the pharmaceutical and fine chemical industries continue to evolve towards greener and more efficient manufacturing paradigms, the ability to execute complex synthetic routes with precision is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging deep technical expertise to bring innovative processes like the isoprene-based synthesis of Vitamin A intermediates to commercial reality. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require pilot quantities for clinical trials or metric tons for industrial formulation, our output remains consistent. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of trans-4-acetoxy-2-methyl-2-butene aldehyde meets the exacting standards required for downstream vitamin synthesis.
We invite global partners to collaborate with us to optimize their supply chains and reduce manufacturing costs through advanced process chemistry. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data, discuss route feasibility assessments, and explore how our commitment to innovation can drive value for your organization.
