Scalable Synthesis of Novel Coumarin Sulfonamides for High-Purity BRD4 Inhibitor Manufacturing
Scalable Synthesis of Novel Coumarin Sulfonamides for High-Purity BRD4 Inhibitor Manufacturing
The pharmaceutical industry is constantly seeking robust and scalable pathways for next-generation epigenetic modulators, particularly those targeting the Bromodomain and extra C-terminal domain (BET) family. Patent CN109824640B introduces a significant advancement in this field by disclosing a series of novel coumarin compounds characterized by the general structure shown in Formula (I). These compounds exhibit potent inhibitory activity against the BRD4 protein, a critical epigenetic reader involved in the transcriptional regulation of oncogenes such as MYC. The structural versatility of this scaffold allows for extensive modification at the sulfonamide nitrogen, enabling fine-tuning of pharmacokinetic properties and binding affinity. As depicted in the general structural formula, the core coumarin nucleus serves as a rigid planar anchor, while the variable R group provides the necessary chemical diversity to optimize interactions within the BRD4 acetyl-lysine binding pocket. This innovation represents a strategic opportunity for developing new anti-tumor therapeutics with improved selectivity profiles compared to existing BET inhibitors.
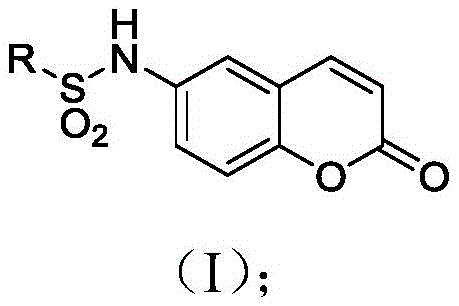
The therapeutic potential of these molecules extends across a broad spectrum of malignancies, including leukemia, multiple myeloma, and various solid tumors where BRD4 dysregulation drives pathogenesis. By effectively blocking the interaction between BRD4 and acetylated histones, these small molecules can silence oncogenic transcription programs, offering a promising avenue for treating cancers that have developed resistance to conventional therapies. The patent highlights that these compounds are not merely theoretical constructs but have been synthesized and validated through rigorous in vitro testing, demonstrating measurable IC50 values in the micromolar range against BRD4. For research and development teams focused on oncology, accessing high-purity intermediates of this specific chemotype is essential for lead optimization and preclinical candidate selection. The ability to rapidly access diverse analogs through a modular synthetic route significantly accelerates the drug discovery timeline, allowing medicinal chemists to explore structure-activity relationships (SAR) with greater efficiency and precision.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for accessing complex heterocyclic sulfonamides often rely on multi-step sequences involving expensive transition metal catalysts, such as palladium or copper, which introduce significant cost and purification burdens. Many existing methods for constructing the coumarin-sulfonamide linkage require harsh reaction conditions, including high temperatures or strong bases, which can compromise the integrity of sensitive functional groups on the aromatic rings. Furthermore, conventional approaches frequently suffer from poor regioselectivity during the functionalization of the coumarin core, leading to difficult-to-separate isomeric impurities that complicate downstream processing. The reliance on precious metal catalysts also raises concerns regarding residual metal contamination in the final active pharmaceutical ingredient (API), necessitating additional and costly scavenging steps to meet stringent regulatory limits. These factors collectively contribute to extended production timelines and inflated manufacturing costs, creating bottlenecks for supply chain managers who need to secure reliable volumes of clinical-grade material.
The Novel Approach
In stark contrast, the methodology described in patent CN109824640B utilizes a streamlined three-step sequence that begins with commercially abundant coumarin as the starting material. This approach eliminates the need for precious metal catalysis entirely, relying instead on classical organic transformations that are well-understood and easily controlled on a large scale. The strategy employs a regioselective nitration to install the necessary handle at the 6-position of the coumarin ring, followed by a reduction to the aniline and a final sulfonylation. This linear progression minimizes the number of unit operations and reduces the overall environmental footprint of the synthesis. By avoiding complex cross-coupling reactions, the process inherently reduces the risk of generating heavy metal impurities, simplifying the quality control workflow. The simplicity of the operation, combined with the use of inexpensive reagents like iron powder for reduction, positions this method as a highly economically viable alternative for the commercial production of BRD4 inhibitor intermediates.
Mechanistic Insights into the Three-Step Synthetic Route
The synthetic pathway leverages fundamental principles of electrophilic aromatic substitution and nucleophilic attack to construct the target molecule with high fidelity. The initial step involves the nitration of the coumarin nucleus using a mixed acid system of nitric and sulfuric acid at low temperatures. This condition is critical for generating the nitronium ion electrophile while preventing the hydrolysis of the lactone ring, which is susceptible to ring-opening under acidic or basic stress. The electron-donating nature of the lactone oxygen directs the incoming nitro group predominantly to the 6-position, ensuring the formation of the desired regioisomer required for subsequent biological activity. Following isolation, the nitro group serves as a precursor for the amine functionality through a reduction process. The use of iron powder in the presence of ammonium chloride provides a mild yet effective reducing environment that converts the nitro group to an amino group without affecting other sensitive moieties on the molecule. This heterogeneous reduction is advantageous for scale-up as the iron byproducts can be easily removed via filtration.
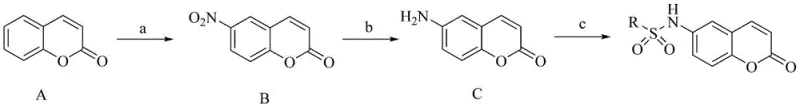
The final transformation involves the reaction of the newly formed 6-amino-coumarin with a variety of sulfonyl chlorides to generate the diverse library of sulfonamides. This step proceeds via a nucleophilic attack of the aniline nitrogen on the sulfur atom of the sulfonyl chloride, displacing the chloride ion. The reaction is typically facilitated by a base such as pyridine, which acts both as a solvent and as a proton scavenger to drive the equilibrium towards product formation. The modularity of this step allows for the rapid incorporation of different R groups, ranging from simple alkyl chains to complex aryl systems, simply by changing the sulfonyl chloride starting material. This flexibility is crucial for SAR studies, enabling the rapid generation of analogs to optimize potency and metabolic stability. The entire sequence, from nitration to final coupling, is designed to be robust and forgiving, tolerating a wide range of substituents on the sulfonyl chloride component, which is a key feature for a versatile pharmaceutical intermediate platform.
How to Synthesize N-(2-oxo-2H-chromen-6-yl)sulfonamides Efficiently
Executing this synthesis requires careful attention to temperature control and stoichiometry to maximize yield and purity at each stage. The process begins with the preparation of the nitrated intermediate, where maintaining the reaction temperature near 0°C is essential to prevent over-nitration or decomposition. Once the nitro-coumarin is isolated, the reduction step utilizes a slurry of iron powder which must be thoroughly washed and filtered to ensure the removal of inorganic residues before proceeding to the final coupling. The final sulfonylation is typically conducted in dichloromethane at room temperature, allowing for easy monitoring by TLC and straightforward workup procedures. Detailed standardized operating procedures for each of these critical unit operations are provided below to ensure reproducibility and safety during scale-up efforts.
- Perform mixed acid nitration on coumarin at 0°C to obtain 6-nitro-2H-benzopyran-2-one.
- Reduce the nitro intermediate using iron powder and ammonium chloride at 80°C to yield the amino-coumarin.
- React the amino-coumarin with the corresponding sulfonyl chloride in dichloromethane with pyridine to form the final sulfonamide.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial advantages by utilizing commodity chemicals that are readily available in the global market. The primary starting material, coumarin, is produced on a massive scale for the fragrance and flavor industries, ensuring a stable and continuous supply chain that is not subject to the volatility often seen with specialized pharmaceutical building blocks. The elimination of precious metal catalysts not only reduces the direct material cost but also removes the logistical complexity associated with sourcing and recovering expensive metals like palladium. This simplification of the bill of materials translates directly into a more predictable cost structure, allowing procurement managers to negotiate better long-term contracts with suppliers. Furthermore, the use of common solvents like ethanol and dichloromethane facilitates solvent recovery and recycling, contributing to overall process sustainability and waste reduction.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the replacement of expensive catalytic systems with stoichiometric reagents like iron powder, which is orders of magnitude cheaper. By removing the need for specialized ligands and inert atmosphere conditions often required for cross-coupling reactions, the operational expenditure (OPEX) is significantly lowered. The high yields reported in the patent examples indicate efficient atom economy, meaning less raw material is wasted as byproduct, further driving down the cost per kilogram of the final intermediate. Additionally, the simplified purification requirements reduce the consumption of silica gel and eluents, which are often hidden cost drivers in chromatographic purifications.
- Enhanced Supply Chain Reliability: The reliance on widely available feedstocks mitigates the risk of supply disruptions that can occur with niche reagents. Since the synthetic steps are chemically robust and do not require exotic equipment, the process can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without significant re-validation. This flexibility ensures business continuity and allows for the rapid scaling of production capacity to meet fluctuating demand during clinical trials. The stability of the intermediates also allows for strategic stockpiling, providing a buffer against unexpected market shortages or logistics delays.
- Scalability and Environmental Compliance: The mild reaction conditions, particularly the ambient temperature sulfonylation and the aqueous workup of the iron reduction, make this process inherently safer and easier to scale from grams to tons. The absence of heavy metal catalysts simplifies the environmental compliance landscape, as there is no need for complex wastewater treatment protocols to remove trace metals. This aligns with modern green chemistry principles and corporate sustainability goals, making the supply chain more attractive to environmentally conscious stakeholders. The straightforward isolation of products via filtration or crystallization reduces energy consumption compared to processes requiring high-vacuum distillation or lyophilization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these coumarin-based BRD4 inhibitors. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the technology's capabilities. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this chemistry into their drug development pipelines.
Q: What is the primary advantage of this coumarin synthesis route?
A: The primary advantage is the use of inexpensive, readily available starting materials like coumarin and iron powder, avoiding expensive transition metal catalysts and enabling cost-effective large-scale production.
Q: How does the compound interact with the BRD4 protein?
A: The coumarin sulfonamide scaffold is designed to bind within the acetylated lysine recognition pocket of the BRD4 Bromodomain, interfering with epigenetic signal transmission and inhibiting tumor cell proliferation.
Q: Is this process suitable for industrial scale-up?
A: Yes, the patent explicitly states the method features mild reaction conditions, simple operation, and wide raw material sources, making it highly suitable for industrialized large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Coumarin Sulfonamides Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for the supply of complex pharmaceutical intermediates like these BRD4 inhibitors. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and timeliness. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the highest standards of quality. Our commitment to technical excellence allows us to navigate the complexities of multi-step syntheses, delivering materials that accelerate your research and development milestones without compromise.
We invite you to engage with our technical procurement team to discuss your specific requirements for coumarin sulfonamide derivatives. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into how our optimized manufacturing processes can reduce your overall project costs. We encourage potential partners to contact us for specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your strategic goals for bringing novel anti-cancer therapies to the market.
