Revolutionizing Apalutamide Intermediate Production with Highly Dispersed Bimetallic Nanomaterials
Revolutionizing Apalutamide Intermediate Production with Highly Dispersed Bimetallic Nanomaterials
The global demand for advanced oncology therapeutics continues to surge, driven by an aging population and the critical need for effective treatments for conditions such as castration-resistant prostate cancer (CRPC). At the forefront of this therapeutic class is Apalutamide, a potent Androgen Receptor inhibitor approved by the FDA, which relies on high-quality intermediates for its synthesis. A groundbreaking technological advancement detailed in patent CN108314646B introduces a novel preparation method for a key urogenital system drug intermediate, specifically 2-cyano-5-nitro-3-(trifluoromethyl)pyridine. This innovation leverages a supported high-dispersion Pd/Ni co-doped two-component catalyst derived from metal-organic frameworks (MOFs), offering a transformative approach to efficiency and purity in pharmaceutical manufacturing.
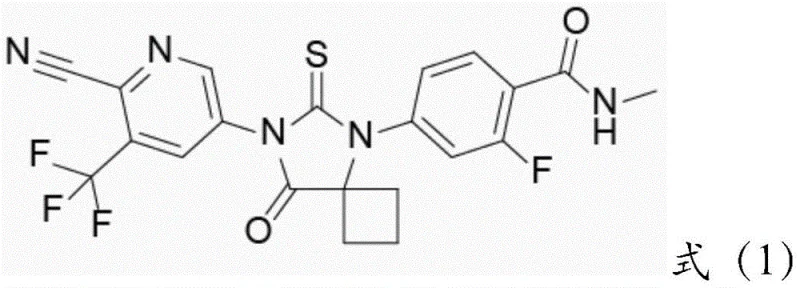
This patent represents a significant leap forward by addressing the longstanding challenges of catalyst recovery and heavy metal residue control, which are paramount for GMP compliance. By utilizing a heterogeneous catalytic system based on 2-amino terephthalic acid and nickel chloride hexahyrdate, the process ensures that the precious palladium component is firmly anchored, allowing for straightforward physical separation via filtration. For R&D directors and procurement specialists alike, this technology signals a shift towards more sustainable and cost-effective supply chains for complex heterocyclic intermediates, ensuring a reliable supply of high-purity materials essential for the final drug substance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
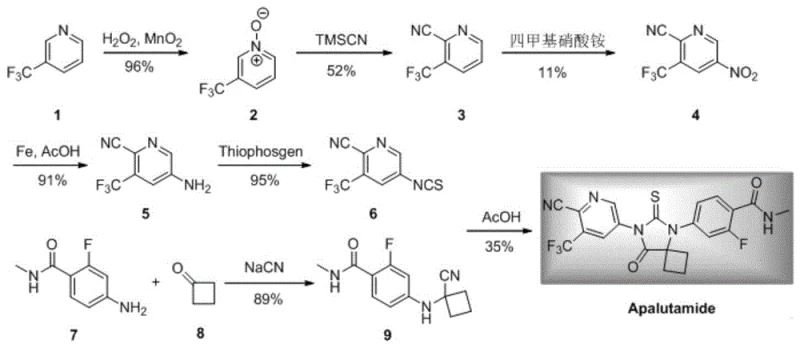
Historically, the synthesis of Apalutamide intermediates has been plagued by inefficient reaction pathways that suffer from low overall yields and hazardous operational conditions. As illustrated in existing literature and prior art schemes, traditional routes often involve multi-step sequences that introduce significant bottlenecks. For instance, one common pathway involves a nitration step that exhibits notoriously low yields, creating a "short plate" effect that drags down the entire process efficiency. Furthermore, alternative routes have relied on the use of Raney Nickel for reduction steps, which presents severe safety hazards due to its pyrophoric nature and the potential for generating dangerous byproducts like dechlorinated impurities.
Another critical drawback of conventional homogeneous catalysis is the difficulty in removing residual heavy metals. Traditional palladium-catalyzed cyanation reactions often require stoichiometric amounts of ligands and result in catalyst residues that are difficult to scrub below the strict ppm levels mandated by regulatory bodies. This necessitates additional downstream processing steps, such as specialized scavenging or repeated recrystallization, which inflate production costs and extend lead times. Moreover, the use of highly toxic cyanation reagents like sodium cyanide or potassium cyanide in these older methods poses substantial environmental and worker safety risks, complicating waste management and increasing the overall carbon footprint of the manufacturing process.
The Novel Approach
In stark contrast, the methodology disclosed in CN108314646B offers a streamlined, robust alternative that circumvents these historical pitfalls through the ingenious design of a bimetallic nanomaterial. The core of this innovation lies in the construction of a Metal-Organic Framework (MOF) support using 2-amino terephthalic acid and nickel, upon which palladium is loaded and subsequently reduced at high temperatures. This architecture creates a highly dispersed active site that maximizes catalytic efficiency while maintaining structural integrity. The result is a cyanation reaction of 2-bromo-5-nitro-3-(trifluoromethyl)pyridine that achieves conversion rates exceeding 99% with exceptional selectivity, effectively solving the yield issues associated with previous nitration-based routes.
Furthermore, this novel approach champions safety and simplicity by employing potassium ferrocyanide (K4[Fe(CN)6]) as the cyanation source. Unlike free cyanides, this reagent is non-toxic, inexpensive, and stable, eliminating the need for complex safety protocols and specialized containment infrastructure. The heterogeneous nature of the catalyst allows it to be removed simply by filtration, and remarkably, the patent data indicates that the catalyst can be recycled and reused multiple times without significant loss of activity. This not only reduces the consumption of precious metals but also simplifies the workup procedure, enabling the isolation of high-purity products through extraction and concentration alone, completely bypassing the need for resource-intensive column chromatography.
Mechanistic Insights into Pd/Ni Co-Doped MOF Catalysis
The superior performance of this system can be attributed to the synergistic interaction between the palladium active sites and the nickel-containing MOF support. The preparation begins with the hydrothermal treatment of 2-amino terephthalic acid and nickel chloride hexahydrate under alkaline conditions, forming a stable salt precursor with a defined metal-organic framework structure. Upon loading with potassium tetrachloropalladate and subsequent high-temperature reduction in a hydrogen atmosphere, ultra-fine palladium nanoparticles are uniformly distributed across the MOF surface. Transmission Electron Microscopy (TEM) characterization confirms this high dispersion, which is critical for maximizing the surface area available for the catalytic cycle. The nickel component likely plays a structural role in stabilizing the palladium nanoparticles against aggregation, ensuring long-term catalyst stability during the rigorous conditions of the cyanation reaction.
From a mechanistic standpoint, the reaction proceeds through a classic palladium-catalyzed cross-coupling cycle, but with enhanced kinetics due to the unique electronic environment provided by the support. The addition of specific catalyst promoters, particularly a mixture of potassium fluoride and potassium iodide in a 1:1 molar ratio, further accelerates the reaction rate. It is hypothesized that the iodide ions facilitate a weak halogen exchange with the bromine substrate, generating a more reactive aryl-iodide intermediate in situ, which then undergoes rapid cyanation. This promoter effect allows the reaction to proceed efficiently at moderate temperatures (around 120°C) while maintaining high selectivity for the target nitrile product, minimizing the formation of hydrodehalogenated or homocoupled byproducts that typically plague such transformations.
Impurity control is inherently built into this catalytic system. The high selectivity (>93%) observed during screening means that fewer side reactions occur, simplifying the impurity profile of the crude reaction mixture. Additionally, the use of a solid-supported catalyst prevents the leaching of palladium into the solution phase, a common issue with homogeneous systems that leads to metal contamination in the final API. The patent data demonstrates that even after multiple recycling runs, the catalyst maintains conversion rates above 99%, indicating that the active sites remain accessible and intact. This robustness is essential for consistent batch-to-batch quality, ensuring that the intermediate meets the stringent purity specifications required for downstream pharmaceutical synthesis without requiring extensive purification efforts.
How to Synthesize 2-Cyano-5-nitro-3-(trifluoromethyl)pyridine Efficiently
The implementation of this advanced catalytic protocol requires precise adherence to the preparation and reaction conditions outlined in the patent to ensure optimal performance. The process begins with the meticulous synthesis of the Pd/Ni-MOF catalyst, followed by its application in the cyanation of the bromo-pyridine substrate using safe cyanide sources and specific promoters. The following guide summarizes the critical operational parameters necessary to replicate the high yields and purity described in the intellectual property, serving as a foundational reference for process chemists aiming to adopt this technology.
- Prepare the catalyst by forming a metal-organic framework from 2-amino terephthalic acid and nickel chloride, followed by Pd loading and high-temperature reduction.
- Conduct the cyanation reaction of 2-bromo-5-nitro-3-(trifluoromethyl)pyridine using K4[Fe(CN)6] and the Pd/Ni catalyst in DMAc solvent with KF/KI promoters.
- Isolate the product via filtration of the catalyst, followed by acidic aqueous back-extraction to achieve high purity without column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Pd/Ni-MOF catalytic technology translates directly into tangible operational efficiencies and risk mitigation. The shift from homogeneous to heterogeneous catalysis fundamentally alters the cost structure of the manufacturing process by eliminating the need for expensive metal scavengers and complex filtration aids. Since the catalyst can be physically separated via simple filtration and reused, the consumption of palladium—a volatile and costly commodity—is drastically reduced. This reduction in precious metal usage, combined with the substitution of toxic cyanide salts with benign potassium ferrocyanide, results in substantial cost savings in raw material procurement and waste disposal fees.
- Cost Reduction in Manufacturing: The elimination of column chromatography is perhaps the most significant economic driver of this technology. Traditional purification methods for heterocyclic intermediates often rely on silica gel chromatography, which is solvent-intensive, time-consuming, and difficult to scale. By achieving high purity through acidic back-extraction and concentration, this method significantly lowers solvent consumption and processing time. Furthermore, the recyclability of the catalyst means that the effective cost per kilogram of product decreases with each reuse cycle, providing a clear path to margin improvement as production volumes increase.
- Enhanced Supply Chain Reliability: The use of commercially available and stable reagents like potassium ferrocyanide and 2-amino terephthalic acid ensures a robust supply chain that is less susceptible to the regulatory restrictions often placed on hazardous chemicals. The simplified workup procedure also shortens the overall cycle time for each batch, allowing for faster turnaround and improved responsiveness to market demand. This agility is crucial for maintaining continuous supply lines for critical oncology medications, reducing the risk of stockouts caused by prolonged manufacturing delays.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process offers a greener alternative to conventional routes. The avoidance of pyrophoric Raney Nickel and toxic free cyanides simplifies regulatory compliance and reduces the burden on wastewater treatment facilities. The heterogeneous nature of the reaction facilitates easier scale-up from laboratory to pilot and commercial scales, as heat and mass transfer issues associated with slurry handling are minimized. This scalability ensures that the technology can meet the growing global demand for Apalutamide intermediates without compromising on safety or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel catalytic system. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on the practical aspects of adopting this technology for industrial applications.
Q: What are the primary advantages of the Pd/Ni-MOF catalyst over homogeneous palladium systems?
A: The supported high-dispersion Pd/Ni co-doped catalyst allows for simple filtration removal, eliminating the need for expensive and complex heavy metal scavenging processes required for GMP compliance. Furthermore, the catalyst demonstrates excellent recyclability, maintaining high activity over multiple cycles, which drastically reduces raw material costs compared to single-use homogeneous catalysts.
Q: How does this method address the safety concerns associated with traditional cyanation reagents?
A: This process utilizes potassium ferrocyanide (K4[Fe(CN)6]) as the cyanation source instead of highly toxic free cyanides like NaCN or KCN. Potassium ferrocyanide is significantly safer to handle, cheaper, and does not require complex pretreatment, thereby enhancing operational safety and reducing regulatory burdens in the manufacturing facility.
Q: Can this synthesis route be scaled for commercial production without column chromatography?
A: Yes, the patent explicitly details a purification strategy involving acidic high-temperature back-extraction that achieves over 99% HPLC purity. This eliminates the bottleneck of column chromatography, making the process highly suitable for large-scale commercial manufacturing where throughput and solvent consumption are critical factors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Cyano-5-nitro-3-(trifluoromethyl)pyridine Supplier
As the pharmaceutical industry continues to evolve towards more efficient and sustainable manufacturing practices, the technology described in CN108314646B stands out as a benchmark for modern intermediate synthesis. NINGBO INNO PHARMCHEM is uniquely positioned to leverage this advanced catalytic methodology to deliver high-quality intermediates for the urogenital therapeutic market. With our state-of-the-art facilities and deep expertise in process chemistry, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that ensure every batch meets the exacting standards required for GMP-grade API synthesis.
We invite global partners to collaborate with us to optimize their supply chains and reduce their cost of goods sold through the adoption of this superior synthetic route. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, and discover how our innovative capabilities can support your long-term strategic goals in the competitive oncology landscape.
