Revolutionizing Secondary Amide Production: A Green Boron-Catalyzed Transamidation Strategy for Industrial Scale-Up
Revolutionizing Secondary Amide Production: A Green Boron-Catalyzed Transamidation Strategy for Industrial Scale-Up
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of modern pharmaceutical and agrochemical manufacturing, where efficiency and sustainability are paramount. A significant breakthrough in this domain is documented in patent CN115304506A, which introduces a novel method for preparing secondary amide compounds through the activation of primary amides using boron salt catalysis. This technology represents a paradigm shift from traditional, resource-intensive protocols to a more streamlined, atom-economical approach. By utilizing boron trifluoride etherate as a catalyst under ambient air conditions, this process achieves high yields of secondary amides without the need for complex pre-activation steps or expensive transition metal catalysts. For industry leaders seeking a reliable secondary amide supplier, understanding the mechanistic advantages and scalability of this patented route is crucial for optimizing supply chains and reducing overall production costs in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the amide bond, particularly the conversion of primary amides to secondary amides, has been fraught with synthetic challenges due to the inherent resonance stability of the amide functional group. Conventional methodologies often necessitate harsh reaction conditions, including elevated temperatures and the use of strong bases, which can compromise sensitive functional groups on the substrate. Furthermore, many established protocols rely heavily on transition metal catalysts, such as nickel complexes, to facilitate the transamidation reaction. While effective in certain contexts, these metal-catalyzed routes introduce significant downstream processing burdens, specifically the rigorous requirement to remove trace metal residues to meet stringent pharmaceutical purity standards. Additionally, the reliance on inert atmospheres and specialized solvents in traditional methods increases operational complexity and safety risks, making them less ideal for large-scale industrial applications where cost reduction in pharmaceutical intermediate manufacturing is a critical objective.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in patent CN115304506A offers a remarkably efficient and environmentally benign alternative. This novel approach leverages the Lewis acidity of boron salts, specifically boron trifluoride etherate, to activate the carbonyl group of the primary amide, thereby lowering the energy barrier for nucleophilic attack by the amine. The reaction proceeds smoothly at a moderate temperature of 80°C and, critically, does not require an inert atmosphere, allowing operations to be conducted under standard air conditions. This simplification drastically reduces the engineering controls needed for production. Moreover, the method exhibits exceptional substrate tolerance, accommodating a wide range of aromatic and aliphatic amines and amides. As illustrated in the general reaction scheme below, the transformation is direct and high-yielding, bypassing the need for additional activating agents or coupling reagents that generate stoichiometric waste.
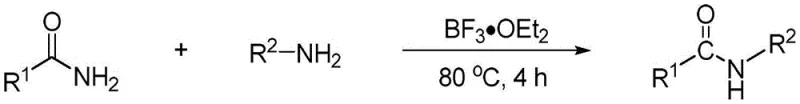
Mechanistic Insights into Boron Salt-Catalyzed Transamidation
The efficacy of this synthetic route lies in the specific interaction between the boron catalyst and the amide substrate. Mechanistically, the boron atom in boron trifluoride etherate acts as a potent Lewis acid, coordinating with the lone pair electrons on the carbonyl oxygen of the primary amide. This coordination withdraws electron density from the carbonyl carbon, rendering it significantly more electrophilic and susceptible to nucleophilic attack by the incoming amine. Unlike transition metal catalysis which often involves complex oxidative addition and reductive elimination cycles, this Lewis acid activation is straightforward and reversible. The absence of redox-active metals means there is no risk of metal-induced side reactions or the formation of difficult-to-remove organometallic impurities. This mechanistic simplicity translates directly to a cleaner reaction profile, which is highly advantageous for R&D teams focused on impurity control and regulatory compliance in high-purity amide intermediates production.
Furthermore, the robustness of this catalytic system allows for excellent control over the reaction selectivity. The mild conditions prevent the degradation of thermally labile functional groups that might otherwise decompose under the harsh conditions required by non-catalyzed thermal transamidation. For instance, the patent data demonstrates successful synthesis of various derivatives, such as N-benzyl-4-methylbenzamide, with yields exceeding 94%. The structural integrity of the products is maintained, as evidenced by detailed spectroscopic analysis including NMR and mass spectrometry. This level of precision ensures that the final API intermediates or agrochemical building blocks meet the rigorous quality specifications required by global markets. The ability to achieve such high conversion rates without extensive purification steps underscores the potential for this chemistry to serve as a platform technology for the commercial scale-up of complex polymer additives and fine chemicals.
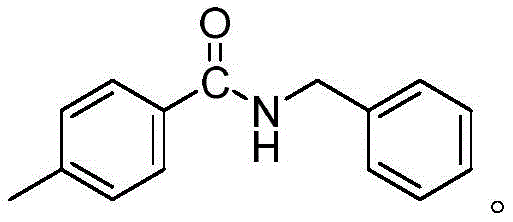
How to Synthesize N-benzyl-4-methylbenzamide Efficiently
Implementing this boron-catalyzed transamidation strategy in a laboratory or pilot plant setting is straightforward and aligns well with green chemistry principles. The protocol typically involves mixing the primary amide and the amine in a specific molar ratio, followed by the addition of the boron trifluoride etherate catalyst. The reaction mixture is then heated to 80°C for a duration of approximately 4 hours, during which the progress can be easily monitored using thin-layer chromatography (TLC). Upon completion, the workup procedure is minimal, often requiring only rotary evaporation to remove volatile components followed by standard column chromatography if high purity is essential. This operational simplicity makes it an attractive candidate for rapid process development. For detailed standardized synthesis steps and specific safety handling procedures, please refer to the guide below.
- Charge a reaction vessel with primary amide, amine, and boron trifluoride etherate in a 1: 2:2 molar ratio under air atmosphere.
- Heat the reaction mixture to 80°C and maintain stirring for approximately 4 hours to ensure complete conversion monitored by TLC.
- Purify the crude product via rotary evaporation followed by silica gel column chromatography using ethyl acetate and petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this boron-catalyzed methodology offers substantial strategic benefits for procurement and supply chain management. The elimination of expensive transition metal catalysts directly impacts the bill of materials, leading to significant cost savings without compromising on reaction efficiency. Moreover, the ability to run reactions under air conditions removes the need for specialized inert gas infrastructure and the associated logistical costs of nitrogen or argon supply. This flexibility enhances supply chain reliability by reducing dependency on specific utility setups that might be bottlenecks in multi-purpose manufacturing facilities. The robustness of the process also implies a lower risk of batch failure due to atmospheric leaks, ensuring consistent delivery schedules for critical intermediates.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the substitution of precious metal catalysts with inexpensive and readily available boron salts. Traditional nickel-catalyzed routes not only incur high raw material costs but also necessitate expensive scavenging resins or complex extraction processes to meet residual metal limits. By avoiding these metals entirely, manufacturers can streamline their downstream processing, reducing solvent consumption and waste disposal fees. Additionally, the solvent-free or low-solvent nature of the reaction minimizes the volume of hazardous waste generated, further lowering environmental compliance costs and contributing to a more sustainable manufacturing footprint.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals as reagents. Boron trifluoride etherate, primary amides, and amines are widely produced bulk chemicals with stable global supply networks, unlike specialized ligands or catalysts that may have single-source suppliers. This abundance ensures that production schedules are not disrupted by raw material shortages. Furthermore, the mild reaction conditions reduce the wear and tear on reactor vessels and heating systems, extending equipment lifespan and reducing maintenance downtime. For supply chain heads, this translates to a more predictable and continuous flow of high-purity amide intermediates, essential for maintaining just-in-time inventory levels.
- Scalability and Environmental Compliance: Scaling chemical processes from gram to ton scale often exposes hidden inefficiencies, but this transamidation method is inherently scalable due to its thermal stability and lack of exothermic hazards associated with strong bases. The high atom economy of the reaction means that the majority of the starting mass is incorporated into the final product, minimizing waste generation. This aligns perfectly with increasingly stringent environmental regulations regarding solvent emissions and heavy metal discharge. Facilities adopting this technology can achieve higher throughput with smaller environmental footprints, facilitating easier permitting and community acceptance for expansion projects aimed at reducing lead time for high-purity amides.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this boron-catalyzed transamidation technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating its suitability for your specific application. Understanding these nuances is vital for technical teams assessing process feasibility and for commercial teams negotiating supply agreements.
Q: What are the advantages of using boron salt catalysis over traditional metal catalysts for transamidation?
A: Traditional methods often require expensive transition metals like nickel or harsh conditions involving strong bases. The boron salt catalyzed method described in patent CN115304506A operates under mild conditions (80°C) in air, eliminating the need for costly metal removal steps and reducing environmental impact.
Q: Is this transamidation process suitable for both aromatic and aliphatic substrates?
A: Yes, the method demonstrates broad substrate scope. It effectively converts various primary amides, including substituted aryl groups like tolyl and naphthyl, as well as alkyl groups, reacting them with both aromatic and aliphatic amines to produce secondary amides in high yields.
Q: Does this synthesis method require an inert atmosphere or specialized solvents?
A: No, one of the key innovations is that the reaction proceeds efficiently under standard air conditions without the need for an inert gas blanket. Furthermore, the process is designed to be solvent-free or requires minimal solvent, significantly simplifying the operational procedure and waste treatment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-benzyl-4-methylbenzamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methods like the boron-salt mediated transamidation described in CN115304506A. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial realities. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. We understand that in the competitive landscape of pharmaceutical and agrochemical intermediates, consistency and purity are non-negotiable, and our infrastructure is designed to deliver exactly that.
We invite you to collaborate with us to leverage this efficient synthesis route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity targets. By partnering with us, you gain access to deep process knowledge and the agility to adapt quickly to market demands. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how we can optimize your supply chain for secondary amide production.
