Advanced Manufacturing of Bisquinazoline Diselenide Anticancer Intermediates for Global Pharma
Introduction to Novel Selenium-Containing Quinazoline Scaffolds
The landscape of oncology drug discovery is continuously evolving, with a renewed focus on heterocyclic compounds incorporating chalcogen elements like selenium to enhance biological potency. Patent CN102627614B introduces a significant advancement in this field by disclosing a series of bisquinazoline diselenide compounds that demonstrate remarkable antitumor properties. These molecules are characterized by a unique structural motif where two quinazoline rings are bridged by a diselenide bond, creating a rigid yet electronically rich framework ideal for interacting with biological targets such as tyrosine kinases. The strategic incorporation of selenium not only modulates the electronic distribution across the quinazoline core but also leverages the known antioxidant and enzyme-modulating capabilities of organoselenium species. For pharmaceutical developers, this patent represents a critical intellectual property foundation for next-generation anticancer agents, offering a versatile platform where substituents at the R1 through R5 positions can be tuned to optimize pharmacokinetics and efficacy.
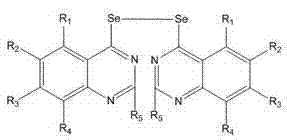
From a supply chain perspective, the accessibility of these complex intermediates is paramount. The patent outlines a synthesis pathway that relies on readily available precursors, mitigating the risks associated with sourcing exotic starting materials. This ensures that as a reliable pharmaceutical intermediate supplier, we can maintain continuity of supply even during global raw material fluctuations. The structural diversity allowed by the general formula means that a single manufacturing platform can produce a library of analogs, accelerating the lead optimization phase for R&D teams. Understanding the precise architecture of these molecules is the first step toward mastering their production, as the diselenide linkage requires careful handling to prevent oxidation or degradation during storage and transport.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of selenium-bridged heterocycles has been fraught with challenges that hinder commercial viability. Traditional methods often involve the use of highly toxic selenium reagents, such as elemental selenium powder in combination with strong reducing agents under harsh conditions, which can lead to poor atom economy and significant safety hazards. Furthermore, conventional routes frequently suffer from low regioselectivity, resulting in complex mixtures of mono-substituted and di-substituted byproducts that are difficult to separate. The reliance on volatile organic solvents and extreme temperatures in older protocols also exacerbates environmental concerns, leading to high waste disposal costs and regulatory scrutiny. For procurement managers, these inefficiencies translate into unpredictable pricing and extended lead times, as purification steps become bottlenecks in the production schedule. Additionally, the instability of intermediate selenolate species often necessitates cryogenic conditions, adding substantial energy costs and operational complexity to the manufacturing process.
The Novel Approach
In contrast, the methodology described in CN102627614B offers a streamlined and robust alternative that addresses these historical pain points. The core innovation lies in the direct nucleophilic substitution of 4-chloroquinazoline derivatives with alkali metal diselenides (M2Se2), where M can be sodium, potassium, or lithium. This approach eliminates the need for in situ generation of unstable selenolates under cryogenic conditions, allowing the reaction to proceed smoothly at moderate temperatures ranging from 20°C to 120°C. The use of common industrial solvents such as ethanol, isopropanol, DMF, or even aqueous mixtures significantly reduces the environmental footprint and simplifies solvent recovery systems. By utilizing 4-chloroquinazoline as the electrophile, the process capitalizes on the high reactivity of the 4-position, ensuring high conversion rates and minimizing the formation of unwanted isomers. This novel route not only enhances safety by avoiding hazardous reagents but also improves overall yield consistency, making it an ideal candidate for cost reduction in API manufacturing.
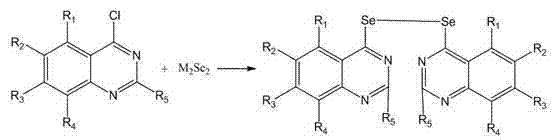
Mechanistic Insights into Diselenide Bond Formation
The chemical mechanism underpinning this synthesis is a classic nucleophilic aromatic substitution (SNAr), facilitated by the electron-deficient nature of the quinazoline ring. The diselenide anion (Se2^2-) acts as a potent nucleophile, attacking the C-4 position of the 4-chloroquinazoline substrate, which is activated by the adjacent nitrogen atoms in the pyrimidine ring. This activation lowers the energy barrier for the displacement of the chloride leaving group, allowing the reaction to proceed efficiently without the need for transition metal catalysts. The absence of heavy metal catalysts is a crucial advantage for pharmaceutical applications, as it removes the necessity for expensive and time-consuming metal scavenging steps to meet strict residual metal limits (e.g., ICH Q3D guidelines). The formation of the Se-Se bond is preserved throughout the reaction, suggesting that the diselenide salt acts as a dianionic equivalent that bridges two quinazoline units simultaneously or sequentially. This mechanistic pathway ensures that the final product retains the specific oxidation state of selenium required for its biological activity, preventing the formation of inactive selenides or over-oxidized selenones.
Impurity control is inherently built into this mechanism due to the high specificity of the SNAr reaction on the quinazoline scaffold. The primary impurities typically arise from incomplete conversion or hydrolysis of the chloro-group, both of which are easily managed through reaction monitoring and optimized workup procedures. The patent data indicates that recrystallization from DMF and water is highly effective at purifying the crude product, yielding orange to brown-red crystals with high purity. This crystallization behavior is driven by the planar nature of the bisquinazoline system and the intermolecular interactions of the diselenide bridge, which promote the formation of a stable crystal lattice. For R&D directors, understanding this mechanism allows for rational modification of the R-groups to further tune solubility and reactivity. For instance, electron-withdrawing groups on the quinazoline ring can further activate the C-4 position, potentially allowing for milder reaction conditions, while bulky substituents might influence the stereochemistry and packing of the final crystal form.
How to Synthesize Bisquinazoline Diselenide Efficiently
Executing this synthesis at a commercial scale requires strict adherence to the parameters outlined in the patent to ensure reproducibility and safety. The process begins with the preparation of the diselenide source, which can be generated in situ from elemental selenium and a reducing agent like sodium borohydride, or introduced directly as a stable salt. The reaction is typically conducted in a stirred vessel equipped with a reflux condenser, where the temperature is carefully controlled to maintain the optimal kinetic window of 20-120°C. The addition of the 4-chloroquinazoline derivative is performed in batches to manage the exotherm and ensure complete consumption of the nucleophile. Following the reaction period of 3-36 hours, the mixture is cooled, and the product precipitates or is isolated via filtration. The detailed standardized synthesis steps, including specific molar ratios and solvent volumes for various derivatives, are provided in the guide below to assist technical teams in replicating these results.
- Prepare a solution of sodium, potassium, or lithium diselenide in a suitable solvent such as ethanol, DMF, or aqueous mixtures under inert atmosphere.
- Add 4-chloroquinazoline derivatives to the diselenide solution in batches while maintaining temperature between 20-120°C.
- Reflux the mixture for 3-36 hours, cool, filter the solid product, and recrystallize using DMF and water to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the significant simplification of the raw material portfolio. By relying on 4-chloroquinazoline derivatives and simple alkali metal salts, manufacturers can source inputs from a broad base of global suppliers, reducing dependency on single-source vendors and mitigating supply disruption risks. The flexibility in solvent selection—allowing for the use of ethanol or water mixtures—further enhances supply chain resilience, as these commodities are less prone to market volatility compared to specialized halogenated solvents. Moreover, the elimination of transition metal catalysts translates directly into cost reduction in API manufacturing by removing the need for costly palladium or copper ligands and the associated analytical testing for metal residues. This streamlining of the bill of materials allows for more accurate cost forecasting and tighter margin control in long-term supply agreements.
- Cost Reduction in Manufacturing: The process design inherently lowers operational expenditures by utilizing ambient pressure conditions and moderate temperatures, which reduces energy consumption compared to high-pressure hydrogenation or cryogenic reactions. The high atom economy of the substitution reaction minimizes waste generation, leading to lower disposal fees and a smaller environmental footprint. Furthermore, the straightforward purification via recrystallization avoids the need for complex chromatographic separations, which are often the most expensive unit operation in fine chemical synthesis. These factors combine to create a highly cost-efficient production model that can withstand pricing pressures in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The robustness of the chemistry ensures consistent batch-to-batch quality, which is critical for maintaining regulatory compliance and avoiding production delays. The use of stable intermediates and common solvents means that inventory can be held safely for longer periods without degradation, allowing for better demand planning and buffer stock management. This reliability extends to the scalability of the process; since the reaction does not rely on sensitive catalysts that might deactivate upon scale-up, the transition from pilot plant to commercial production is seamless. Consequently, lead times for high-purity pharmaceutical intermediates can be significantly shortened, enabling faster time-to-market for downstream drug products.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this route is superior to traditional methods that generate hazardous selenium byproducts. The ability to use aqueous workups and green solvents aligns with modern sustainability goals and regulatory trends towards greener chemistry. The solid nature of the final product facilitates safe handling, packaging, and transport, reducing the risks associated with liquid intermediates. Scalability is further supported by the wide operating temperature window, which provides process engineers with the flexibility to optimize throughput without compromising safety. This makes the technology suitable for commercial scale-up of complex heterocyclic compounds ranging from 100 kgs to 100 MT annual capacities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of bisquinazoline diselenide compounds. These answers are derived directly from the experimental data and claims within patent CN102627614B, providing a factual basis for decision-making. Understanding these nuances is essential for partners looking to integrate these intermediates into their oncology pipelines. We encourage technical teams to review these points closely to assess the fit for their specific project requirements.
Q: What are the primary biological activities of bisquinazoline diselenide compounds?
A: According to patent CN102627614B, these compounds exhibit significant antitumor activity, specifically showing proliferation inhibitory effects on breast cancer cell lines like MDA-MB-435, with some derivatives demonstrating superior efficacy compared to standard controls like oxaliplatin.
Q: Can the synthesis process be scaled for commercial production?
A: Yes, the process utilizes common industrial solvents like ethanol and DMF and operates at moderate temperatures (20-120°C), making it highly amenable to scale-up from laboratory grams to multi-ton commercial production without requiring exotic high-pressure equipment.
Q: How is product purity ensured in this synthetic route?
A: The protocol specifies a robust purification strategy involving suction filtration followed by recrystallization from N,N-dimethylformamide and water mixtures, which effectively removes unreacted starting materials and inorganic salts to achieve high-purity specifications required for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bisquinazoline Diselenide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving anticancer therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We are committed to delivering stringent purity specifications through our rigorous QC labs, which are equipped with advanced analytical instrumentation to verify identity, assay, and impurity profiles. Our facility is designed to handle reactive chemistries safely, including selenium-based transformations, adhering to the highest international safety and environmental standards. By partnering with us, you gain access to a supply chain that is both resilient and responsive, capable of adapting to the dynamic demands of the pharmaceutical industry.
We invite you to engage with our technical procurement team to discuss your specific requirements for bisquinazoline diselenide derivatives. Whether you need a Customized Cost-Saving Analysis for your current route or require specific COA data to support your regulatory filings, we are ready to assist. We can also provide route feasibility assessments to help you optimize your synthesis strategy for maximum efficiency. Contact us today to request a quote and discover how our expertise in fine chemical manufacturing can accelerate your drug development timeline while reducing overall project costs.
