Advanced Synthesis of Furan Macrocyclic Compounds for Optical and Pharma Applications
Advanced Synthesis of Furan Macrocyclic Compounds for Optical and Pharma Applications
The landscape of heterocyclic macrocycle synthesis is undergoing a significant transformation driven by the urgent need for more efficient and atom-economical processes in both pharmaceutical and electronic material sectors. Patent CN113214283B introduces a groundbreaking preparation method for furan macrocyclic compounds that addresses the longstanding challenges associated with constructing these complex molecular skeletons. Traditionally, the synthesis of such structures has been plagued by low yields, harsh reaction conditions, and the necessity for tedious pre-functionalization steps. This new technology leverages a highly efficient metal-catalyzed ring-forming reaction starting from macrocyclic alkyne alkenal compounds, achieving an atom utilization rate of up to 100 percent. By streamlining the synthetic pathway, this innovation not only enhances the feasibility of producing high-purity furan macrocyclic compounds but also opens new avenues for their application as base units in the synthesis of oligofuran compounds with promising optical properties. As a reliable fine chemical intermediates supplier, understanding such pivotal technological shifts is crucial for maintaining competitiveness in the global market.
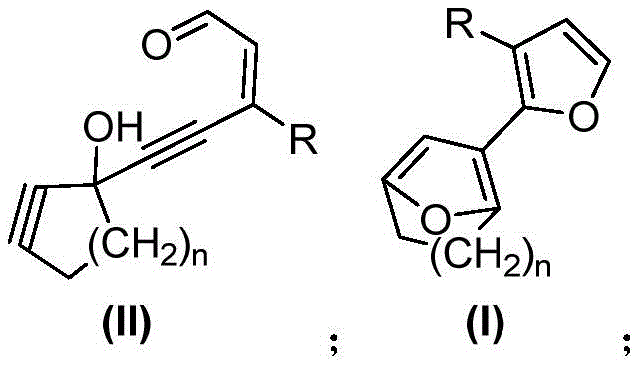
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of furan macrocyclic structures has relied heavily on methods that are inherently inefficient and operationally complex. The first category of conventional techniques typically involves starting with a furan ring and introducing side chains on both sides, followed by ring closure through reactions such as Nozaki-Hiyama-Kishi, metathesis, Michael, or Wittig reactions. These approaches suffer from a critical drawback: they require the selective pre-functionalization of side chains prior to cyclization, which drastically increases the number of synthetic steps and reduces the overall yield. Furthermore, the second category, which attempts to construct the furan ring from non-furan precursors, often utilizes methods like the Paal-Knorr reaction or mercury-catalyzed cycloisomerization. While effective in specific contexts, these older methodologies frequently involve toxic reagents, stringent condition controls, and limited substrate scope, making them less suitable for the commercial scale-up of complex heterocyclic scaffolds required by modern industry standards.
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes a direct and elegant cyclization strategy. The process begins with a macrocyclic alkynal-enal compound, which undergoes a ring-forming reaction under the catalysis of gold or platinum metals. This method eliminates the need for extensive pre-functionalization, allowing for a much more direct assembly of the macrocyclic framework. The versatility of this approach is demonstrated by its tolerance to various substituents; the methylene groups within the macrocycle can be replaced by heteroatoms like oxygen or nitrogen, or even unsaturated bonds, without compromising the reaction efficiency. This flexibility is paramount for cost reduction in electronic chemical manufacturing, as it allows for the rapid generation of diverse libraries of compounds from readily available starting materials such as cyclic ketones or alkyl alcohols. The simplicity of the operation and the high efficiency of the reaction make this a superior choice for industrial applications.
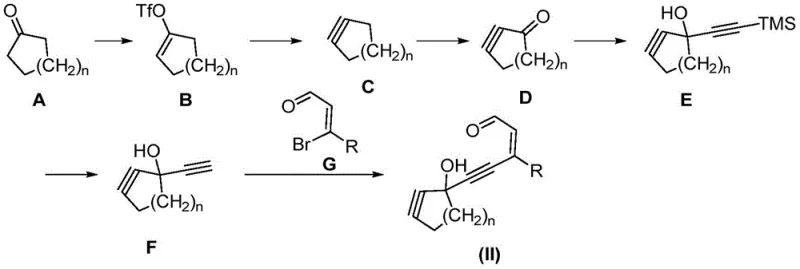
Mechanistic Insights into Metal-Catalyzed Cycloisomerization
The core of this technological breakthrough lies in the mechanism of the metal-catalyzed cycloisomerization. When the macrocyclic alkynal-enal precursor is exposed to a gold or platinum catalyst, the metal center coordinates with the alkyne moiety, activating it towards nucleophilic attack. This activation lowers the energy barrier for the subsequent intramolecular cyclization, where the carbonyl oxygen or an enol equivalent attacks the activated alkyne. This process leads to the formation of the furan ring directly within the macrocyclic structure. The use of catalysts such as IPrAuBF4, PPh3AuSbF6, PtCl4, or PtCl2 ensures high selectivity and turnover, minimizing the formation of by-products. The reaction can be further promoted by the addition of protonation promoting additives like methanol or isopropanol, which facilitate the final aromatization or stabilization of the furan ring. This mechanistic pathway is distinct from radical-based or high-temperature thermal cyclizations, offering a much cleaner reaction profile that is easier to control on a large scale.
From an impurity control perspective, this catalytic system offers significant advantages. Traditional methods often generate complex mixtures of regioisomers or polymeric by-products due to the high reactivity of intermediates under harsh conditions. However, the mild conditions employed in this novel method (typically 40-100°C in solvents like toluene or dichloroethane) suppress side reactions. The high atom economy implies that nearly all atoms from the starting materials are incorporated into the final product, drastically reducing the burden on downstream purification processes. For R&D teams focused on developing high-purity furan macrocyclic compounds, this means fewer chromatography steps and higher overall recovery rates. The ability to tolerate diverse functional groups on the aryl ring (R group), including halogens, nitro groups, and alkoxy groups, further underscores the robustness of this catalytic cycle, ensuring consistent quality across different derivatives.
How to Synthesize Furan Macrocyclic Compounds Efficiently
The synthesis of these valuable compounds follows a logical progression from simple cyclic ketones to the final macrocyclic furan. The process begins with the conversion of a macrocyclic cyclic ketone into an enol triflate, which is then transformed into an internal alkyne. Subsequent oxidation and coupling reactions install the necessary aldehyde and alkyne functionalities to create the key macrocyclic alkynal-enal precursor. The final step involves the critical metal-catalyzed cyclization. This streamlined route avoids the pitfalls of traditional macrocyclization, such as high dilution requirements or unstable intermediates. For detailed procedural specifics regarding reagent quantities, temperature profiles, and workup procedures, please refer to the standardized synthesis guide provided below, which outlines the exact operational parameters validated in the patent examples.
- Convert macrocyclic cyclic ketone to macrocyclic enol triflate using N-phenyl bis(trifluoromethanesulfonyl) imide and LHMDS.
- Transform the enol triflate into a macrocyclic internal alkyne using lithium chloride, followed by oxidation to a macrocyclic alkynone.
- Perform nucleophilic addition with trimethylsilyl acetylene, deprotect, and couple with an aryl bromo-aldehyde to form the macrocyclic alkynal-enal precursor.
- Execute the final ring-closing reaction using a gold or platinum catalyst in an organic solvent to yield the target furan macrocycle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method translates into tangible strategic benefits beyond mere technical novelty. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Since the starting materials are common cyclic ketones or alkyl alcohols, sourcing is straightforward and less susceptible to the volatility associated with specialized, custom-synthesized intermediates. This stability in raw material availability directly contributes to enhanced supply chain reliability, ensuring that production schedules can be maintained without unexpected delays caused by precursor shortages. Furthermore, the elimination of toxic heavy metal catalysts like mercury, which were used in older cycloisomerization methods, simplifies waste management and regulatory compliance, reducing the environmental footprint of the manufacturing process.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the high atom economy and the reduction in synthetic steps. By avoiding the need for pre-functionalizing side chains and utilizing a direct cyclization strategy, the consumption of reagents and solvents is significantly minimized. The removal of expensive and hazardous reagents typically associated with macrocyclization further drives down the cost of goods sold. Additionally, the high yields reported in the patent examples mean that less raw material is wasted, leading to substantial cost savings in the overall production budget. This efficiency makes the commercial production of these complex molecules financially viable for the first time.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent output quality, which is critical for maintaining long-term contracts with downstream clients in the pharmaceutical and electronic materials sectors. The tolerance of the reaction to various substituents allows for a flexible manufacturing platform where different derivatives can be produced using the same core infrastructure. This adaptability reduces the lead time for high-purity optical materials, enabling faster response to market demands. Moreover, the scalability of the process from gram to kilogram scales without significant loss in efficiency guarantees a steady supply of materials for clinical trials or pilot production runs.
- Scalability and Environmental Compliance: As the industry moves towards greener chemistry, this method stands out for its environmental compatibility. The use of gold or platinum catalysts, while precious, allows for low loading levels (1-10 mol%), and the reactions proceed in standard organic solvents that are easily recoverable. The absence of stoichiometric toxic waste streams simplifies the treatment of effluent, aligning with strict environmental regulations. This ease of scale-up from laboratory to commercial production (100 kgs to 100 MT) ensures that the technology can meet the growing demand for advanced functional materials without compromising on safety or sustainability standards.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and commercial viability of this synthesis method. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is essential for partners looking to integrate these intermediates into their own development pipelines.
Q: What are the primary advantages of this new cyclization method over traditional macrocyclization techniques?
A: Unlike traditional methods such as Nozaki-Hiyama-Kishi or metathesis reactions which require extensive pre-functionalization of side chains, this novel approach utilizes a direct metal-catalyzed cycloisomerization of macrocyclic alkynal-enals. This results in significantly higher atom economy (up to 100%), simpler operational steps, and milder reaction conditions, thereby reducing the overall complexity and cost of producing complex heterocyclic scaffolds.
Q: Can the methylene groups in the macrocyclic ring be substituted without affecting the reaction?
A: Yes, experimental data confirms that the cyclization reaction is robust against structural variations. One or more methylene groups in the (CH2)n chain can be replaced by heteroatoms such as oxygen (-O-) or nitrogen (-NTs-), and adjacent methylene groups can even be replaced by unsaturated bonds like -CH=CH- or -C≡C- without hindering the ring formation, offering great flexibility for designing diverse molecular architectures.
Q: What are the potential applications of the synthesized furan macrocyclic compounds?
A: These compounds serve as excellent structural units for synthesizing oligofuran compounds, which exhibit superior fluorescence properties. They hold significant potential in the fields of optical materials, including hydrological detection, nondestructive inspection, biological tracking, and fluorescent probes, due to their large Stokes shifts and high fluorescence efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Furan Macrocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this novel synthesis technology in advancing the fields of optical materials and pharmaceutical intermediates. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering high-purity furan macrocyclic compounds that meet stringent purity specifications, supported by our rigorous QC labs which employ state-of-the-art analytical instrumentation to verify every batch. Our capability to handle complex heterocyclic scaffolds positions us as a strategic partner for companies seeking to innovate in the realm of functional materials.
We invite you to explore how this advanced manufacturing process can optimize your supply chain and reduce your overall production costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can accelerate your development timeline and secure your position in the competitive global market.
