Advanced Stereoselective Transaminase Technology for Commercial Chiral Amine Production
The pharmaceutical industry continuously seeks innovative pathways to manufacture high-purity chiral amines, which serve as critical building blocks for numerous life-saving medications. Patent CN110914417B introduces a groundbreaking stereoselective transaminase derived from Mycobacterium species that addresses long-standing challenges in asymmetric synthesis. This biocatalyst exhibits strict R-stereoselectivity and a remarkably broad substrate spectrum, enabling the efficient conversion of prochiral carbonyl compounds into valuable chiral amines under mild conditions. For R&D Directors and Procurement Managers, this technology represents a significant shift away from traditional metal-catalyzed processes, offering a greener, more sustainable, and potentially more cost-effective route for producing complex pharmaceutical intermediates such as Sitagliptin precursors. The ability to achieve high optical purity without the need for expensive chiral resolution steps or toxic heavy metals positions this enzymatic approach as a superior alternative for modern drug manufacturing supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of chiral amines has relied heavily on chemical asymmetric synthesis utilizing transition metal catalysts such as rhodium complexes. These conventional methods present substantial drawbacks, including the high cost of precious metal catalysts and the stringent requirement for their complete removal from the final product to meet regulatory safety standards. The presence of metal residues necessitates additional purification steps, such as specialized filtration or chromatography, which increase both production time and operational expenses. Furthermore, chemical hydrogenation often requires harsh reaction conditions, including high pressure and elevated temperatures, posing safety risks and limiting the compatibility with sensitive functional groups found in complex drug molecules. The environmental impact of metal waste disposal also adds a significant burden to the overall manufacturing footprint, making these traditional routes less attractive in an era of increasing green chemistry regulations.
The Novel Approach
In contrast, the novel biocatalytic approach disclosed in the patent utilizes a highly active omega-transaminase that operates efficiently under ambient temperature and atmospheric pressure. This enzymatic method eliminates the need for transition metals entirely, thereby removing the risk of metal contamination and the associated costly removal processes. The transaminase employs pyridoxal phosphate as a cofactor, which is stable and does not require complex regeneration cycles, simplifying the reaction setup significantly. By leveraging the inherent stereospecificity of the enzyme, manufacturers can achieve high enantiomeric excess directly during the synthesis step, reducing the need for downstream chiral separation. This shift from chemical catalysis to biocatalysis not only enhances the safety profile of the manufacturing process but also aligns with global sustainability goals by reducing energy consumption and hazardous waste generation.
Mechanistic Insights into Omega-Transaminase Catalyzed Asymmetric Synthesis
The core of this technology lies in the unique mechanistic action of the omega-transaminase, which facilitates the reversible transfer of an amino group from an amino donor to a prochiral carbonyl acceptor. Unlike alpha-transaminases that are limited to alpha-amino groups, this omega-transaminase can catalyze the transfer of non-alpha amino groups, granting it access to a wider range of substrate structures essential for pharmaceutical synthesis. The enzyme demonstrates strict R-stereoselectivity, a trait that is relatively rare among natural transaminases, making it particularly valuable for synthesizing specific drug intermediates that require the R-configuration. The catalytic cycle involves the formation of a Schiff base intermediate with the pyridoxal phosphate cofactor, ensuring precise spatial orientation of the substrate to guarantee high optical purity of the resulting amine product.
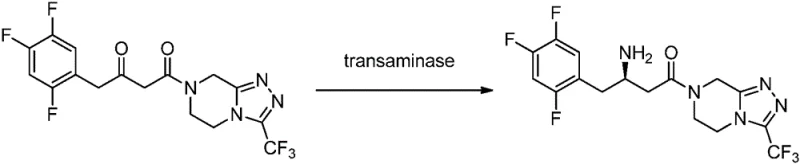
Impurity control is inherently superior in this enzymatic system due to the high specificity of the biocatalyst towards the target carbonyl group. In chemical synthesis, side reactions such as over-reduction or racemization can occur, leading to complex impurity profiles that are difficult to separate. The transaminase, however, recognizes specific structural features of the substrate, minimizing the formation of by-products and ensuring a cleaner reaction profile. This high selectivity translates directly to reduced purification burdens for the supply chain team, as fewer chromatographic steps are required to achieve the stringent purity specifications demanded by regulatory agencies. The ability to maintain high conversion rates while preserving stereochemical integrity makes this process robust and reliable for the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Chiral Amines Efficiently
The synthesis protocol outlined in the patent provides a clear pathway for implementing this technology in a production environment, starting with the cultivation of the engineered host strain. The process involves expressing the specific transaminase polypeptide in a robust bacterial host, such as Escherichia coli BL21, which allows for high-yield production of the biocatalyst through standard fermentation techniques. Once the enzyme is prepared, the reaction is conducted in a liquid system where the prochiral carbonyl compound is mixed with an amino donor in the presence of the cofactor. Detailed standardized synthesis steps see the guide below.
- Cultivate the engineered Escherichia coli BL21 strain expressing the specific transaminase polypeptide under controlled fermentation conditions to accumulate the biocatalyst.
- Conduct the asymmetric transamination reaction in a liquid system using prochiral carbonyl compounds and an amino donor, maintaining pH 7.5-8.5 and temperature 25-35°C.
- Separate the resulting high-purity chiral amine from the reaction mixture through standard extraction or crystallization processes, optionally recycling the aqueous phase.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this transaminase technology offers tangible strategic advantages beyond mere technical feasibility. The elimination of expensive noble metal catalysts directly impacts the bill of materials, leading to substantial cost savings in raw material procurement without compromising on quality. Additionally, the mild reaction conditions reduce energy consumption and equipment wear, contributing to lower operational expenditures over the lifecycle of the product. The simplified downstream processing, resulting from the absence of metal residues and high reaction selectivity, shortens the overall production cycle time, enhancing the responsiveness of the supply chain to market demands. These factors collectively improve the margin structure and reliability of the supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The removal of rhodium or other precious metal catalysts from the process workflow eliminates a significant cost driver associated with traditional chemical synthesis. Without the need for specialized metal scavenging resins or extensive washing protocols to meet residual metal limits, the overall processing cost is drastically simplified. This reduction in auxiliary material consumption and waste treatment expenses allows for a more competitive pricing structure for the final chiral amine products. Furthermore, the high efficiency of the enzyme means that less catalyst is required per unit of product, optimizing the utilization of biological resources and driving down the variable cost of goods sold significantly.
- Enhanced Supply Chain Reliability: Relying on fermentation-derived enzymes reduces dependency on the volatile market prices of precious metals, which are subject to geopolitical fluctuations and mining constraints. The biological production of the catalyst can be scaled up rapidly using established industrial fermentation infrastructure, ensuring a consistent and secure supply of the critical reagent. This stability in raw material availability mitigates the risk of production delays caused by catalyst shortages, thereby guaranteeing continuous manufacturing operations. The robustness of the E. coli expression system also ensures that the biocatalyst can be produced in large quantities to meet surging demand for key drug intermediates without bottlenecks.
- Scalability and Environmental Compliance: The process operates under mild aqueous conditions, which significantly reduces the generation of hazardous organic solvent waste compared to traditional organic synthesis routes. This alignment with green chemistry principles simplifies environmental compliance and reduces the costs associated with waste disposal and regulatory reporting. The scalability of the fermentation process allows for seamless transition from pilot scale to multi-ton commercial production, supporting the growing volume requirements of blockbuster drugs. By adopting this eco-friendly technology, companies can enhance their corporate sustainability profiles while maintaining high efficiency and productivity in their manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this stereoselective transaminase technology in pharmaceutical manufacturing. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation to provide clarity on its practical application. Understanding these aspects helps stakeholders evaluate the feasibility and advantages of integrating this biocatalytic route into their existing supply chains.
Q: How does this transaminase technology eliminate heavy metal residues compared to traditional chemical synthesis?
A: Traditional methods often rely on expensive rhodium or other transition metal catalysts which leave toxic residues requiring complex removal steps. This patented biocatalytic process uses a protein-based enzyme with pyridoxal phosphate as a cofactor, completely eliminating heavy metal contamination risks and simplifying downstream purification for pharmaceutical grade compliance.
Q: What is the substrate scope of this stereoselective transaminase for industrial applications?
A: The enzyme demonstrates a broad substrate spectrum, effectively catalyzing the conversion of various prochiral carbonyl compounds including ketones with trifluorophenyl groups and heterocyclic structures. This versatility allows it to produce key intermediates for drugs like Sitagliptin and Dolutegravir with high stereoselectivity.
Q: Is this biocatalytic process scalable for commercial manufacturing of pharmaceutical intermediates?
A: Yes, the patent describes the use of robust E. coli expression systems and mild reaction conditions (ambient temperature and pressure) which are inherently suitable for large-scale fermentation and batch processing. The stability of the enzyme and the simplicity of the reaction setup facilitate reliable commercial scale-up without the safety hazards associated with high-pressure chemical hydrogenation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amines Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced biocatalytic patents into commercial reality, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team possesses the expertise to optimize the fermentation and reaction conditions described in CN110914417B to ensure stringent purity specifications are met consistently. With rigorous QC labs and state-of-the-art manufacturing facilities, we are equipped to handle the complexities of chiral amine synthesis, delivering high-quality intermediates that comply with global regulatory standards. Our commitment to technological innovation ensures that our clients benefit from the latest advancements in green chemistry and process efficiency.
We invite you to contact our technical procurement team to discuss how this transaminase technology can be tailored to your specific project needs. Request a Customized Cost-Saving Analysis to understand the potential economic benefits of switching to this enzymatic route for your chiral amine requirements. We are ready to provide specific COA data and route feasibility assessments to support your decision-making process. Partner with us to leverage cutting-edge biocatalysis for a more sustainable and cost-effective supply chain.
