Advanced Manufacturing of Chiral Alpha-Chloroaziridine Intermediates for Global Pharma Supply Chains
The global demand for high-purity chiral building blocks in the pharmaceutical sector continues to drive innovation in asymmetric synthesis methodologies. A pivotal advancement in this domain is documented in Chinese patent CN103694157A, which discloses a highly efficient method for preparing chiral α-chloroaziridines. This technology represents a significant leap forward for manufacturers seeking reliable pharmaceutical intermediate supplier partnerships, as it addresses long-standing challenges regarding stereocontrol and process safety. The core innovation lies in the utilization of chiral (Rs)-N-(tert-butylsulfinyl)imines reacting with trimethyl(dichloromethyl)silane (TMSCCl2H) in the presence of a base. By operating within a温和 temperature window of -80°C to 30°C, this protocol achieves remarkable optical purity without the need for extreme cryogenic conditions often associated with sensitive organometallic chemistry. For R&D directors evaluating new synthetic routes, the ability to generate these strained three-membered rings with high diastereoselectivity offers a robust platform for developing next-generation bioactive molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of α-chloroaziridines has been fraught with significant technical and safety hurdles that impede large-scale manufacturing. Traditional approaches often rely on the generation of chlorocarbene species, which are inherently unstable and hazardous to handle in bulk quantities, posing severe risks to plant safety and operational continuity. Furthermore, alternative strategies involving Darzens-type reactions between imines and reactive dichloromethyl compounds frequently suffer from poor regioselectivity and low stereoselectivity. These deficiencies result in complex product mixtures that require extensive and costly purification steps, drastically inflating the cost reduction in API manufacturing calculations. Additionally, methods involving the addition of nitrenes to chloroalkenes or reactions with acid chlorides often lack the generality required for diverse substrate scopes, limiting their utility in the rapid iteration cycles of modern drug discovery. Consequently, the industry has long sought a method that combines safety, efficiency, and high fidelity in stereochemical outcomes.
The Novel Approach
The methodology outlined in patent CN103694157A fundamentally transforms this landscape by leveraging the powerful directing effects of the tert-butylsulfinyl group. This novel approach utilizes a straightforward condensation of chiral sulfinyl imines with TMSCCl2H, facilitated by common bases such as sodium tert-butoxide or lithium bis(trimethylsilyl)amide. The reaction proceeds smoothly in standard organic solvents like tetrahydrofuran (THF), toluene, or dimethylformamide (DMF), eliminating the need for exotic or prohibitively expensive reagents. As illustrated in the general reaction scheme below, the process delivers the desired chiral α-chloroaziridine scaffolds with exceptional efficiency.
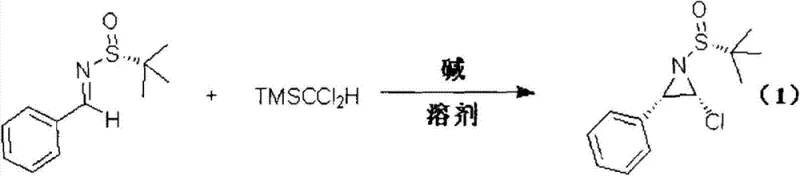
The versatility of this system is evidenced by its tolerance for a wide array of functional groups, including aryl, alkyl, alkenyl, and heteroaryl substituents. Experimental data from the patent indicates that yields can reach up to 90% with diastereomeric ratios (dr) consistently exceeding 98:2, and often reaching 99:1. This level of precision effectively bypasses the need for difficult chiral separations downstream, providing a streamlined pathway for the commercial scale-up of complex pharmaceutical intermediates. For supply chain heads, this translates to a more predictable production timeline and reduced waste generation, aligning perfectly with modern green chemistry initiatives.
Mechanistic Insights into Sulfinyl Imine-Mediated Cyclization
To fully appreciate the value of this technology for high-purity OLED material or pharmaceutical synthesis, one must understand the underlying mechanistic drivers. The reaction initiates with the deprotonation of trimethyl(dichloromethyl)silane by the base to generate a nucleophilic dichloromethyl anion equivalent. This species attacks the electrophilic carbon of the chiral sulfinyl imine. The bulky tert-butylsulfinyl group plays a critical role here, creating a rigid chiral environment that dictates the facial selectivity of the nucleophilic attack. Following the initial addition, an intramolecular nucleophilic substitution occurs where the nitrogen lone pair displaces one of the chloride atoms, closing the three-membered aziridine ring. This concerted mechanism ensures that the stereochemical information encoded in the starting imine is faithfully transferred to the final aziridine product. The stability of the sulfinyl group under the reaction conditions prevents racemization, ensuring that the optical integrity of the molecule is maintained throughout the transformation.
Furthermore, the impurity profile of this reaction is remarkably clean compared to carbene-based methods. Because the reaction does not involve free radical intermediates or highly reactive singlet carbenes, side reactions such as polymerization or non-specific insertion into C-H bonds are minimized. The primary byproducts are typically siloxanes and salts derived from the base, which are easily removed during the aqueous workup and subsequent column chromatography. This high level of chemoselectivity is crucial for reducing lead time for high-purity pharmaceutical intermediates, as it simplifies the isolation process. For quality control teams, the consistent appearance of characteristic NMR signals and mass spectrometry data across different substrates confirms the robustness of the mechanism, allowing for reliable batch-to-batch reproducibility essential for GMP manufacturing environments.
How to Synthesize Chiral Alpha-Chloroaziridine Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and stereoselectivity. The patent provides detailed embodiments covering a broad range of substrates, from simple phenyl imines to complex heterocycles like furan and pyridine derivatives. The standardized protocol involves dissolving the chiral imine and the silane reagent in an anhydrous solvent, cooling the mixture to the specified temperature (often between -20°C and 0°C for optimal balance of rate and selectivity), and then adding the base solution dropwise. Detailed standardized synthesis steps are provided in the guide below to assist process chemists in replicating these results.
- Prepare the reaction mixture by dissolving chiral (Rs)-N-(tert-butylsulfinyl)imine and trimethyl(dichloromethyl)silane (TMSCCl2H) in a suitable organic solvent such as THF or toluene.
- Cool the reaction system to a temperature between -80°C and 30°C depending on the specific substrate reactivity, then slowly add a strong base like sodium tert-butoxide or potassium bis(trimethylsilyl)amide.
- Stir the reaction for 0.5 to 10 hours to ensure complete cyclization, then quench with water and purify the resulting chiral alpha-chloroaziridine via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this patented methodology offers tangible economic and operational benefits that extend beyond mere chemical elegance. The shift towards this sulfinyl imine-based route addresses several critical pain points in the sourcing of chiral nitrogen heterocycles. By utilizing commercially available starting materials and avoiding hazardous reagents, companies can mitigate supply risks and reduce the regulatory burden associated with handling dangerous chemicals. The following points detail how this technology drives value across the organization.
- Cost Reduction in Manufacturing: The most significant financial advantage stems from the elimination of expensive chiral resolution steps. Since the reaction produces products with diastereomeric ratios often exceeding 99:1, the need for preparative chiral HPLC or multiple recrystallizations is virtually eliminated. This drastic simplification of the purification train reduces solvent consumption, labor hours, and equipment occupancy time. Furthermore, the reagents used, such as TMSCCl2H and various tert-butoxides, are commodity chemicals available from multiple global suppliers, preventing single-source bottlenecks and ensuring competitive pricing. The mild reaction conditions also imply lower energy consumption for heating or deep cooling, contributing to a leaner overall cost structure for the production of these valuable intermediates.
- Enhanced Supply Chain Reliability: The robustness of this chemical process directly translates to improved supply security. Unlike methods relying on unstable intermediates that must be generated in situ with tight controls, this route uses stable, shelf-stable reagents that can be stocked in bulk. The wide tolerance for solvents—including ethers, hydrocarbons, and polar aprotic solvents—means that production can continue even if a specific solvent faces temporary market shortages. Additionally, the short reaction times, ranging from 0.5 to 10 hours, allow for high throughput in existing reactor fleets. This agility enables manufacturers to respond rapidly to fluctuating demand from downstream API producers, ensuring that critical project timelines are met without delay.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this method is superior to legacy technologies. The avoidance of chlorocarbene precursors removes the risk of generating toxic gas leaks, a major concern for plant safety audits. The waste stream is primarily composed of benign siloxanes and inorganic salts, which are easier to treat and dispose of compared to the heavy metal waste often associated with transition-metal catalyzed alternatives. This alignment with green chemistry principles facilitates smoother regulatory approvals and reduces the environmental compliance costs associated with waste treatment. The process has been demonstrated to work effectively on scales ranging from grams to multi-gram batches in the patent examples, indicating a clear path for linear scale-up to tonnage production without fundamental changes to the chemistry.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is vital for stakeholders evaluating its integration into their supply chains. The following questions address common inquiries regarding the practical application, scope, and downstream utility of the technology described in patent CN103694157A. These answers are derived directly from the experimental data and technical specifications provided in the intellectual property documentation.
Q: What are the primary advantages of this synthesis method over traditional chlorocarbene routes?
A: Unlike traditional methods utilizing hazardous chlorocarbene or non-selective Darzens-type reactions, this patented approach employs chiral tert-butylsulfinyl imines. This ensures exceptional stereocontrol with diastereomeric ratios (dr) often exceeding 99:1, significantly reducing the need for costly chiral separation processes.
Q: Is this process scalable for industrial production of API intermediates?
A: Yes, the process utilizes commercially available reagents like TMSCCl2H and operates under mild temperatures ranging from -80°C to 30°C. The use of common organic solvents such as THF and toluene, combined with robust base systems, makes the protocol highly adaptable for commercial scale-up in standard reactor setups.
Q: What is the downstream utility of the synthesized alpha-chloroaziridines?
A: These compounds serve as versatile building blocks for nitrogen-containing heterocycles. As demonstrated in the patent data, they can be efficiently converted into unsubstituted aziridines or other complex amine derivatives through selective ring-opening or reduction reactions, making them critical for drug discovery pipelines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Alpha-Chloroaziridine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process expertise and unwavering commitment to quality. Our team of expert process chemists has extensively analyzed the sulfinyl imine route and is fully prepared to adapt this technology to meet your specific project requirements. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of chiral alpha-chloroaziridine meets the highest international standards for enantiomeric excess and chemical purity.
We invite you to leverage our technical capabilities to optimize your supply chain for nitrogen-containing heterocycles. Whether you require custom synthesis of specific derivatives or large-scale toll manufacturing, our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. By collaborating with us, you gain access to a secure, compliant, and cost-effective source of high-value chiral building blocks, empowering your R&D efforts to bring life-saving therapies to market faster.
