Advanced Manufacturing of 2-Arylimidazo[2,1-a]isoquinoline for Scalable Pharmaceutical Applications
Advanced Manufacturing of 2-Arylimidazo[2,1-a]isoquinoline for Scalable Pharmaceutical Applications
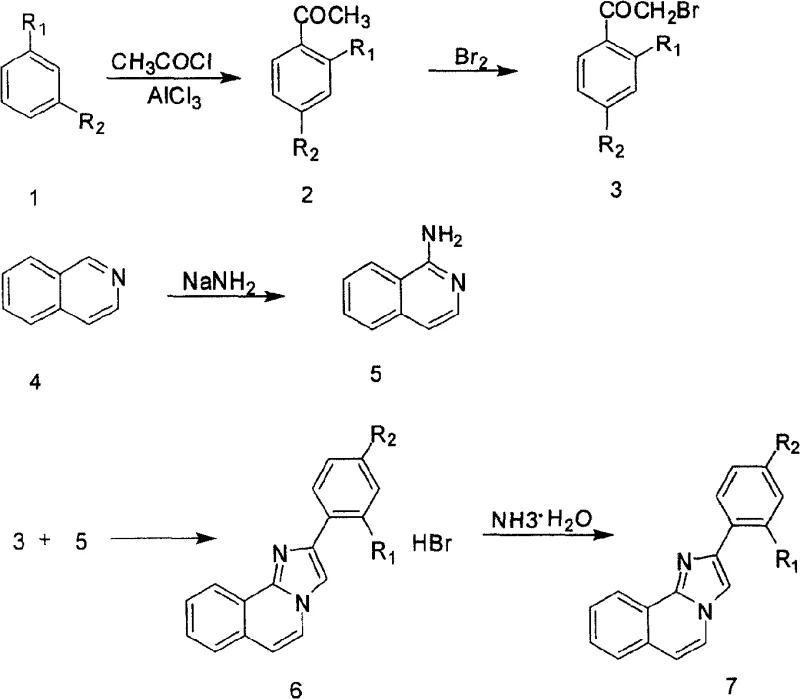
The development of non-steroidal anti-early pregnancy agents represents a significant paradigm shift in reproductive health pharmacology, moving away from traditional steroid-based compounds toward more targeted heterocyclic structures. Patent CN100434429C introduces a robust and environmentally superior methodology for the preparation of 2-arylimidazo[2,1-a]isoquinoline derivatives, which serve as critical scaffolds in this therapeutic class. This technical disclosure addresses the urgent industrial need for synthetic routes that balance high chemical efficiency with stringent environmental safety standards, particularly by eliminating the generation of toxic nitrogen oxide byproducts common in earlier iterations of this chemistry. For R&D directors and process chemists, understanding the nuances of this four-step sequence—from Friedel-Crafts acylation to final cyclization—is essential for optimizing yield and purity profiles in complex heterocyclic synthesis.
![General chemical structure of 2-arylimidazo[2,1-a]isoquinoline derivatives showing variable R groups](/insights/img/2-arylimidazo-isoquinoline-synthesis-pharma-supplier-20260307014525-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those disclosed in Chinese patent CN1872855, relied heavily on the use of potassium nitrite, acetic anhydride, and dimethyl sulfoxide (DMSO) to construct the necessary molecular framework. While chemically feasible on a small laboratory scale, these conventional approaches suffer from severe drawbacks when translated to industrial manufacturing environments. The most critical limitation is the inevitable generation of nitrogen oxide gases during the reaction process, which poses significant occupational health hazards and necessitates expensive, complex scrubbing systems to meet modern environmental regulations. Furthermore, the use of DMSO complicates downstream processing due to its high boiling point and difficulty in removal, often leading to solvent retention issues in the final active pharmaceutical ingredient (API) intermediate. These factors collectively result in inflated operational expenditures and extended production cycles, rendering older methods economically unviable for competitive generic drug manufacturing.
The Novel Approach
The innovative strategy outlined in patent CN100434429C circumvents these historical bottlenecks by employing a streamlined pathway that utilizes substituted benzene and isoquinoline as primary feedstocks. By shifting to a bromination-condensation mechanism, the process entirely eliminates the need for nitrosating agents, thereby eradicating the source of toxic NOx emissions at the molecular level. The reaction sequence leverages widely available commodity chemicals like acetyl chloride and liquid bromine, reacting them under controlled conditions to form alpha-brominated ketones which then undergo cyclization with 1-aminoisoquinoline. This approach not only simplifies the purification workflow through standard crystallization techniques but also significantly reduces the E-factor (environmental factor) of the synthesis. The resulting process is inherently safer, cleaner, and more aligned with Green Chemistry principles, offering a distinct competitive advantage for manufacturers seeking to minimize their environmental footprint while maximizing throughput.
Mechanistic Insights into the Cyclization and Amination Strategy
The core of this synthetic innovation lies in the efficient construction of the imidazo[2,1-a]isoquinoline ring system, achieved through a nucleophilic substitution followed by intramolecular cyclization. The process begins with the generation of 1-aminoisoquinoline via the reaction of isoquinoline with alkali metal amides, such as sodium amide, in a high-boiling xylene solvent. This Chichibabin-type amination is critical, as it installs the necessary nucleophilic nitrogen atom at the C1 position of the isoquinoline ring, activating it for subsequent attack. The reaction conditions, typically maintained between 100°C and 135°C, ensure complete conversion while minimizing thermal degradation of the sensitive heterocyclic core. Following isolation, the 1-aminoisoquinoline acts as a bidentate nucleophile, attacking the electrophilic alpha-carbon of the brominated acetophenone derivative. This displacement of the bromide ion initiates the ring closure, forming the fused imidazole ring with high regioselectivity.
Impurity control is meticulously managed through the physical properties of the intermediate hydrobromate salt. Upon completion of the cyclization in chloroform, the product precipitates as a stable hydrobromate salt, which serves as a self-purifying step. This salt formation effectively traps the basic product while leaving neutral organic impurities in the mother liquor. Subsequent recrystallization from methanol and ether precipitation further refines the solid state, removing trace halogenated byproducts and unreacted starting materials. Finally, the free base is liberated through a mild aqueous workup using strong ammonia water, ensuring that the final 2-arylimidazo[2,1-a]isoquinoline product meets rigorous purity specifications without the need for chromatographic purification. This reliance on crystallization-driven purification is a hallmark of scalable process chemistry, ensuring consistent quality across large production batches.
How to Synthesize 2-Arylimidazo[2,1-a]isoquinoline Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for producing high-quality intermediates suitable for contraceptive drug development. The procedure is divided into four distinct operational units: acylation, bromination, amination, and cyclization. Each step has been optimized to maximize yield while maintaining safety, utilizing standard glass-lined or stainless steel reactors common in fine chemical facilities. The following guide summarizes the critical operational parameters required to replicate this success, emphasizing temperature control and stoichiometric precision to prevent side reactions.
- Perform Friedel-Crafts acylation of substituted benzene with acetyl chloride and aluminum chloride to form substituted acetophenone.
- Brominate the substituted acetophenone using liquid bromine in methanol to generate alpha-brominated ketone.
- React isoquinoline with sodium amide in xylene at elevated temperatures to synthesize 1-aminoisoquinoline.
- Condense the alpha-brominated ketone with 1-aminoisoquinoline in chloroform, followed by hydrobromate salt formation and neutralization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers tangible economic benefits driven by simplified logistics and reduced regulatory overhead. The elimination of hazardous gas generation fundamentally alters the cost structure of the manufacturing process, removing the need for specialized abatement equipment and lowering insurance premiums associated with toxic chemical handling. Furthermore, the reliance on commodity feedstocks such as substituted benzenes, acetyl chloride, and isoquinoline ensures a stable and resilient supply chain, insulating production schedules from the volatility often seen with exotic or highly regulated reagents. This stability allows for more accurate forecasting and long-term contracting, essential for maintaining continuity in the pharmaceutical supply network.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the removal of expensive and difficult-to-handle reagents like DMSO and potassium nitrite. By avoiding the generation of nitrogen oxides, manufacturers save substantially on waste treatment costs and environmental compliance fees, which can be a major hidden expense in traditional heterocyclic synthesis. Additionally, the high yields observed in the bromination and cyclization steps reduce the overall consumption of raw materials per kilogram of finished product, directly improving the gross margin. The ability to isolate the product as a salt intermediate also minimizes product loss during purification, ensuring that the theoretical yield is closely approached in commercial practice.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis are bulk chemicals produced by a wide global network of suppliers, reducing the risk of single-source dependency. Isoquinoline and various acetophenones are established commodities with mature supply chains, ensuring that production can be scaled up rapidly without facing raw material bottlenecks. The use of common solvents like xylene, methanol, and chloroform further simplifies procurement, as these can be sourced locally in most major chemical manufacturing hubs. This ubiquity of inputs translates to shorter lead times and greater flexibility in responding to sudden increases in market demand for the final API.
- Scalability and Environmental Compliance: From an operational perspective, the process is designed for seamless scale-up, utilizing unit operations that are easily transferred from pilot plants to multi-ton reactors. The absence of high-pressure hydrogenation or cryogenic conditions means that existing infrastructure can often be repurposed for this synthesis with minimal capital investment. Moreover, the cleaner reaction profile aligns perfectly with increasingly stringent global environmental standards, future-proofing the manufacturing site against tightening regulations on volatile organic compounds (VOCs) and toxic emissions. This sustainability advantage is increasingly becoming a key differentiator when qualifying suppliers for major multinational pharmaceutical contracts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for decision-making. Understanding these details is crucial for assessing the feasibility of integrating this intermediate into your current product portfolio.
Q: What are the primary environmental advantages of this synthesis route?
A: Unlike previous methods utilizing potassium nitrite and acetic anhydride which generate hazardous nitrogen oxide gases, this patented process avoids toxic gas emissions, significantly reducing environmental compliance costs and waste treatment complexity.
Q: What is the expected purity profile for the final API intermediate?
A: The process utilizes multiple recrystallization steps, specifically using ethanol or ether precipitation, to achieve high-purity specifications suitable for pharmaceutical grade applications, effectively removing halogenated impurities and unreacted starting materials.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the methodology relies on standard unit operations such as filtration, vacuum distillation, and reflux, utilizing common industrial solvents like xylene and chloroform, making it highly adaptable for metric-ton scale manufacturing without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Arylimidazo[2,1-a]isoquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of novel contraceptive agents depends on the availability of high-quality, consistently manufactured intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2-arylimidazo[2,1-a]isoquinoline meets the exacting standards required for pharmaceutical applications. Our commitment to process excellence means we can deliver the cost and environmental advantages of this patented route at an industrial scale.
We invite you to collaborate with us to leverage this advanced technology for your next project. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and accelerate your time to market.
