Advanced Synthesis of Quercetin Dimer Derivatives for Commercial Pharmaceutical Applications
Advanced Synthesis of Quercetin Dimer Derivatives for Commercial Pharmaceutical Applications
The pharmaceutical industry is constantly seeking novel modifications of natural products to overcome inherent limitations such as poor solubility and low bioavailability. Patent CN108047271B introduces a groundbreaking approach to synthesizing quercetin dimer derivatives, effectively addressing these challenges through a sophisticated molecular design. This technology leverages the potent biological activities of quercetin, a well-known flavonoid, and amplifies them by creating dimeric structures linked via stable triazole and phosphoramidate bridges. The resulting compounds exhibit superior anticancer, antioxidant, and anti-inflammatory properties compared to the parent molecule. For R&D teams and procurement specialists alike, this patent represents a significant opportunity to access high-value pharmaceutical intermediates with enhanced therapeutic profiles. The strategic modification of the quercetin scaffold not only improves its physicochemical properties but also opens new avenues for drug development in oncology and cardiovascular health.
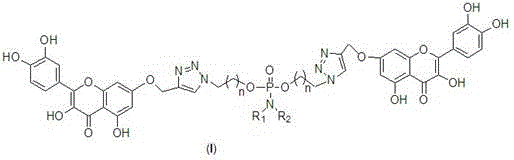
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to utilizing quercetin in therapeutic applications have been severely hampered by its intrinsic physicochemical drawbacks. Native quercetin is practically insoluble in water, which drastically limits its absorption and distribution within the human body, leading to poor oral bioavailability. Furthermore, simple monomeric forms often suffer from rapid metabolic clearance and limited potency against aggressive tumor lines. Conventional synthesis methods for flavonoid derivatives frequently rely on harsh acidic or basic conditions that can degrade the sensitive polyphenolic structure, resulting in complex impurity profiles that are difficult to purify. Additionally, many existing modification strategies fail to introduce functional groups that can actively participate in biological targeting, rendering the derivatives less effective than desired. The lack of a robust, scalable linkage strategy has historically prevented the commercial viability of quercetin-based dimers, leaving a gap in the market for high-performance flavonoid therapeutics.
The Novel Approach
The methodology disclosed in CN108047271B offers a transformative solution by employing a modular "click chemistry" strategy combined with phosphoramidate linkage. This novel approach constructs a symmetric dimer where two quercetin units are tethered by a central phosphorus-containing bridge, flanked by 1,2,3-triazole rings. This specific architecture is designed to enhance water solubility through the introduction of polar phosphate and hydroxyl groups while maintaining the structural integrity of the active flavonoid cores. The use of the triazole ring, formed via copper-catalyzed azide-alkyne cycloaddition (CuAAC), provides exceptional metabolic stability, ensuring the drug candidate remains intact long enough to reach its target. By systematically varying the substituents on the phosphorus atom (R1 and R2), chemists can fine-tune the lipophilicity and electronic properties of the molecule, optimizing it for specific biological environments. This level of structural control was previously unattainable with conventional esterification or etherification techniques.
Mechanistic Insights into Cu-Catalyzed Azide-Alkyne Cycloaddition
The core of this synthesis lies in the efficient formation of the 1,4-disubstituted 1,2,3-triazole ring, a reaction renowned for its high fidelity and atom economy. The mechanism initiates with the generation of a copper(I) acetylide species from the terminal alkyne of the 7-propargyloxyquercetin intermediate. In the presence of sodium ascorbate, copper(II) sulfate is reduced in situ to the active copper(I) catalyst, which coordinates with the organic azide component. This coordination lowers the activation energy for the cycloaddition, facilitating a concerted [3+2] cyclization that is strictly regioselective. Unlike thermal Huisgen cycloadditions which yield mixtures of 1,4- and 1,5-isomers, this catalytic system exclusively produces the 1,4-isomer, simplifying downstream purification and ensuring batch-to-batch consistency. The triazole ring thus formed acts as a rigid, non-hydrolyzable spacer that mimics the peptide bond, making it an ideal bioisostere for pharmaceutical applications.
Following the click reaction, the phosphoramidate center plays a critical role in the molecule's biological activity and stability. The phosphoryl dichloride intermediate reacts with the azido-alcohol to form a P-N or P-O bond, depending on the specific reagents used (e.g., nitrogen mustard or amino acid derivatives). This phosphorus center can act as a prodrug moiety, potentially undergoing enzymatic hydrolysis in vivo to release the active quercetin monomers or the intact dimer at the tumor site. The careful selection of R1 and R2 groups allows for the modulation of the molecule's charge and steric bulk, influencing its ability to penetrate cell membranes. Impurity control is inherently managed by the high selectivity of the click reaction and the crystallization properties of the intermediates, which can be purified via standard column chromatography or recrystallization, ensuring a high-purity final product suitable for clinical evaluation.
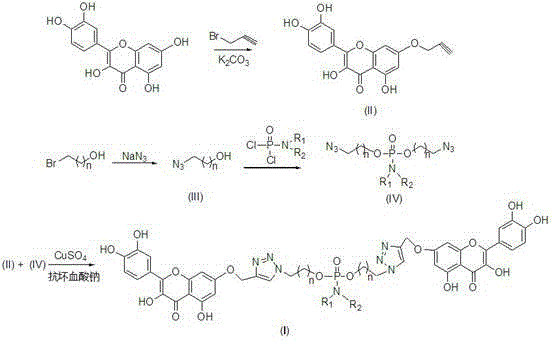
How to Synthesize Quercetin Dimer Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step roadmap for producing these high-value derivatives with excellent yields. The process begins with the selective alkylation of quercetin at the 7-position using propargyl bromide, followed by the preparation of the azide linker. These two key fragments are then united in the final coupling step. The operational simplicity of this route, utilizing common laboratory reagents and moderate temperatures, makes it highly attractive for process chemists looking to transfer technology from the bench to the pilot plant. Detailed standardized synthesis steps, including precise stoichiometry, solvent volumes, and workup procedures, are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Alkylate quercetin with bromopropyne in the presence of potassium carbonate to form 7-propargyloxyquercetin.
- React bromohydrin with sodium azide to generate the azide alcohol intermediate, followed by phosphorylation with phosphoryl dichloride.
- Perform the final coupling using CuSO4 and sodium ascorbate to link the alkyne and azide components via a triazole ring.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the manufacturing process described in this patent offers substantial advantages over traditional flavonoid modification routes. The reliance on readily available starting materials, such as bulk quercetin and simple bromo-alcohols, ensures a stable and resilient supply base that is not subject to the volatility of exotic reagent markets. The elimination of precious metal catalysts like palladium or platinum in favor of inexpensive copper sulfate significantly reduces the raw material cost profile. Furthermore, the aqueous workup procedures minimize the consumption of organic solvents, aligning with modern green chemistry initiatives and reducing waste disposal costs. These factors collectively contribute to a more sustainable and economically viable manufacturing model, allowing for competitive pricing in the global pharmaceutical intermediates market.
- Cost Reduction in Manufacturing: The substitution of expensive transition metal catalysts with a copper-sulfate/sodium-ascorbate system drastically lowers the cost of goods sold (COGS). Since copper is abundant and cheap, and the ligand (sodium ascorbate) is a commodity chemical, the catalytic cycle is extremely cost-efficient. Additionally, the high regioselectivity of the reaction minimizes the formation of isomeric byproducts, which reduces the burden on purification processes and increases the overall yield of the desired active pharmaceutical ingredient (API) intermediate. This efficiency translates directly into significant cost savings for large-scale production runs.
- Enhanced Supply Chain Reliability: The synthetic route relies on robust, well-understood chemical transformations that are less prone to failure compared to sensitive organometallic couplings. The intermediates, such as the azido-alcohol and propargyloxy-quercetin, are stable and can be stockpiled if necessary, providing flexibility in production scheduling. The use of common solvents like DMF, dichloromethane, and tert-butanol ensures that solvent supply chains remain uninterrupted even during global shortages. This reliability is crucial for maintaining continuous manufacturing operations and meeting strict delivery deadlines for downstream drug manufacturers.
- Scalability and Environmental Compliance: The process operates at moderate temperatures (50-60°C) and does not require high-pressure equipment or cryogenic conditions, making it easily scalable from kilogram to tonnage quantities. The workup involves simple phase separations and washes with brine or dilute acid, generating waste streams that are easier to treat and dispose of in compliance with environmental regulations. The absence of toxic heavy metals in the final product simplifies the regulatory filing process, as residual metal limits are easier to meet, thereby accelerating the time to market for new drug candidates based on this scaffold.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these quercetin dimer derivatives. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this technology into your existing R&D pipeline or supply chain network.
Q: What is the primary advantage of the quercetin dimer structure over monomeric quercetin?
A: The dimerization significantly improves water solubility and bioavailability while enhancing anticancer and antioxidant potency through a bivalent mechanism.
Q: Is the copper catalyst used in this synthesis difficult to remove?
A: No, the process utilizes a simple aqueous workup with HCl and standard extraction, allowing for efficient removal of copper residues without complex scavenging resins.
Q: Can this synthesis be scaled for industrial production?
A: Yes, the reaction conditions utilize common solvents like DMF and tert-Butanol/water mixtures and operate at moderate temperatures (50-60°C), making it highly amenable to scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quercetin Dimer Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of the quercetin dimer platform in developing next-generation anticancer and anti-inflammatory therapies. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory discovery to industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate we deliver adheres to the highest international standards. We understand the critical nature of timeline and quality in the pharmaceutical sector and are committed to being a dependable extension of your team.
We invite you to collaborate with us to unlock the full commercial potential of this innovative chemistry. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary analogs. Let us help you accelerate your drug development program with our superior manufacturing capabilities and deep technical expertise.
