Advanced Preparation Method for Pranlukast Intermediates via Palladium Catalysis
Introduction to Patent CN108947984B and Pranlukast Manufacturing
The pharmaceutical landscape for asthma management continues to evolve, with Pranlukast standing out as a potent leukotriene receptor antagonist. The preparation method detailed in patent CN108947984B represents a significant technological leap in the synthesis of this critical active pharmaceutical ingredient (API). Unlike traditional routes that rely on scarce and expensive precursors, this innovation leverages a robust bromo-intermediate strategy to achieve superior yields and purity. For R&D directors and procurement managers alike, understanding the nuances of this patent is essential for securing a reliable pranlukast intermediate supplier. The method addresses long-standing challenges in the supply chain by simplifying the synthetic pathway and utilizing commercially abundant raw materials. This report provides a deep technical analysis of the process, highlighting its potential for cost reduction in pharmaceutical intermediates manufacturing and ensuring a stable supply of high-purity pranlukast for global markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Pranlukast has been hindered by reliance on complex and costly starting materials. The most common conventional method utilizes 3-((4-(4-phenylbutoxy) ammonium benzoate)-2-hydroxyacetophenone (PBHA) as a key raw material. As illustrated in the reaction scheme below, this route necessitates an ester ketone condensation followed by acidic ring closure. The fundamental flaw lies in the sourcing of PBHA, which is not only expensive but also difficult to procure in bulk quantities, creating a bottleneck for large-scale production. Furthermore, this traditional pathway requires the use of both strong bases and strong acids, which introduces significant safety hazards and complicates waste treatment protocols. The harsh reaction conditions often lead to the formation of difficult-to-remove impurities, thereby compromising the final purity of the API and increasing downstream purification costs. These factors collectively result in a fragile supply chain vulnerable to raw material price fluctuations and regulatory scrutiny regarding environmental compliance.
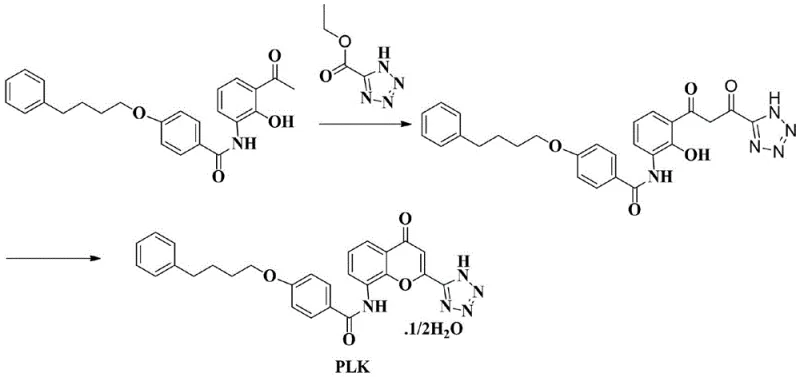
The Novel Approach
In stark contrast, the novel approach disclosed in patent CN108947984B circumvents these bottlenecks by initiating the synthesis with 4-bromobenzoic acid, a commodity chemical with excellent availability and pricing stability. This strategic shift allows for a more linear and controllable synthesis trajectory. The new method employs a sequential amidation, cyclization, and finally a palladium-catalyzed etherification to construct the target molecule. By avoiding the pre-formation of the complex PBHA ether linkage, the process significantly reduces the number of purification steps required early in the synthesis. The reaction conditions are notably milder, utilizing organic bases like pyridine or triethylamine rather than corrosive inorganic acids or bases. This transition not only enhances operator safety but also drastically simplifies the workup procedures, leading to higher overall recovery rates. The adoption of this route signifies a move towards greener chemistry principles, aligning with modern regulatory expectations for sustainable pharmaceutical manufacturing.
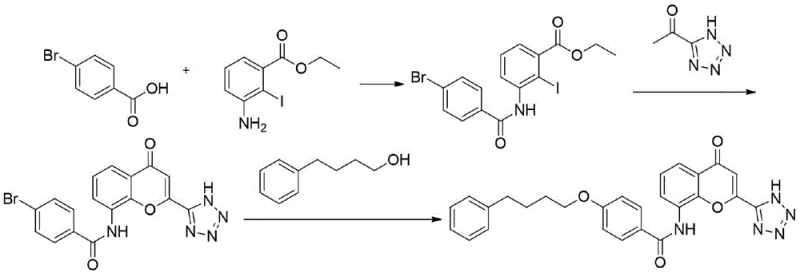
Mechanistic Insights into Pd-Catalyzed Etherification and Cyclization
The core of this innovative synthesis lies in the precise execution of the cyclization and subsequent etherification steps. In Step C, the formation of the 4H-benzopyrone core is achieved through the reaction of the iodo-benzamide intermediate with 1-(1H-tetrazole-5-yl)ethanone. The use of lithium bis(trimethylsilyl)amide (LiHMDS) or potassium carbonate as the base is critical here; these reagents facilitate the deprotonation and subsequent nucleophilic attack required to close the pyrone ring without degrading the sensitive tetrazole moiety. Maintaining the reaction temperature between 70°C and 110°C in solvents like toluene or DMF ensures optimal kinetic energy for the cyclization while preventing thermal decomposition. This careful balance is what allows the process to achieve yields upwards of 86% with purities exceeding 97%, a metric that is crucial for minimizing impurity carryover into the final API.
Following the core construction, Step D employs a sophisticated palladium-catalyzed cross-coupling reaction to install the 4-phenylbutoxy side chain. This transformation replaces the traditional nucleophilic substitution which often requires harsh conditions. By using palladium acetate or cuprous iodide as a catalyst in the presence of 4-phenylbutanol and a base like cesium carbonate, the reaction proceeds via a coordinated oxidative addition and reductive elimination cycle. This mechanism is highly selective for the aryl bromide bond, leaving other functional groups untouched. The result is a clean conversion to the final Pranlukast structure with exceptional regioselectivity. The ability to perform this coupling at moderate temperatures (80°C to 110°C) demonstrates the robustness of the catalytic system, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates where reproducibility is paramount.
How to Synthesize Pranlukast Efficiently
Implementing this synthesis route requires strict adherence to the stoichiometric ratios and thermal profiles outlined in the patent data. The process begins with the activation of 4-bromobenzoic acid using thionyl chloride, followed by immediate amidation to prevent hydrolysis. The subsequent cyclization demands anhydrous conditions to protect the tetrazole ring integrity. Finally, the palladium-catalyzed step requires careful degassing of solvents to maintain catalyst activity. For process chemists looking to replicate this success, the detailed standardized synthesis steps provided in the patent serve as a foundational guide. The following section outlines the specific operational parameters required to achieve the reported high yields and purity specifications.
- React 4-bromobenzoic acid with thionyl chloride to form 4-bromobenzoyl chloride, then couple with ethyl 3-amino-2-iodobenzoate.
- Perform cyclization using 1-(1H-tetrazole-5-yl)ethanone and a strong base like lithium bis(trimethylsilyl)amide to form the benzopyrone core.
- Execute a palladium-catalyzed etherification with 4-phenylbutanol and potassium carbonate to finalize the Pranlukast structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the shift to this novel synthesis route offers profound benefits for supply chain resilience and cost management. The primary driver of value is the substitution of the exotic PBHA starting material with 4-bromobenzoic acid and 4-phenylbutanol, both of which are produced on a massive industrial scale globally. This change effectively decouples the production of Pranlukast from the volatility of niche intermediate markets. Furthermore, the elimination of strong mineral acids and bases reduces the burden on waste treatment facilities, lowering the environmental compliance costs associated with neutralization and disposal. The high purity achieved directly from the crystallization steps (often exceeding 99%) means that resource-intensive recrystallization cycles can be minimized or eliminated, further driving down processing time and solvent consumption.
- Cost Reduction in Manufacturing: The economic impact of this route is driven by the drastic simplification of the raw material portfolio. By utilizing commodity chemicals instead of custom-synthesized precursors, the direct material cost is significantly reduced. Additionally, the high yield of the amidation step (reported up to 95%) ensures that very little valuable intermediate is lost to side reactions. The avoidance of expensive heavy metal scavengers, which are often needed when using less selective catalysts, further contributes to substantial cost savings. The overall process efficiency translates to a lower cost of goods sold (COGS), providing a competitive margin advantage in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Supply continuity is a critical concern for procurement managers, and this method addresses it by diversifying the source of key inputs. Since 4-bromobenzoic acid and 4-phenylbutanol are available from multiple global suppliers, the risk of single-source dependency is mitigated. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in utility supply, such as cooling water temperature fluctuations. This stability ensures consistent batch-to-batch quality and reliable delivery schedules, which is essential for maintaining uninterrupted API production lines for asthma medications.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing solvents like ethyl acetate and toluene which are easily recovered and recycled in large-scale distillation units. The absence of hazardous reagents simplifies the safety profile of the plant, reducing the need for specialized containment equipment. From an environmental standpoint, the atom economy of the palladium-catalyzed step is superior to traditional substitution methods, generating less salt waste. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals, making the manufacturing site more resilient to future environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of patent CN108947984B. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these details is vital for technical teams evaluating the feasibility of technology transfer. The answers reflect the specific advantages of the bromo-intermediate route over legacy methods.
Q: What are the primary advantages of the bromo-intermediate route over the traditional PBHA method?
A: The bromo-intermediate route utilizes readily available starting materials like 4-bromobenzoic acid, avoiding the expensive and hard-to-source PBHA. It also eliminates the need for harsh strong acid and strong base conditions required in the traditional cyclization, leading to safer operations and higher purity profiles exceeding 99%.
Q: How does the new method control impurities during the cyclization step?
A: By utilizing specific molar ratios of lithium bis(trimethylsilyl)amide or potassium carbonate in solvents like toluene or DMF at controlled temperatures between 70°C and 110°C, the process minimizes side reactions. This precise control ensures the tetrazole ring remains intact while forming the benzopyrone core efficiently.
Q: Is this synthesis route scalable for commercial production?
A: Yes, the process is designed for scalability. The use of common solvents like ethyl acetate, toluene, and DMF, combined with robust catalysts like palladium acetate, allows for straightforward scale-up from laboratory grams to multi-ton commercial batches without significant yield loss.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pranlukast Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the global pharmaceutical supply chain. Our technical team has extensively analyzed the pathways described in CN108947984B and possesses the expertise to optimize this chemistry for industrial application. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to reactor is seamless. Our facility is equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Pranlukast intermediate meets the highest international standards for safety and efficacy.
We invite procurement leaders and R&D directors to collaborate with us to leverage this advanced technology. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, high-quality supply of Pranlukast intermediates that drives your business forward.
