Scalable Synthesis of High-Purity Marchantin S Derivatives for Anti-Inflammatory Drug Development
The pharmaceutical industry is constantly seeking novel scaffolds that offer dual therapeutic potential, particularly in the realms of oncology and chronic inflammation management. Patent CN113549046B introduces a groundbreaking class of bisbibenzyl compounds centered around the core structure of Marchantin S, a cyclic bisbibenzyl originally isolated from the liverwort Reboulia hemisphaerica. This intellectual property details not only the isolation of this unique natural product but, more critically for industrial application, provides a robust total synthesis pathway and a diverse library of derivatives designed to optimize pharmacological performance. The strategic value of this technology lies in its ability to transform a scarce natural extract into a commercially viable pharmaceutical intermediate through precise organic synthesis. By leveraging this patented methodology, manufacturers can access high-purity compounds that demonstrate exceptional inhibition of pro-inflammatory cytokines such as IL-1β and IL-6, alongside potent cytotoxicity against various cancer cell lines including HepG2 and A549.
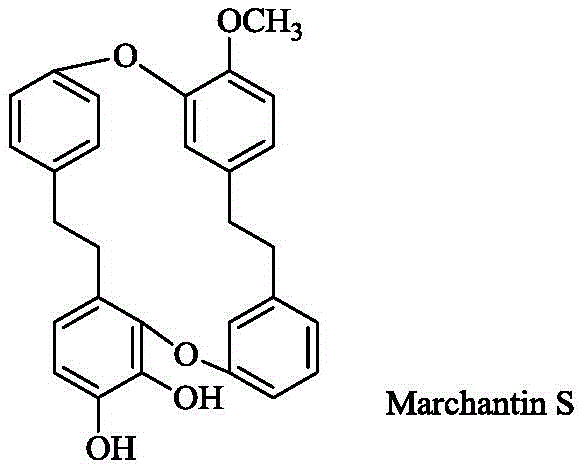
For research and development directors evaluating new pipeline candidates, the structural versatility offered by this patent is paramount. The core Marchantin S scaffold serves as a rigid, lipophilic framework that can be systematically modified at multiple positions to fine-tune bioavailability and target affinity. The patent outlines specific substitution patterns where phenolic hydroxyl groups are converted into ethers, esters, or aminomethyl derivatives, creating a structure-activity relationship (SAR) map that guides rational drug design. This level of chemical control is essential for developing next-generation anti-inflammatory agents that avoid the side effects associated with traditional NSAIDs. Furthermore, the synthesis route is designed to be scalable, utilizing standard organic transformations that are well-understood in process chemistry, thereby reducing the technical risk associated with scaling up complex natural product analogs for clinical trials and eventual commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the supply of bioactive bisbibenzyls has been severely constrained by reliance on natural extraction from bryophytes, a process fraught with inconsistency and environmental variability. Extracting Marchantin S directly from plant sources yields minute quantities, making it impossible to secure the kilogram-to-ton scales required for preclinical and clinical development. Moreover, natural extracts often contain complex mixtures of structurally similar congeners, necessitating expensive and time-consuming purification steps that drastically increase the cost of goods sold (COGS). The seasonal availability of the source material further exacerbates supply chain vulnerabilities, creating bottlenecks that can delay critical drug discovery programs. From a regulatory perspective, relying on a botanical source introduces challenges in defining a consistent impurity profile, which is a stringent requirement for FDA and EMA approval of new molecular entities. These factors collectively render natural extraction an unsustainable strategy for the commercial manufacturing of high-value pharmaceutical intermediates.
The Novel Approach
The synthetic methodology disclosed in CN113549046B represents a paradigm shift by establishing a fully synthetic route that bypasses the limitations of natural sourcing. This approach utilizes readily available aromatic aldehydes and phenols as starting materials, employing key reactions such as Ullmann coupling, Wittig olefination, and catalytic hydrogenation to construct the complex macrocyclic architecture. By controlling every step of the synthesis, manufacturers can ensure batch-to-batch reproducibility and achieve purity levels that exceed those possible with extraction. The ability to introduce specific functional groups at defined positions allows for the creation of a focused library of derivatives, enabling rapid optimization of biological activity without the need for semi-synthetic modification of scarce natural products. This synthetic flexibility not only secures the supply chain but also opens avenues for patent protection on novel derivatives, providing a competitive moat for pharmaceutical companies investing in this technology.
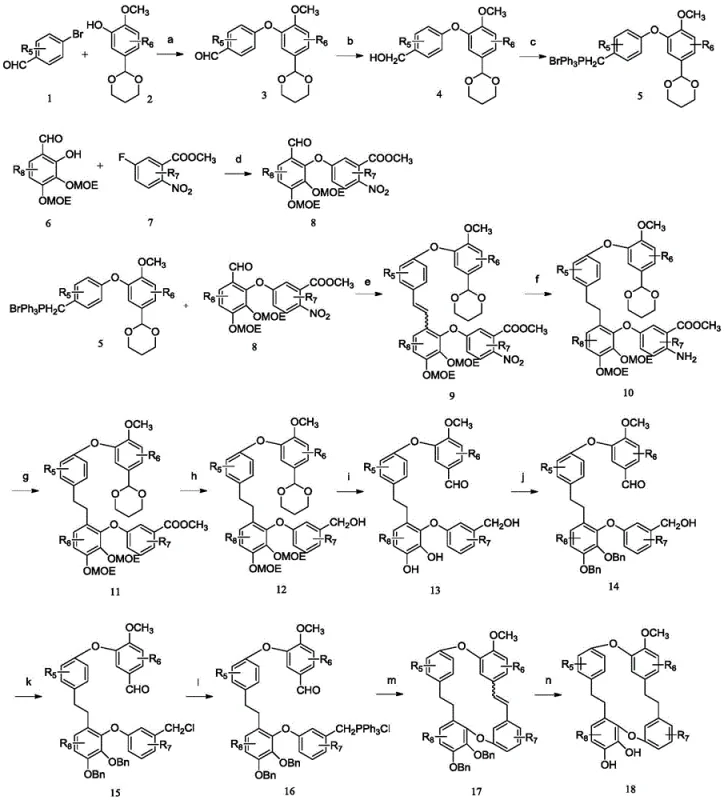
Mechanistic Insights into Bisbibenzyl Macrocyclization and Functionalization
The core of the synthetic strategy involves a sophisticated sequence of bond-forming reactions that construct the 14-membered macrocyclic ring characteristic of bisbibenzyls. A critical step in this process is the formation of the diaryl ether linkage, typically achieved through copper-catalyzed coupling reactions which require precise temperature control and ligand selection to maximize yield and minimize homocoupling byproducts. Following the assembly of the linear precursor, the macrocyclization is often facilitated by intramolecular reactions that close the ring, a step that demands high dilution conditions to favor cyclization over polymerization. The patent details the use of protecting group strategies, such as methoxymethyl (MOM) or benzyl ethers, to mask reactive phenolic hydroxyls during these harsh conditions, ensuring that the final deprotection steps reveal the active pharmacophore without damaging the sensitive macrocyclic core. Understanding these mechanistic nuances is vital for process chemists aiming to optimize reaction parameters for large-scale production.
Impurity control is another cornerstone of this synthetic approach, particularly given the complexity of the target molecules. The use of catalytic hydrogenation with palladium on carbon (Pd/C) for the reduction of nitro groups and olefins is highlighted as a clean and efficient method that generates minimal waste compared to stoichiometric reducing agents. Furthermore, the final derivatization steps, such as the demethylation using boron tribromide (BBr3) or the introduction of amine side chains via Mannich reactions, are designed to be highly selective. This selectivity reduces the formation of regioisomers, simplifying the downstream purification process and improving the overall mass balance of the synthesis. For R&D teams, this means a clearer path to obtaining reference standards and drug substance with well-defined impurity profiles, which is crucial for toxicological assessments and regulatory filings. The robustness of these chemical transformations ensures that the process can withstand the rigors of commercial manufacturing while maintaining high product quality.
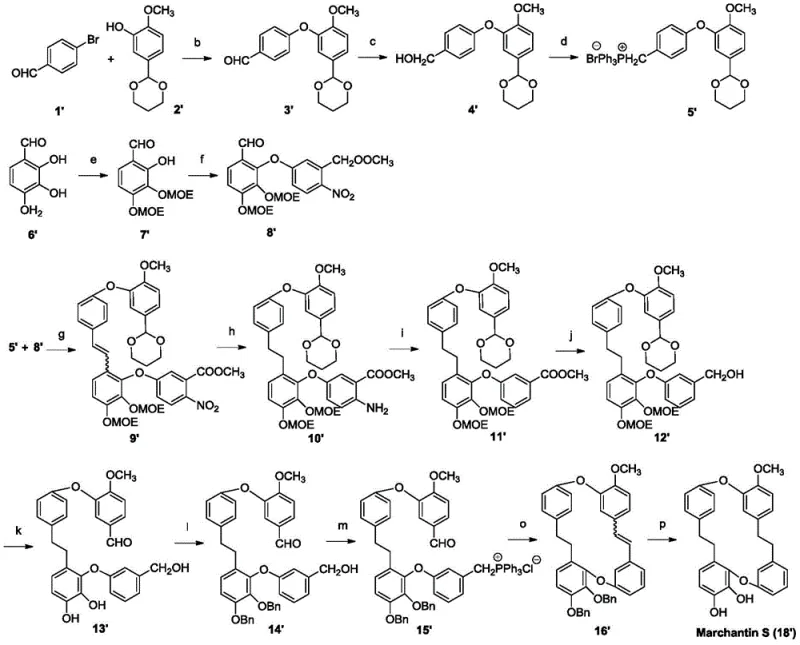
How to Synthesize Marchantin S Derivatives Efficiently
The synthesis of these high-value intermediates requires a meticulous adherence to the reaction conditions outlined in the patent to ensure optimal yields and purity. The process begins with the preparation of key building blocks through etherification and reduction steps, followed by the assembly of the macrocyclic skeleton via sequential coupling and cyclization reactions. Each stage of the synthesis is optimized to minimize side reactions, with specific attention paid to solvent selection and temperature profiles to drive the equilibrium towards the desired product. The final stages involve the strategic installation of functional groups that confer biological activity, such as the introduction of nitrogen-containing heterocycles which have been shown to enhance solubility and target binding. For laboratories looking to replicate or scale this chemistry, it is essential to utilize high-purity reagents and maintain strict anhydrous conditions during sensitive steps like the Wittig reaction and Lewis acid-mediated deprotections.
- Preparation of key intermediates via Ullmann coupling and reduction steps to form the biphenyl ether backbone.
- Cyclization and macrocyclization reactions utilizing Wittig olefination and hydrogenation to close the bisbibenzyl ring system.
- Final derivatization through selective demethylation, esterification, or Mannich reactions to introduce pharmacophores.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from natural extraction to total synthesis offers profound strategic advantages that extend beyond mere cost savings. The primary benefit is the stabilization of the supply chain; by relying on commodity chemicals rather than seasonal plant harvests, manufacturers can guarantee continuous production schedules and eliminate the risk of raw material shortages. This reliability is critical for maintaining the continuity of drug development pipelines, where delays in intermediate supply can cascade into significant financial losses and missed market windows. Furthermore, the synthetic route allows for the decoupling of production capacity from geographical constraints associated with botanical sourcing, enabling manufacturing to be located in regions with established chemical infrastructure and favorable regulatory environments. This flexibility enhances the resilience of the supply network against geopolitical disruptions and logistical challenges.
- Cost Reduction in Manufacturing: The implementation of a fully synthetic route eliminates the exorbitant costs associated with the collection, extraction, and purification of natural products from biomass. By utilizing scalable organic synthesis techniques, the cost per gram of the active intermediate is drastically reduced, making the final drug product more economically viable. The avoidance of expensive chromatographic separations required for complex natural mixtures further contributes to substantial cost savings, allowing for more competitive pricing in the global marketplace. Additionally, the ability to synthesize analogs directly means that resources are not wasted on isolating inactive congeners, focusing investment solely on the most promising therapeutic candidates.
- Enhanced Supply Chain Reliability: Synthetic manufacturing provides a predictable and controllable production timeline that is immune to the vagaries of agriculture and climate. Procurement teams can negotiate long-term contracts with confidence, knowing that production volumes can be ramped up quickly to meet demand without waiting for growing seasons. The use of standard chemical reactors and purification equipment means that production can be easily transferred between different contract manufacturing organizations (CMOs) if necessary, reducing single-source dependency. This redundancy is a key component of a robust supply chain strategy, ensuring that patient access to critical medications is never compromised by upstream supply issues.
- Scalability and Environmental Compliance: The synthetic pathways described are amenable to scale-up from laboratory to commercial tonnage using standard process engineering principles. The reactions utilize common solvents and catalysts that can be recovered and recycled, aligning with green chemistry initiatives and reducing the environmental footprint of manufacturing. By avoiding the land-use changes and biodiversity impacts associated with wild-harvesting medicinal plants, this synthetic approach supports corporate sustainability goals. Moreover, the well-defined waste streams from chemical synthesis are easier to treat and manage compared to the complex organic waste generated by botanical extraction, facilitating compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Marchantin S derivatives. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on the feasibility and potential of this technology. Understanding these aspects is crucial for stakeholders evaluating the integration of these intermediates into their existing drug discovery platforms or supply chains. The information covers aspects ranging from biological efficacy to process scalability, providing a holistic view of the value proposition.
Q: What are the primary biological activities of Marchantin S derivatives?
A: According to patent CN113549046B, these derivatives exhibit significant anti-inflammatory activity by inhibiting IL-1β and IL-6 transcription, as well as potent antitumor effects against HepG2, A549, and MCF-7 cell lines.
Q: How does the synthetic route improve supply chain stability?
A: The total synthesis route replaces unreliable natural extraction from mosses with a robust chemical process using commercially available starting materials, ensuring consistent quality and uninterrupted supply for large-scale manufacturing.
Q: What structural modifications enhance potency?
A: The patent indicates that introducing nitrogen-containing heterocycles such as piperazinyl or morpholinyl groups via Mannich reactions, or modifying phenolic hydroxyl groups, significantly enhances cytotoxicity and anti-inflammatory efficacy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Marchantin S Supplier
As the demand for novel anti-inflammatory and antitumor agents continues to rise, securing a reliable supply of high-quality intermediates is more critical than ever. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in complex organic synthesis to deliver Marchantin S derivatives that meet the rigorous standards of the global pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your needs from early-stage development through to full-scale commercialization. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of material we supply is consistent, safe, and ready for formulation.
We invite you to collaborate with us to unlock the full potential of these promising bisbibenzyl compounds. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project requirements, demonstrating how our optimized synthetic routes can reduce your overall development costs. We encourage you to contact us today to request specific COA data for our available derivatives and to discuss route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a trusted ally dedicated to accelerating your drug development timeline and ensuring the success of your therapeutic programs.
