Advanced Synthesis of (E)-Substituted Styrene Compounds for Pharmaceutical Applications
Introduction to Advanced Stilbene Synthesis
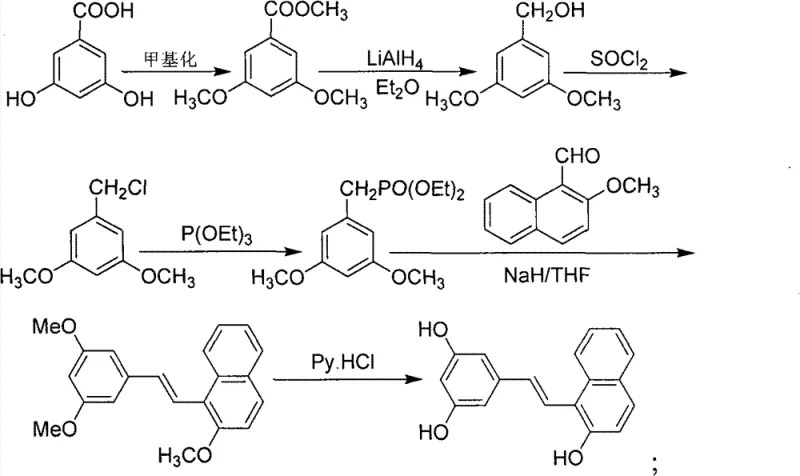
The pharmaceutical industry continuously seeks robust and scalable pathways for bioactive molecules, particularly those exhibiting potent antioxidant and antitumor properties. Patent CN101544591B introduces a sophisticated yet practical methodology for the preparation of (E)-substituted styrene compounds, a class of structures that includes renowned bioactive agents like trans-resveratrol and its analogs. This intellectual property outlines a versatile synthetic strategy that transforms readily available 3,5-dihydroxy benzoic acid into complex stilbene derivatives through a sequence of methylation, alkylation, reduction, and crucially, Wittig-Horner condensation. For R&D directors and process chemists, this patent represents a significant advancement in accessing high-purity intermediates with defined stereochemistry, addressing the critical need for reliable supply chains in drug development.
The technical breakthrough lies in the modularity of the approach, allowing for the systematic variation of substituents on the aromatic rings to tune biological activity. By employing a phosphonate-based olefination strategy, the process ensures high selectivity for the desired (E)-isomer, which is often the pharmacologically active form. This level of control is paramount for regulatory compliance and efficacy in final drug products. Furthermore, the reaction conditions described are notably mild, avoiding extreme pressures or temperatures that often complicate scale-up, thereby positioning this technology as a cornerstone for cost reduction in pharmaceutical intermediate manufacturing.
![Synthetic route for (E)-1-[3-(1-skatole)]-2-(3,5-dimethoxy benzene) ethene showing methylation, reduction, and Wittig-Horner steps](/insights/img/e-substituted-styrene-wittig-pharma-supplier-20260307064518-01.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to substituted styrenes often rely on Heck coupling or classic Wittig reactions that suffer from significant drawbacks in an industrial setting. Heck couplings, while powerful, typically require expensive palladium catalysts and rigorous exclusion of oxygen and moisture, leading to higher operational costs and potential heavy metal contamination issues that are difficult to purge to ppm levels. Similarly, classical Wittig reactions using unstabilized ylids can result in mixtures of (E) and (Z) isomers, necessitating energy-intensive chromatographic separations that drastically reduce overall yield and throughput. These inefficiencies create bottlenecks in the supply chain, increasing lead times and compromising the economic viability of producing complex API intermediates at a commercial scale.
The Novel Approach
In contrast, the methodology disclosed in CN101544591B leverages the Horner-Wadsworth-Emmons (HWE) modification of the Wittig reaction, utilizing dialkyl phosphonates which are more stable and easier to handle than phosphonium salts. This novel approach facilitates the formation of the carbon-carbon double bond with excellent (E)-selectivity under relatively mild basic conditions using sodium hydride in tetrahydrofuran. The synthetic pathway begins with the protection of phenolic hydroxyl groups via methylation, followed by reduction of the carboxylic acid moiety to a primary alcohol using lithium aluminum hydride. This alcohol is then converted to a chloride and subsequently to a phosphonate, creating a versatile building block that can react with various aldehydes, such as indole-3-carboxaldehyde or methoxynaphthaldehyde, to generate diverse stilbene scaffolds efficiently.
Mechanistic Insights into Wittig-Horner Condensation
The core of this synthetic innovation is the Wittig-Horner condensation mechanism, which proceeds through the formation of a stabilized carbanion from the benzyl phosphonate precursor. Upon treatment with a strong base like sodium hydride at low temperatures ranging from -20°C to 20°C, the acidic proton alpha to the phosphorus atom is abstracted, generating a nucleophilic species. This carbanion then attacks the carbonyl carbon of the aldehyde electrophile, forming a betaine intermediate that collapses into an oxaphosphetane four-membered ring. Unlike the irreversible nature of some classical Wittig reactions, the HWE reaction is often under thermodynamic control, favoring the elimination of the phosphate byproduct to yield the more stable trans-alkene geometry. This mechanistic feature is critical for ensuring high stereochemical purity without the need for downstream isomerization or separation steps.
Furthermore, the patent details a robust deprotection strategy for converting the methoxy-protected intermediates back to the biologically relevant phenolic hydroxyl groups. This is achieved by heating the methoxy-substituted stilbenes with pyridine hydrochloride at temperatures between 150°C and 230°C. This thermal demethylation cleaves the methyl ethers effectively, restoring the hydrogen-bonding capability of the phenol groups which is often essential for the compound's interaction with biological targets. The ability to perform this deprotection as a final step allows chemists to carry out the preceding carbon-carbon bond-forming reactions on protected substrates, thereby preventing side reactions at the phenolic sites and improving overall process robustness and yield.
How to Synthesize (E)-Substituted Styrene Efficiently
The synthesis of these high-value intermediates requires precise control over reaction parameters to maximize yield and purity. The process initiates with the methylation of 3,5-dihydroxybenzoic acid using dimethyl sulfate in the presence of anhydrous potassium carbonate and acetone, followed by reflux to ensure complete conversion to the dimethyl ester. Subsequent reduction with lithium aluminum hydride in anhydrous diethyl ether must be conducted under an inert nitrogen atmosphere to prevent oxidation, yielding the corresponding benzyl alcohol. This alcohol is then activated by conversion to a benzyl chloride using thionyl chloride, which serves as the precursor for the Arbuzov reaction with triethyl phosphite to generate the key phosphonate intermediate. For the detailed standardized operating procedures and specific stoichiometric ratios required for GMP-compliant production, please refer to the technical guide below.
- Methylation of 3,5-dihydroxy benzoic acid using dimethyl sulfate and potassium carbonate to form the dimethyl ester.
- Reduction of the ester to the corresponding benzyl alcohol using Lithium Aluminum Hydride (LiAlH4) in anhydrous ether.
- Conversion of the alcohol to benzyl chloride using thionyl chloride (SOCl2), followed by Arbuzov reaction with triethyl phosphite to form the phosphonate.
- Wittig-Horner condensation of the phosphonate with various aldehydes (e.g., indole-3-carboxaldehyde) using NaH in THF to form the stilbene backbone.
- Optional deprotection of methoxy groups using pyridine hydrochloride at elevated temperatures to yield the final polyhydroxy product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers substantial strategic benefits regarding cost stability and material availability. The reliance on commodity chemicals such as 3,5-dihydroxybenzoic acid, dimethyl sulfate, and triethyl phosphite ensures that the raw material supply chain is resilient against market volatility. Unlike processes dependent on scarce noble metals or exotic ligands, this methodology utilizes reagents that are globally sourced and available in bulk quantities, significantly mitigating the risk of production stoppages due to raw material shortages. This reliability is crucial for maintaining continuous manufacturing schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts, such as palladium or platinum, directly translates to lower raw material costs and simplified waste management protocols. By avoiding heavy metals, the downstream purification process becomes less complex, reducing the consumption of silica gel and solvents required for chromatography. This streamlined workflow results in substantial cost savings per kilogram of produced intermediate, enhancing the overall profit margin for large-scale commercial production runs.
- Enhanced Supply Chain Reliability: The synthetic steps described involve standard unit operations such as reflux, distillation, and crystallization, which are easily implemented in existing multipurpose chemical plants without the need for specialized reactor modifications. This compatibility with standard infrastructure allows for rapid technology transfer and flexible capacity allocation, ensuring that supply can be scaled up quickly to meet surges in demand. The robustness of the chemistry also implies fewer batch failures, leading to more predictable inventory levels and improved service levels for downstream customers.
- Scalability and Environmental Compliance: The process generates waste streams that are well-characterized and manageable within standard environmental treatment facilities. The absence of persistent organic pollutants associated with certain catalytic systems simplifies regulatory compliance and reduces the environmental footprint of the manufacturing site. Furthermore, the high atom economy of the condensation steps and the ability to recover and recycle solvents like tetrahydrofuran and ethyl acetate contribute to a more sustainable and economically efficient production model.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its implementation. The following questions address common inquiries regarding the scalability, purity, and specific reaction conditions outlined in the patent documentation. These insights are derived directly from the experimental data and procedural descriptions provided in the intellectual property, ensuring accuracy and relevance for technical decision-making.
Q: What are the key advantages of the Wittig-Horner method described in CN101544591B?
A: The method offers mild reaction conditions, simple operation, and high stereoselectivity for the (E)-isomer. It avoids the use of expensive transition metal catalysts often required in cross-coupling reactions, significantly simplifying the purification process and reducing heavy metal impurities in the final API intermediate.
Q: Can this synthesis route be scaled for commercial production?
A: Yes, the route utilizes common industrial reagents such as dimethyl sulfate, thionyl chloride, and triethyl phosphite. The reaction steps, including reflux and standard aqueous workups, are highly amenable to scale-up from laboratory grams to multi-ton commercial batches without requiring specialized high-pressure equipment.
Q: How is the stereochemistry controlled to ensure the (E)-configuration?
A: The Wittig-Horner reaction inherently favors the formation of the thermodynamically more stable (E)-alkene isomer, especially when using stabilized phosphonate carbanions and appropriate bases like sodium hydride. This eliminates the need for complex isomer separation steps typically associated with traditional Wittig reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (E)-Substituted Styrene Compound Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific reagents and conditions required for this Wittig-Horner based synthesis, maintaining stringent purity specifications through our rigorous QC labs. We understand that the quality of the intermediate directly impacts the efficacy and safety of the final drug product, which is why we adhere to the highest standards of process control and documentation throughout the entire production lifecycle.
We invite you to engage with our technical procurement team to discuss how this advanced synthetic route can be optimized for your specific application needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and innovation in your pharmaceutical development pipeline.
