Mastering Regioselective Lithiation of 1,3-Bis(trifluoromethyl)benzene for Commercial API Production
Mastering Regioselective Lithiation of 1,3-Bis(trifluoromethyl)benzene for Commercial API Production
The pharmaceutical and fine chemical industries constantly seek robust synthetic routes for fluorinated building blocks, particularly those serving as precursors for antimalarial and other bioactive agents. Patent CN1028994C presents a groundbreaking advancement in the lithiation of 1,3-bis(trifluoromethyl)benzene, addressing a long-standing challenge in regioselectivity that has plagued manufacturers for decades. By utilizing lithium salts of specific amines rather than traditional alkyllithium reagents, this technology enables the preferential formation of the valuable 2,4-isomer over the undesired 2,6-isomer with unprecedented efficiency. For R&D directors and procurement specialists, this innovation translates directly into higher purity profiles and streamlined downstream processing, essential for meeting the stringent quality standards of modern API manufacturing. The ability to control the substitution pattern on the electron-deficient aromatic ring opens new avenues for synthesizing complex intermediates without the burden of difficult chromatographic separations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of 1,3-bis(trifluoromethyl)benzene has relied heavily on n-butyllithium as the lithiating agent, a method documented in prior art such as Kodaira et al. and Coleman et al. These conventional approaches suffer from a critical deficiency: poor regiocontrol. When n-butyllithium is employed, the resulting lithiated species reacts with electrophiles to produce a statistical mixture of isomers, typically yielding a ratio of 2,4-isomer to 2,6-isomer ranging from roughly 1:1 to 3:2. This lack of selectivity creates a significant bottleneck in commercial production, as separating these structurally similar fluorinated isomers is notoriously difficult and costly. The presence of nearly equal amounts of the unwanted 2,6-isomer necessitates extensive purification efforts, often involving multiple recrystallizations or preparative chromatography, which drastically reduces overall yield and increases the cost of goods sold. Furthermore, the handling of large quantities of n-butyllithium at cryogenic temperatures poses inherent safety risks and operational complexities that supply chain managers strive to avoid in large-scale facilities.
The Novel Approach
The methodology disclosed in Patent CN1028994C fundamentally alters the reaction landscape by replacing small, aggressive alkyllithiums with sterically hindered amine lithium salts, such as lithium 2,2,6,6-tetramethylpiperidide (LiTMP). This strategic shift in reagent selection leverages steric bulk to direct the lithiation exclusively to the less hindered positions on the aromatic ring. Experimental data within the patent demonstrates that this approach can achieve a 2,4-isomer to 2,6-isomer ratio ranging from 4:1 to greater than 100:1, depending on the specific electrophile and conditions used. Such a dramatic improvement in selectivity means that the crude reaction mixture is already enriched with the desired product, often exceeding 90% purity before any purification steps are undertaken. This breakthrough not only simplifies the isolation process but also significantly enhances the economic viability of producing 2,4-disubstituted trifluoromethyl benzene derivatives, making it a highly attractive route for the reliable pharmaceutical intermediate supplier seeking to optimize their portfolio.
Mechanistic Insights into Amine Salt-Mediated Regioselective Lithiation
The superior performance of this lithiation protocol stems from the unique interplay between the steric properties of the amine base and the electronic effects of the trifluoromethyl groups. In the conventional n-butyllithium pathway, the small size of the butyl group allows it to access multiple positions on the ring, leading to a mixture of kinetic and thermodynamic products. In contrast, the bulky amine lithium salts, generated in situ from secondary amines like 2,2,6,6-tetramethylpiperidine or diisopropylamine, create a massive steric shield around the lithium center. This steric bulk prevents the base from approaching the more crowded positions adjacent to both trifluoromethyl groups, effectively forcing the deprotonation to occur at the position that yields the 2,4-substitution pattern upon quenching. The reaction is typically conducted in ether solvents like tetrahydrofuran (THF) at temperatures ranging from -80°C to room temperature, with optimal results often observed between -10°C and -30°C. This temperature flexibility is crucial for scale-up, as it reduces the energy load associated with maintaining deep cryogenic conditions while maintaining high fidelity in product formation.
Furthermore, the mechanism facilitates excellent impurity control by minimizing side reactions such as nucleophilic attack on the trifluoromethyl groups or ring metalation at unintended sites. The patent highlights that the resulting lithiated intermediate is stable enough to be reacted with a wide variety of electrophiles, including carbon dioxide, DMF, methyl iodide, and halogens, without losing its regiochemical integrity. For instance, when quenched with solid carbon dioxide, the process yields 2,4-bis(trifluoromethyl)benzoic acid with high selectivity, while reaction with DMF produces the corresponding benzaldehyde. The ability to tolerate diverse electrophiles while maintaining the 2,4-configuration ensures that this platform technology can be adapted for the synthesis of various high-purity OLED material precursors or agrochemical intermediates. The robustness of the catalytic cycle and the stability of the intermediates provide R&D teams with a versatile tool for constructing complex molecular architectures with precision.
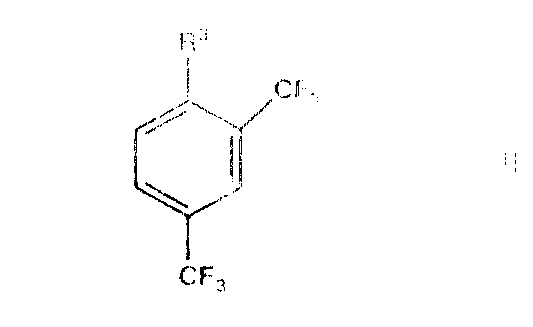
How to Synthesize 2,4-Bis(trifluoromethyl)benzene Derivatives Efficiently
Implementing this advanced lithiation strategy requires careful attention to reagent preparation and temperature management to fully realize the benefits of regioselectivity. The process begins with the generation of the active lithium amide base, typically by reacting the chosen secondary amine with a standard alkyllithium solution in an anhydrous ether solvent under an inert atmosphere. Once the base is formed, the substrate, 1,3-bis(trifluoromethyl)benzene, is introduced at controlled low temperatures to initiate the deprotonation. The detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios, addition rates, and workup procedures necessary to achieve the high yields and purities reported in the patent literature. Adhering to these protocols ensures that the steric advantages of the amine base are fully utilized, preventing the formation of the unwanted 2,6-isomer and maximizing the output of the valuable 2,4-product.
- Prepare the lithiation reagent by reacting a secondary amine, such as 2,2,6,6-tetramethylpiperidine, with n-butyllithium in an ether solvent like THF at low temperatures ranging from -40°C to 0°C.
- Add 1,3-bis(trifluoromethyl)benzene to the cooled amine lithium salt solution, maintaining the temperature below 0°C to ensure high regioselectivity for the 2,4-position.
- Quench the resulting lithiated intermediate with the desired electrophile, such as solid carbon dioxide or DMF, followed by acidic workup and purification via distillation or crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this amine-mediated lithiation technology offers substantial advantages that resonate deeply with procurement managers and supply chain heads focused on cost reduction in pharmaceutical intermediate manufacturing. The primary driver of value is the drastic simplification of the purification train. Because the reaction inherently produces a crude mixture dominated by the desired 2,4-isomer, the need for expensive and time-consuming separation techniques is virtually eliminated. This translates directly into lower processing costs, reduced solvent consumption, and shorter batch cycles, allowing manufacturers to increase throughput without expanding facility footprint. Additionally, the patent describes a viable method for recovering the expensive amine reagent from the aqueous waste stream, which can be recycled back into the process. This closed-loop capability significantly lowers the raw material cost per kilogram of finished product, providing a competitive edge in pricing negotiations with downstream API producers.
- Cost Reduction in Manufacturing: The elimination of complex isomer separation steps removes a major cost center from the production budget. Traditional methods often require multiple distillation columns or crystallization stages to isolate the target isomer from a near-1:1 mixture, consuming vast amounts of energy and solvents. By shifting the selectivity to favor the target product >90%, this technology allows for simpler workups, often requiring only a single crystallization or distillation to achieve specification-grade purity. Furthermore, the ability to recover and reuse the bulky amine base mitigates the impact of volatile raw material prices, ensuring stable long-term costing models for large volume contracts.
- Enhanced Supply Chain Reliability: The robustness of this chemical process contributes significantly to supply continuity. Unlike cryogenic processes that are sensitive to minor temperature fluctuations and equipment failures, this method operates effectively at moderately low temperatures (e.g., -10°C to -30°C), which are easier to maintain reliably in multi-purpose reactors. The use of commercially available starting materials and the tolerance for a range of electrophiles mean that supply chains are less vulnerable to single-source bottlenecks. For supply chain heads, this reliability reduces the risk of production delays and ensures that delivery commitments to global pharmaceutical partners can be met consistently, even during periods of high market demand.
- Scalability and Environmental Compliance: Scaling this reaction from laboratory to commercial production is facilitated by the manageable exotherm and the use of standard organic solvents like THF and hexane. The patent examples demonstrate successful execution on scales sufficient to produce hundreds of grams to kilograms, indicating a clear path to tonnage production. From an environmental standpoint, the reduction in solvent usage for purification and the recycling of the amine reagent align with green chemistry principles, reducing the overall E-factor of the process. This compliance with environmental standards simplifies regulatory approvals and waste disposal logistics, making it a sustainable choice for long-term manufacturing partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this lithiation technology, derived directly from the experimental data and claims of Patent CN1028994C. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this route into their existing manufacturing platforms. The answers provided reflect the specific capabilities of the amine-salt method, highlighting its superiority over legacy n-butyllithium processes in terms of selectivity, yield, and operational safety. We encourage potential partners to review these details to appreciate the depth of optimization achieved in this proprietary methodology.
Q: How does this lithiation method improve isomer selectivity compared to traditional n-BuLi?
A: Traditional n-butyllithium methods typically yield a mixture of 2,4- and 2,6-isomers with ratios close to 1:1 or 3:2, making separation difficult. The patented method using bulky amine lithium salts (like LiTMP) achieves a 2,4-to-2,6 ratio of 4:1 to over 100:1, drastically simplifying purification.
Q: What are the optimal temperature conditions for this reaction?
A: While conventional lithiation often requires cryogenic temperatures like -78°C, this process operates effectively between -80°C and room temperature, with preferred embodiments showing excellent results at -10°C to -30°C, reducing energy costs.
Q: Can the amine reagent be recovered to reduce material costs?
A: Yes, the patent explicitly describes a recovery process where the amine (e.g., 2,2,6,6-tetramethylpiperidine) is extracted from the acidic aqueous waste phase, basified, and distilled for reuse, achieving recovery yields of approximately 94%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Bis(trifluoromethyl)benzene Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality fluorinated intermediates in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature can be faithfully reproduced on an industrial scale. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of 1,3-bis(trifluoromethyl)benzene derivative meets the exacting standards required for API synthesis. Our commitment to process excellence means we can offer this advanced lithiation technology as a value-added service, delivering cost-effective solutions without compromising on the chemical integrity of your supply chain.
We invite you to engage with our technical procurement team to discuss how this regioselective lithiation method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this high-selectivity route for your specific application. We are ready to provide specific COA data and route feasibility assessments to support your R&D and sourcing decisions, ensuring a seamless transition to a more efficient and reliable supply model for your critical fluorinated building blocks.
