Transition-Metal-Free Borylation of Alkyl Bromides: A Scalable Route for High-Purity Intermediates
Transition-Metal-Free Borylation of Alkyl Bromides: A Scalable Route for High-Purity Intermediates
The landscape of organic synthesis is undergoing a significant transformation with the emergence of sustainable, metal-free methodologies that address the stringent purity requirements of the pharmaceutical industry. Patent CN110885341B introduces a groundbreaking approach for the boron esterification of alkyl bromides without the reliance on transition metal catalysts, marking a pivotal shift from traditional cross-coupling paradigms. This innovation leverages a radical-mediated mechanism utilizing silanes and radical initiators to effectively convert carbon-bromine bonds into valuable carbon-boron bonds under mild heating conditions. The significance of this technology lies in its ability to bypass the complexities associated with metal-ligand optimization and the subsequent removal of toxic metal residues, which are critical pain points in the manufacture of active pharmaceutical ingredients. By enabling the direct use of readily available alkyl bromides, this method opens new avenues for the efficient construction of complex molecular architectures essential for drug discovery and development.
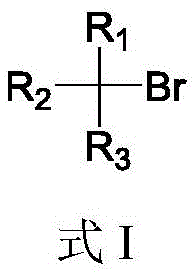
Traditional methods for synthesizing alkyl boronic esters have long been plagued by significant limitations that hinder their widespread adoption in large-scale industrial settings. Historically, the preparation of these vital intermediates relied heavily on the use of highly reactive organometallic reagents such as Grignard reagents or organolithium compounds, which suffer from poor functional group tolerance and require cryogenic conditions that are energy-intensive and difficult to manage safely. Furthermore, while transition metal-catalyzed borylation using copper, nickel, or palladium has offered a more versatile alternative, these processes introduce their own set of challenges, including the necessity for expensive ligands, sensitivity to air and moisture, and the critical issue of residual metal contamination in the final product. The screening of optimal metal-ligand combinations is often a time-consuming and laborious process that delays project timelines, and the reliance on alkyl iodides as substrates further escalates raw material costs due to the lower abundance and higher price of iodine compared to bromine. These cumulative factors create a substantial bottleneck for procurement and supply chain teams seeking reliable and cost-effective sources of high-quality building blocks.
In stark contrast, the novel approach detailed in the patent data presents a robust and streamlined solution that circumvents these historical obstacles through a cleverly designed radical chain mechanism. This method operates efficiently in the absence of any transition metal catalyst, utilizing a combination of a diboron source, a silane reductant, and a radical initiator to drive the transformation. The reaction conditions are remarkably forgiving, proceeding effectively in polar aprotic solvents like N,N-dimethylacetamide at temperatures ranging from 20 to 150°C, with optimal results often achieved around 80°C. This thermal stability allows for simpler reactor configurations and reduces the energy burden associated with cryogenic cooling. Moreover, the system exhibits exceptional functional group compatibility, accommodating primary, secondary, and even tertiary alkyl bromides, as well as substrates containing esters, ethers, and other sensitive moieties that would typically decompose under classical organometallic conditions. The use of alkyl bromides instead of iodides represents a strategic advantage, leveraging cheaper and more abundant starting materials to drive down the overall cost of goods sold.
Mechanistic Insights into Radical-Mediated Borylation
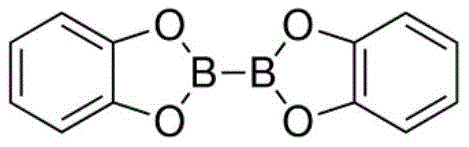
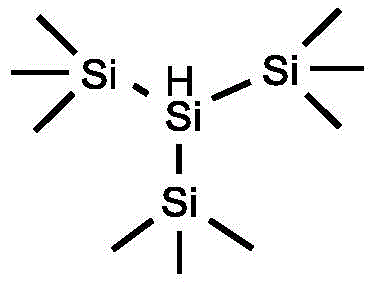
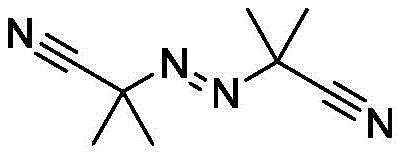
A deep understanding of the underlying reaction mechanism is crucial for R&D directors evaluating the feasibility and robustness of this new synthetic route. The proposed pathway operates via a free-radical chain mechanism initiated by the thermal decomposition of an azo compound, such as azobisisobutyronitrile (AIBN), which generates the initial radical species required to kickstart the cycle. These primary radicals abstract a hydrogen atom from the silane reagent, specifically tris(trimethylsilyl)silane (TTMSS), to generate a highly reactive silyl radical. This silyl radical then engages in a halogen atom transfer process with the alkyl bromide substrate, cleaving the carbon-bromine bond to release a bromosilane byproduct and, critically, generating the desired alkyl radical intermediate. This alkyl radical subsequently attacks the boron-boron bond of the diboron reagent, forming the new carbon-boron bond and propagating the radical chain by regenerating a boryl radical or interacting with another silane molecule. This elegant cascade avoids the formation of high-energy organometallic intermediates that are prone to beta-hydride elimination, a common side reaction in transition metal catalysis that leads to alkene byproducts and reduced yields.
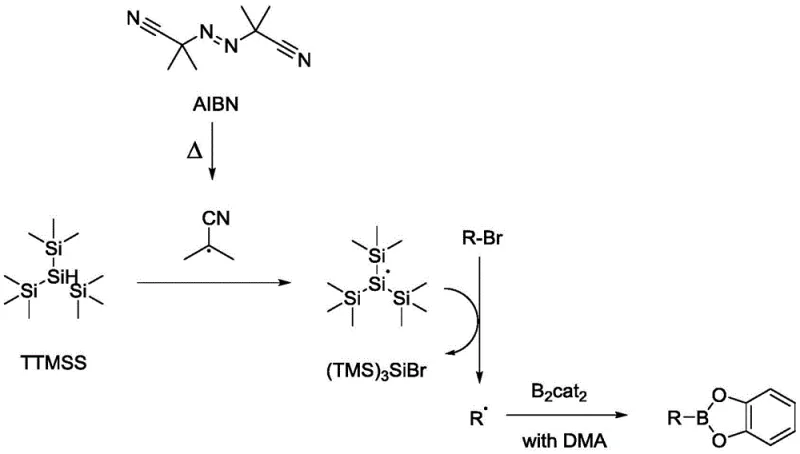
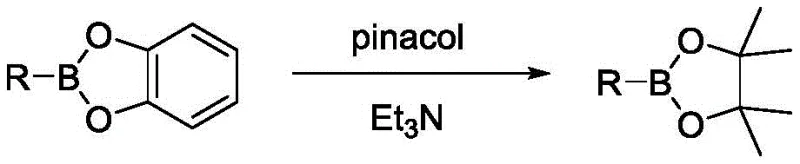
The control of impurities and byproducts is inherently superior in this metal-free system due to the absence of transition metal complexes that can coordinate with product molecules or catalyze undesired decomposition pathways. In traditional palladium-catalyzed reactions, trace amounts of metal can remain trapped in the crystal lattice of the product or form stable complexes that are difficult to remove, necessitating additional purification steps like scavenging resins or recrystallization which lower overall yield. In this radical process, the primary byproducts are silicon-containing species and the spent radical initiator fragments, which are generally easier to separate from the organic product during the aqueous workup phase. The patent describes a specific post-reaction treatment where the initially formed catechol boronate ester, which may be air-sensitive, is converted into the more stable pinacol boronate ester by treatment with pinacol and triethylamine. This derivatization step not only stabilizes the product for storage and transport but also facilitates purification via standard column chromatography or crystallization techniques. The ability to isolate the product as a stable pinacol ester ensures that the material meets the stringent stability specifications required for global supply chains without degradation during shipping.
How to Synthesize Alkyl Boronic Esters Efficiently
The practical implementation of this synthesis route is designed to be straightforward and adaptable to existing manufacturing infrastructure, requiring no specialized equipment beyond standard glass-lined or stainless steel reactors capable of maintaining an inert atmosphere. The process begins with the charging of the reactor with the alkyl bromide substrate, the diboron reagent, the silane, and the radical initiator dissolved in a suitable polar aprotic solvent such as DMA. The detailed standardized synthesis steps involve precise stoichiometric control, typically using a slight excess of the diboron and silane reagents to ensure complete conversion of the valuable alkyl bromide. Following the reaction period at elevated temperature, the mixture is cooled, and the stabilization step with pinacol is performed directly in the pot or after a simple solvent swap, minimizing unit operations. For a comprehensive guide on the exact molar ratios, temperature profiles, and workup procedures validated by experimental data, please refer to the technical protocol below.
- Mix alkyl bromide, bis-catechol diboron (B2cat2), tris(trimethylsilyl)silane (TTMSS), and AIBN in DMA solvent under inert atmosphere.
- Heat the sealed reaction mixture to 80°C and stir for approximately 6 hours to facilitate radical generation and carbon-boron bond formation.
- Perform workup by adding pinacol and triethylamine to convert the intermediate to a stable pinacol ester, followed by extraction and chromatographic purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology offers compelling advantages that directly address the key performance indicators of procurement managers and supply chain heads, particularly regarding cost structure and supply reliability. The elimination of transition metal catalysts removes a significant cost center associated with the purchase of precious metals like palladium or nickel, as well as the expensive phosphine or N-heterocyclic carbene ligands often required to sustain their activity. Furthermore, the avoidance of these metals negates the need for costly downstream processing steps dedicated to metal scavenging and validation of low residual metal levels, which are mandatory for pharmaceutical grade materials. This simplification of the purification train translates into substantial cost savings in terms of both material consumption and processing time, allowing for a more competitive pricing structure for the final alkyl boronic ester intermediates. The use of alkyl bromides, which are commodity chemicals with established global supply chains, further insulates the production process from the volatility and scarcity issues often associated with specialized alkyl iodides or custom-synthesized organometallic reagents.
- Cost Reduction in Manufacturing: The economic benefits of this metal-free process are driven primarily by the removal of expensive catalytic systems and the simplification of the purification workflow. By avoiding the use of precious metals, manufacturers eliminate the capital tie-up associated with catalyst recovery systems and the operational costs of metal analysis and certification. Additionally, the reaction proceeds with high efficiency and broad substrate scope, reducing the waste generated from failed optimization runs or low-yielding transformations common in sensitive metal-catalyzed protocols. The ability to use cheaper alkyl bromide feedstocks instead of iodides provides a direct reduction in raw material costs, which is a significant factor when scaling to multi-ton production volumes. These cumulative efficiencies result in a leaner manufacturing process that maximizes asset utilization and minimizes the cost per kilogram of the active intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the reliance on robust, commercially available reagents that are not subject to the geopolitical or logistical constraints often affecting specialty catalysts. Alkyl bromides, silanes, and radical initiators are produced by multiple suppliers globally, ensuring that production schedules are not disrupted by single-source bottlenecks. The operational simplicity of the reaction, which does not require rigorous exclusion of moisture to the same extent as organolithium chemistry or sensitive palladium cycles, reduces the risk of batch failures due to environmental fluctuations. This robustness ensures consistent delivery timelines and allows for flexible production planning, enabling suppliers to respond rapidly to changes in demand without compromising on quality or lead times. The stability of the final pinacol ester products further enhances supply chain security by allowing for longer shelf lives and safer transportation.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the homogeneous nature of the reaction mixture and the absence of heterogeneous catalysts that can cause filtration issues or reactor fouling at large scales. The reaction conditions are mild enough to be handled in standard chemical reactors without the need for exotic materials of construction, lowering the barrier to entry for contract manufacturing organizations. From an environmental standpoint, the metal-free nature of the process aligns perfectly with green chemistry principles, reducing the heavy metal load in wastewater streams and simplifying waste disposal protocols. This compliance with increasingly stringent environmental regulations reduces the administrative burden and potential liability associated with hazardous waste management, making the process more sustainable and socially responsible for long-term operations.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the integration of this technology into their existing pipelines, we have compiled answers to common inquiries regarding the operational parameters and scope of this metal-free borylation method. These responses are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a transparent view of the process capabilities. Understanding these details is essential for assessing the fit of this methodology for specific target molecules and for planning the necessary resource allocation for technology transfer. The following questions address the core concerns regarding substrate compatibility, reaction conditions, and product handling that typically arise during the feasibility assessment phase.
Q: Why is this metal-free borylation method superior to traditional transition-metal catalysis?
A: This method eliminates the need for expensive and toxic transition metal catalysts like Palladium or Nickel, thereby removing the requirement for rigorous metal scavenging steps and reducing heavy metal contamination risks in the final API.
Q: Can this process accommodate sensitive functional groups on the alkyl bromide substrate?
A: Yes, the radical mechanism demonstrates broad functional group compatibility, successfully tolerating esters, ethers, and various alkyl chains without the side reactions often seen with Grignard or organolithium reagents.
Q: What are the scalability advantages of using alkyl bromides over alkyl iodides in this process?
A: Alkyl bromides are generally more commercially available and cost-effective than the corresponding alkyl iodides, which simplifies the supply chain and significantly lowers the raw material costs for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyl Boronic Esters Supplier
As the pharmaceutical and fine chemical industries continue to demand higher purity standards and more sustainable manufacturing practices, the adoption of innovative synthetic routes like the one described in CN110885341B becomes a strategic imperative. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring these advanced metal-free technologies to market. Our state-of-the-art facilities are equipped to handle the specific requirements of radical chemistry, ensuring stringent purity specifications are met through our rigorous QC labs and advanced analytical capabilities. We understand that the transition to new synthetic methods requires a partner who can navigate the complexities of process optimization and regulatory compliance, and our team is dedicated to delivering high-purity alkyl boronic esters that meet the exacting needs of global drug developers.
We invite you to explore the potential of this cost-effective and environmentally friendly borylation technology for your next project by contacting our technical procurement team. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how switching to this metal-free route can optimize your budget and reduce lead time for high-purity building blocks. We are prepared to provide specific COA data and route feasibility assessments tailored to your target molecules, ensuring that you have all the information needed to make informed sourcing decisions. Partner with us to secure a reliable supply of critical intermediates that drive innovation while adhering to the highest standards of quality and sustainability.
