Advanced Beta-Dicarbonyl Organosilicon Coupling Agents: Technical Breakthroughs and Commercial Scalability
Advanced Beta-Dicarbonyl Organosilicon Coupling Agents: Technical Breakthroughs and Commercial Scalability
The landscape of silicone material modification is undergoing a significant transformation driven by the need for higher performance adhesion promoters, particularly for challenging substrates like fluororubber. Patent CN102491991B introduces a groundbreaking class of beta-dicarbonyl-containing organosilicon coupling agents that address longstanding limitations in the industry. These novel compounds leverage the unique coordination and chelation capabilities of the beta-dicarbonyl functional group to generate coplanar forces, thereby drastically increasing adhesion strength where traditional silanes fail. The structural versatility allows for R1 and R2 groups to be varied between phenoxy and alkoxy chains (C1-C11), while the silicon center can be modified with chloro, alkoxy, or alkyl groups, offering a broad spectrum of tunable properties for specific industrial requirements.
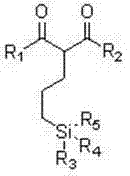
From a supply chain perspective, the robustness of this molecular architecture ensures stability under diverse processing conditions, making it an ideal candidate for high-performance composite materials. The presence of short-chain methoxy or ethoxy groups, as opposed to the long-chain alkoxy substituents found in older generations like KH550 or KH560, results in significantly higher hydrolytic activity. This enhanced reactivity facilitates rapid covalent bonding with inorganic fillers such as glass fibers and silica, ensuring that the resulting composite materials maintain superior mechanical integrity even in wet environments. For R&D directors seeking to optimize formulation efficiency, this patent provides a clear pathway to developing next-generation sealants and adhesives with improved weather resistance and electrical stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional organosilane coupling agents, such as the widely used KH series, often rely on long-chain alkoxy substitutions that inherently limit their reactivity and bonding efficiency. These conventional molecules frequently exhibit reduced activity when interacting with inorganic fillers, leading to suboptimal dispersion and weaker interfacial adhesion in the final composite matrix. Furthermore, the historical synthesis routes for functionalized silanes have often depended on harsh conditions, including the use of highly toxic noble metal catalysts for allylation reactions at elevated temperatures. Such processes not only incur high energy costs but also generate complex waste streams that require expensive remediation, creating a bottleneck for scalable and environmentally compliant manufacturing. The low purity and modest yields associated with these legacy methods further constrain their utility in high-specification applications like electronic encapsulation or aerospace composites.
The Novel Approach
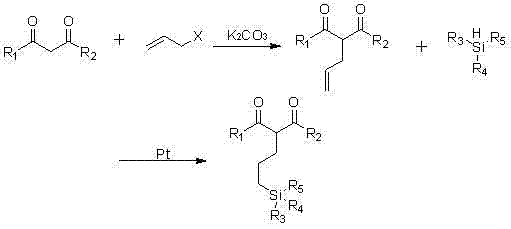
The methodology outlined in CN102491991B represents a paradigm shift by decoupling the synthesis into two highly efficient stages that prioritize both yield and environmental safety. The first stage utilizes anhydrous potassium carbonate as a benign catalyst for the allylation of beta-dicarbonyl compounds, completely avoiding the need for toxic heavy metals in this step. This is followed by a platinum-catalyzed hydrosilylation that attaches the silane moiety under mild conditions, typically between 0°C and 110°C. This two-step strategy not only simplifies the operational workflow but also achieves impressive yields, with experimental data showing up to 95% conversion in the allylation step and substantial recovery in the hydrosilylation phase. By replacing aggressive reagents with accessible bases like potassium carbonate, the process drastically reduces the thermal load and hazard profile of the manufacturing facility.
Mechanistic Insights into Platinum-Catalyzed Hydrosilylation
The core of the second synthetic step involves a sophisticated hydrosilylation mechanism mediated by platinum complexes, which is critical for installing the silicon functionality without compromising the sensitive beta-dicarbonyl group. The reaction proceeds through the coordination of the olefinic double bond of the allyl-beta-dicarbonyl intermediate to the platinum center, followed by the oxidative addition of the Si-H bond from the hydrosiloxane. This forms a transient platinum-hydride-silyl species that undergoes migratory insertion, effectively linking the silicon atom to the terminal carbon of the allyl chain. The choice of catalyst, ranging from chloroplatinic acid to zero-valent phosphine complexes, allows for fine-tuning of the reaction kinetics to prevent side reactions such as isomerization or dehydrogenative silylation. Maintaining strict control over the molar ratio of hydrosiloxane to allyl compound, typically between 1:1 and 5:1, ensures that the anti-Markovnikov addition product is favored, preserving the linear geometry required for optimal surface coverage on fillers.
Impurity control is paramount in this mechanism, as residual platinum or unreacted silanes can degrade the thermal stability of the final silicone resin. The patent specifies the use of polar organic solvents like tetrahydrofuran or acetonitrile, which help stabilize the transition states and facilitate the dissolution of the ionic intermediates. Post-reaction distillation at 60°C to 120°C is employed to separate the high-boiling coupling agent from volatile byproducts and solvent residues, ensuring a product of high chemical purity. This rigorous purification protocol is essential for applications in electronic materials, where ionic contaminants could lead to circuit failure. The mechanistic understanding of this catalytic cycle enables process engineers to scale the reaction from gram-scale laboratory batches to multi-ton industrial production while maintaining consistent quality and minimizing catalyst loading to as low as 0.1% molar equivalent.
How to Synthesize Beta-Dicarbonyl Organosilicon Coupling Agents Efficiently
The synthesis protocol described in the patent offers a reproducible framework for producing these high-value intermediates, starting with the preparation of the allyl-beta-dicarbonyl precursor. Operators should begin by reacting the chosen beta-dicarbonyl substrate, such as diethyl malonate or acetylacetone, with allyl chloride in the presence of potassium carbonate in acetonitrile at reflux temperatures. Following the isolation of the allylated intermediate, the subsequent hydrosilylation step requires careful addition of the hydrosiloxane to a solution containing the platinum catalyst and the intermediate, maintaining temperatures below 110°C to prevent thermal decomposition. Detailed standardized operating procedures regarding stoichiometry, solvent removal, and fractional distillation are critical for achieving the reported yields of 43% to 86% depending on the specific silane variant. For a comprehensive guide on the exact parameters and safety precautions, please refer to the structured synthesis steps provided below.
- Perform allylation of beta-dicarbonyl compounds (e.g., diethyl malonate) with allyl chloride using anhydrous potassium carbonate in polar solvents like acetonitrile at 0-110°C.
- Isolate the intermediate allyl-beta-dicarbonyl compound via extraction and drying, achieving yields up to 95% without heavy metal catalysts.
- Conduct hydrosilylation by reacting the intermediate with hydrosiloxanes (e.g., dimethoxymethylsilane) using a platinum complex catalyst at 0-110°C to form the final coupling agent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers compelling economic and logistical benefits that extend beyond simple raw material costs. The elimination of expensive and toxic noble metal catalysts in the initial allylation step translates directly into a leaner cost structure, reducing the dependency on volatile precious metal markets. Furthermore, the use of common inorganic bases like potassium carbonate simplifies the supply chain by sourcing readily available commodities rather than specialized organometallic reagents. The simplified post-treatment process, which avoids complex extraction and purification sequences associated with heavy metal removal, significantly shortens the production cycle time. This efficiency gain allows manufacturers to respond more agilely to market demand fluctuations, ensuring a steady flow of high-quality coupling agents to downstream customers in the rubber and coatings industries.
- Cost Reduction in Manufacturing: The strategic replacement of toxic noble metal catalysts with inexpensive potassium carbonate in the first reaction step creates a substantial reduction in direct material costs. By avoiding the high-temperature conditions traditionally required for noble metal-catalyzed allylation, the process also achieves significant energy savings, lowering the overall utility burden on the production facility. The high yields reported in the patent examples minimize waste generation and maximize the throughput of valuable intermediates, effectively driving down the cost per kilogram of the final product. These cumulative efficiencies result in a more competitive pricing structure for the end-user without compromising on the performance specifications of the silicone coupling agent.
- Enhanced Supply Chain Reliability: Sourcing reliability is greatly improved as the key reagents, including allyl chloride, beta-dicarbonyl compounds, and hydrosiloxanes, are bulk chemicals with stable global supply networks. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent choices, reduces the risk of batch failures due to minor process deviations. This operational resilience ensures consistent delivery schedules, mitigating the risk of production stoppages for customers who rely on these additives for continuous manufacturing lines. Additionally, the simplified logistics of handling non-hazardous catalysts reduce regulatory burdens and transportation complexities, further securing the supply chain against disruptions.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, with reaction parameters that are easily transferable from laboratory glassware to large-scale stainless steel reactors. The avoidance of highly toxic reagents aligns with increasingly stringent environmental regulations, reducing the costs associated with hazardous waste disposal and emissions control. The ability to produce methoxy and ethoxy-substituted silanes, which are more biodegradable and less persistent than long-chain analogs, enhances the environmental profile of the final composite products. This compliance advantage positions manufacturers as preferred suppliers for green-conscious multinational corporations seeking to reduce the carbon footprint of their material sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this beta-dicarbonyl silane technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this new coupling agent into existing formulations and supply chains. For further technical clarification or custom synthesis requests, our team is prepared to provide detailed support.
Q: What distinguishes this beta-dicarbonyl silane from conventional coupling agents?
A: Unlike conventional agents substituted with long-chain alkoxy groups which suffer from reduced activity, this novel structure features methoxy or ethoxy groups that provide higher reactivity and faster bonding with fillers. Additionally, the beta-dicarbonyl functional group offers unique coordination and chelation effects, significantly enhancing adhesion to difficult substrates like fluororubber.
Q: How does the patented synthesis method reduce production costs?
A: The process eliminates the need for highly toxic and expensive noble metal catalysts in the initial allylation step by utilizing inexpensive potassium carbonate. This substitution not only lowers raw material costs but also simplifies post-treatment and waste disposal, leading to substantial operational savings.
Q: What are the primary industrial applications of this coupling agent?
A: This agent is specifically designed to solve adhesion challenges in fluororubber and fluorosilicone rubber applications. It serves as a critical additive for preparing special silicone oils and resins, acting as a tackifier in sealants, adhesives, and coatings to improve mechanical strength and weather resistance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Dicarbonyl Organosilicon Coupling Agent Supplier
As a leader in the fine chemical sector, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure to translate the innovative pathways described in CN102491991B into commercial reality. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of coupling agent meets the exacting standards required for high-performance silicone applications. Whether you require standard variants or custom modifications of the R-groups to suit specific polymer matrices, our CDMO team is ready to execute your vision with precision.
We invite you to initiate a dialogue with our technical procurement team to explore how this advanced coupling agent can optimize your current formulations. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this more efficient synthesis route. We encourage you to contact us for specific COA data and route feasibility assessments tailored to your project requirements. Let us partner with you to enhance the adhesion performance and durability of your next-generation silicone materials.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
