Scalable Synthesis of Substituted Indolinones for Next-Generation Kinase Inhibitor Manufacturing
Scalable Synthesis of Substituted Indolinones for Next-Generation Kinase Inhibitor Manufacturing
The pharmaceutical landscape is continuously evolving towards more targeted therapies, particularly in the realm of oncology where kinase inhibitors play a pivotal role. Patent CN100338036C discloses a robust class of substituted indolinones exhibiting potent inhibitory effects on various kinases and cyclin/CDK complexes. These compounds, characterized by their unique structural framework, offer significant promise as therapeutic agents for treating diseases associated with abnormal cell proliferation, including various forms of cancer. The core innovation lies in the versatile substitution patterns allowed on the indolinone scaffold, enabling fine-tuning of pharmacological properties. As a reliable pharmaceutical intermediate supplier, understanding the depth of this chemical space is crucial for developing cost-effective manufacturing routes. The general structure, as defined in the patent, allows for extensive modification at the nitrogen atom and the benzene ring, providing a rich canvas for medicinal chemists to optimize bioavailability and potency.
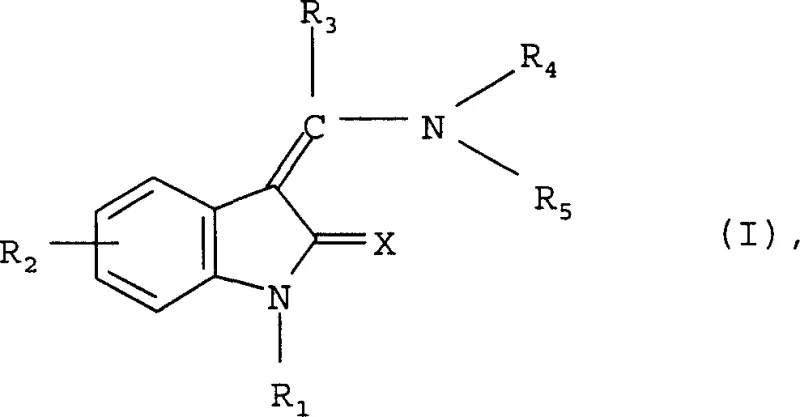
This technical insight report delves into the synthetic methodologies outlined in the patent, highlighting how these processes can be leveraged for commercial scale-up. The ability to synthesize these complex molecules efficiently is paramount for reducing lead time for high-purity pharmaceutical intermediates. By analyzing the reaction conditions and substrate scope, we can identify key opportunities for process intensification and cost reduction in API manufacturing. The patent not only covers solution-phase synthesis but also embraces solid-phase techniques, reflecting a modern approach to drug discovery that balances speed with purity. For procurement managers and supply chain heads, the implications of these synthetic routes extend beyond the laboratory, influencing raw material sourcing strategies and production timelines. The detailed exploration of these methods underscores the commitment to delivering high-quality chemical building blocks that meet the rigorous standards of the global pharmaceutical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of indolinone derivatives often suffers from multi-step sequences that involve harsh reaction conditions and expensive reagents. Conventional routes may require protecting group strategies that add unnecessary complexity and reduce overall yield. For instance, introducing specific substituents on the aromatic ring or the nitrogen atom can be challenging due to regioselectivity issues and the sensitivity of the indolinone core to strong acids or bases. Furthermore, purification of intermediates in traditional solution-phase synthesis frequently relies on extensive column chromatography, which is difficult to scale and generates significant solvent waste. These factors contribute to higher production costs and longer cycle times, creating bottlenecks in the supply chain for critical oncology drugs. The reliance on stoichiometric amounts of activating agents and the generation of stoichiometric byproducts further exacerbate environmental concerns and waste disposal costs. Consequently, there is a pressing need for more streamlined approaches that minimize step count and maximize atom economy while maintaining high purity standards required for clinical applications.
The Novel Approach
The methodology presented in CN100338036C offers a transformative alternative by utilizing direct condensation reactions between activated indolinone precursors and diverse amine components. This approach significantly simplifies the construction of the target molecule by forming the critical exocyclic double bond in a single operational step. The use of activated species, such as those containing halogen or alkoxy leaving groups at the 3-position, facilitates nucleophilic attack by amines under relatively mild conditions. Moreover, the patent highlights the versatility of this method by accommodating a wide range of amines, including cyclic amines, aryl amines, and functionalized alkyl amines, without compromising yield. The integration of solid-phase synthesis using resins like Rink or Sieber represents a paradigm shift, enabling rapid library generation and simplified workup procedures where filtration replaces complex extraction and chromatography. This novel approach not only enhances the efficiency of the synthesis but also improves the scalability, making it highly attractive for commercial scale-up of complex pharmaceutical intermediates. The flexibility to perform reactions in common organic solvents like DMF, dichloromethane, or toluene further supports easy adaptation to existing manufacturing infrastructure.
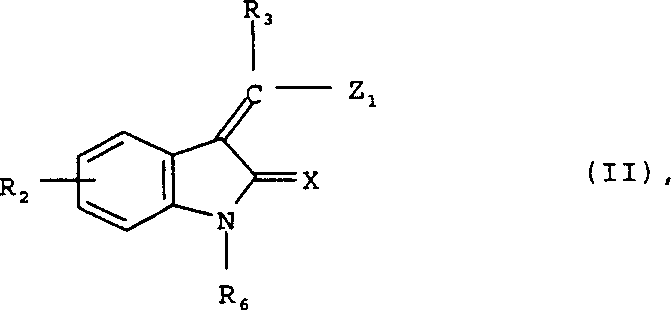
Mechanistic Insights into Condensation and Reduction Pathways
The core mechanistic pathway involves the nucleophilic addition-elimination reaction where the amine nitrogen attacks the electrophilic carbon at the 3-position of the activated indolinone. The leaving group, typically a halogen or alkoxy moiety, is displaced to form the stable exocyclic enamine structure. This reaction is driven by the thermodynamic stability of the conjugated system formed between the indolinone carbonyl and the newly formed double bond. The presence of electron-withdrawing or donating groups on the amine partner can influence the reaction kinetics, necessitating careful control of temperature and base selection. For example, less nucleophilic anilines may require elevated temperatures up to 175°C or the use of stronger bases to drive the reaction to completion. Conversely, highly reactive alkyl amines can proceed at ambient temperature, showcasing the adaptability of the process. Understanding these electronic effects is critical for R&D teams aiming to optimize reaction parameters for new analogues. The mechanism ensures high regioselectivity, preventing the formation of unwanted isomers that could complicate downstream purification and impact the safety profile of the final drug product.
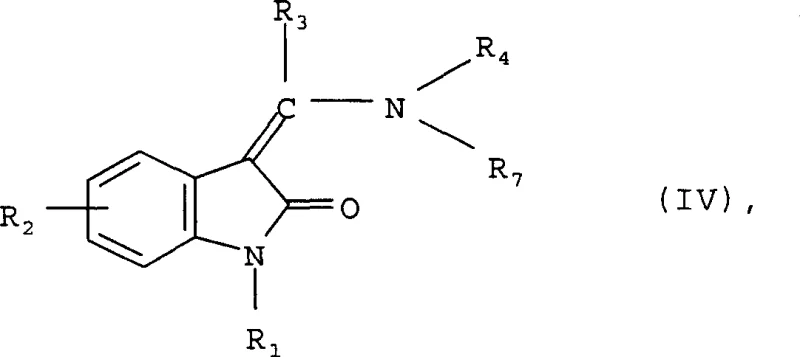
For derivatives containing amino-methyl groups, the patent describes a reduction strategy that is both efficient and scalable. This involves the catalytic hydrogenation of nitro or cyano precursors using heterogeneous catalysts such as palladium on carbon or Raney nickel. The mechanism entails the adsorption of hydrogen and the substrate onto the catalyst surface, followed by sequential reduction steps. This method is particularly advantageous because it avoids the use of hazardous reducing agents like lithium aluminum hydride, which pose significant safety risks on a large scale. The reaction can be conducted in protic solvents like methanol or ethanol, often with the addition of acid to facilitate the reduction of basic amines. The ability to tune the hydrogen pressure from 1 to 7 bar allows for precise control over the reaction rate and selectivity. Furthermore, the removal of the catalyst by simple filtration ensures that the final product is free from heavy metal residues, a critical quality attribute for pharmaceutical ingredients. This reduction step exemplifies a green chemistry approach that aligns with modern regulatory expectations for impurity control and process safety.
How to Synthesize Substituted Indolinones Efficiently
The synthesis of these valuable kinase inhibitors can be achieved through a systematic approach that prioritizes yield and purity. The process begins with the preparation of the activated indolinone synthon, followed by coupling with the desired amine partner. Detailed protocols for both solution-phase and solid-phase variations are available, allowing manufacturers to choose the best fit for their production volume. The following guide outlines the critical operational parameters derived from the patent data to ensure successful replication and scale-up. Adhering to these standardized steps minimizes variability and ensures consistent quality across batches. For comprehensive procedural details, refer to the structured synthesis guide below.
- Prepare the activated indolinone precursor (Formula II) by reacting 2-dihydroindole ketone derivatives with acetic anhydride or triethyl orthoformate to introduce the leaving group Z1.
- Conduct the condensation reaction by mixing the activated indolinone with the appropriate amine component (Formula III) in solvents like DMF or dichloromethane, optionally using mild bases.
- For amino-methyl derivatives, perform catalytic hydrogenation on nitro or cyano precursors using palladium/carbon or Raney nickel under controlled hydrogen pressure.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of the synthetic routes described in this patent offers substantial strategic benefits for procurement and supply chain management within the pharmaceutical sector. By streamlining the synthesis of complex indolinone scaffolds, manufacturers can achieve significant cost savings through reduced material consumption and shorter processing times. The elimination of cumbersome protecting group manipulations and the use of readily available starting materials contribute to a more lean and agile supply chain. This efficiency translates directly into improved margin potential and competitive pricing for the final active pharmaceutical ingredients. Furthermore, the robustness of the reaction conditions ensures high reliability in production scheduling, mitigating the risk of delays caused by sensitive chemical transformations. The ability to source raw materials from established chemical suppliers enhances supply security and reduces dependency on niche vendors. Overall, these process improvements foster a more resilient supply chain capable of meeting the dynamic demands of the global drug market.
- Cost Reduction in Manufacturing: The streamlined condensation reaction eliminates the need for multiple intermediate isolation steps, drastically reducing solvent usage and labor costs associated with purification. By avoiding expensive transition metal catalysts in the main coupling step and utilizing simple heterogeneous catalysts for reduction, the process minimizes the cost of goods sold. The high atom economy of the direct coupling method ensures that a larger proportion of raw materials ends up in the final product, reducing waste disposal fees. Additionally, the compatibility with common industrial solvents allows for solvent recovery and recycling, further driving down operational expenses. These cumulative efficiencies result in a markedly lower production cost per kilogram compared to traditional multi-step syntheses.
- Enhanced Supply Chain Reliability: The use of commercially available anilines and indolinone derivatives as starting materials ensures a stable and diversified supply base. The reaction conditions are tolerant to minor variations in reagent quality, reducing the risk of batch failures due to raw material specifications. The solid-phase option provides an alternative manufacturing route that can be rapidly deployed for small-batch production, offering flexibility in responding to fluctuating demand. The simplicity of the workup procedures, often involving just filtration and crystallization, shortens the manufacturing cycle time significantly. This agility allows supply chain managers to maintain lower inventory levels while still meeting delivery commitments, optimizing working capital utilization.
- Scalability and Environmental Compliance: The processes described are inherently scalable, having been demonstrated from gram to multi-kilogram scales in the patent examples. The avoidance of hazardous reagents and the use of catalytic hydrogenation align with green chemistry principles, simplifying regulatory compliance and environmental permitting. Waste streams are primarily composed of benign salts and organic solvents that can be treated using standard effluent management systems. The high purity of the crude product reduces the burden on downstream purification units, lowering energy consumption and water usage. This environmentally friendly profile enhances the corporate sustainability image and meets the increasingly stringent ecological standards imposed by global regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these substituted indolinones. The answers are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these aspects is vital for making informed decisions about process adoption and partnership opportunities. For further technical clarification, our team is available to provide detailed route feasibility assessments.
Q: What is the primary biological target of these substituted indolinones?
A: These compounds are designed to inhibit various cyclin-dependent kinases (CDKs) such as CDK1, CDK2, CDK4, and CDK6, as well as cyclin/CDK complexes, showing potent activity against tumor cell proliferation.
Q: Can this synthesis be adapted for solid-phase combinatorial chemistry?
A: Yes, the patent explicitly details the use of Rink or Sieber resins for solid-phase synthesis, allowing for efficient library generation and simplified purification workflows suitable for high-throughput screening.
Q: Are heavy metal catalysts required for the final reduction steps?
A: The process utilizes standard heterogeneous catalysts like palladium on carbon or Raney nickel for hydrogenation, which are easily removable via filtration, minimizing residual metal contamination in the final API intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Indolinones Supplier
The technological advancements detailed in CN100338036C represent a significant leap forward in the synthesis of kinase inhibitors, offering a pathway to more affordable and accessible cancer therapies. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate these patented methods into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of substituted indolinones meets the highest industry standards. Our commitment to quality assurance extends throughout the entire manufacturing process, from raw material inspection to final product release, providing you with peace of mind and regulatory confidence.
We invite you to collaborate with us to optimize your supply chain and reduce costs associated with API intermediate procurement. By leveraging our process development capabilities, we can conduct a Customized Cost-Saving Analysis tailored to your specific molecular targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. Together, we can accelerate the development of life-saving medications and bring innovative treatments to patients faster. Let us be your partner in navigating the complexities of fine chemical manufacturing and achieving your commercial goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
