Advanced Synthesis of Dimethylamino Phenyl Acetic Acid Intermediates for Commercial Scale-Up
Advanced Synthesis of Dimethylamino Phenyl Acetic Acid Intermediates for Commercial Scale-Up
The pharmaceutical industry constantly seeks robust synthetic routes that balance safety, efficiency, and scalability, particularly for critical building blocks like multi-substituted dimethylamino phenyl acetic acid compounds. A groundbreaking approach detailed in patent CN111777520B offers a transformative solution to longstanding challenges in producing these valuable intermediates. This novel methodology replaces hazardous reductive amination protocols with a safer, two-step sequence involving methyl esterification followed by phase-transfer catalyzed N-methylation. By systematically addressing the toxicity and flammability issues associated with traditional sodium cyanoborohydride methods, this innovation provides a pathway for reliable pharmaceutical intermediate supplier operations to enhance their production capabilities. The technical breakthrough ensures that manufacturers can achieve product yields exceeding 80 percent while maintaining stringent safety standards required for modern GMP facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dimethylamino phenyl acetic acid derivatives has relied heavily on reductive amination strategies that present significant operational risks and environmental burdens. The conventional pathway typically involves reacting primary amine precursors with formaldehyde in the presence of sodium cyanoborohydride, a reagent known for its high flammability and toxicity profile. As illustrated in the traditional reaction scheme below, this process necessitates rigorous safety controls to manage the release of toxic formaldehyde gas and the handling of pyrophoric reducing agents.
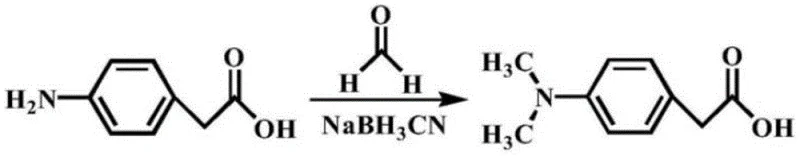
Furthermore, the conventional method often suffers from inconsistent mixing requirements, necessitating ultrasonic assistance or vigorous shaking to ensure proper dissolution of reactants in ultrapure water. These operational complexities not only increase energy consumption but also introduce variability in batch-to-batch reproducibility, which is unacceptable for high-purity API intermediate manufacturing. The reliance on such hazardous chemistry creates substantial liability for supply chain managers, as storage and transportation of sodium cyanoborohydride require specialized infrastructure and compliance with strict hazardous material regulations, thereby inflating the overall cost of goods sold.
The Novel Approach
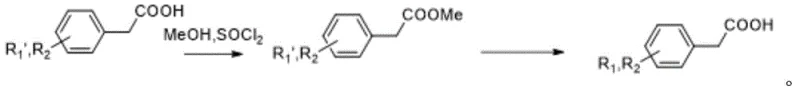
In stark contrast, the innovative method disclosed in the patent data introduces a streamlined, two-step synthetic route that fundamentally alters the risk profile of the manufacturing process. The new strategy begins with the conversion of the starting aminophenylacetic acid into a methyl ester intermediate, effectively protecting the carboxylic acid moiety and enhancing the solubility of the substrate in organic media. This esterification step sets the stage for a highly efficient N-methylation reaction conducted under phase transfer catalysis conditions, utilizing benign alkali bases like potassium hydroxide instead of dangerous reducing agents. By shifting the chemistry from reductive amination to nucleophilic substitution, the process eliminates the need for formaldehyde gas and cyanoborohydrides entirely.
This strategic pivot allows for milder reaction conditions, typically operating between 70 to 90 degrees Celsius, which significantly reduces thermal stress on equipment and lowers energy demands. The use of solid methylating agents, such as methyl p-toluenesulfonate, simplifies dosing and handling procedures, removing the complexity of gas-liquid reactions. Consequently, this novel approach not only improves the safety footprint of the facility but also enhances the economic viability of producing cost reduction in pharmaceutical intermediate manufacturing by minimizing waste treatment costs associated with toxic heavy metal or cyanide residues.
Mechanistic Insights into Phase Transfer Catalyzed N-Methylation
The core of this technological advancement lies in the sophisticated application of phase transfer catalysis (PTC) to drive the N-methylation of the aromatic amine. In the second step of the synthesis, the methyl ester intermediate reacts with an N-methylating agent in a biphasic system comprising an organic solvent and an aqueous alkaline solution. The phase transfer catalyst, preferably tetrabutylammonium bromide (TBAB), plays a pivotal role by shuttling hydroxide ions from the aqueous phase into the organic phase, where they deprotonate the amine nitrogen to generate a highly nucleophilic anionic species. This activated amine then attacks the methylating agent, such as methyl p-toluenesulfonate, facilitating the formation of the dimethylamino group with high regioselectivity.
Crucially, the initial esterification step serves a dual purpose beyond mere protection; it modifies the electronic and steric environment of the molecule to favor the desired alkylation over potential side reactions at the carboxylate position. The mechanism ensures that the reaction proceeds smoothly even with sterically hindered substrates, as evidenced by the successful synthesis of various ortho-substituted derivatives. Impurity control is inherently built into this mechanism, as the mild alkaline conditions prevent the degradation of sensitive functional groups that might occur under the acidic or strongly reducing conditions of the old method. This mechanistic robustness guarantees a cleaner crude product profile, reducing the burden on downstream purification processes and ensuring consistent quality for high-purity pharmaceutical intermediates.
How to Synthesize Dimethylamino Phenyl Acetic Acid Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and purity, leveraging the specific conditions outlined in the patent examples. The process is designed to be scalable, moving seamlessly from laboratory benchtop experiments to industrial reactor vessels without losing efficiency. Operators must first ensure the complete conversion of the starting acid to the methyl ester using thionyl chloride and methanol at controlled temperatures between 50 and 70 degrees Celsius. Following isolation of the ester, the subsequent methylation step demands careful monitoring of the molar ratios between the substrate, the methylating agent, and the phase transfer catalyst to maintain optimal kinetics.
- Perform methyl esterification of the aminophenylacetic acid starting material using thionyl chloride and methanol at 50-70°C.
- React the resulting methyl ester intermediate with an N-methylating agent under alkaline conditions using a phase transfer catalyst like TBAB.
- Purify the final product via extraction and chromatography to achieve yields exceeding 80%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis method translates directly into tangible operational improvements and risk mitigation strategies. The elimination of highly hazardous reagents like sodium cyanoborohydride removes a major bottleneck in raw material sourcing and storage, allowing facilities to operate with lower insurance premiums and reduced regulatory compliance overhead. By substituting dangerous gases and pyrophoric solids with stable, commercially available liquids and salts, the supply chain becomes more resilient against disruptions caused by hazardous material transport restrictions. This shift enables a more agile response to market demands, ensuring continuous availability of critical intermediates without the fear of safety-related shutdowns.
- Cost Reduction in Manufacturing: The transition to a phase transfer catalyzed process significantly lowers the cost of goods by removing the need for expensive and specialized reducing agents. Since the reaction utilizes inexpensive bases like potassium hydroxide and common quaternary ammonium salts, the raw material cost structure is drastically optimized. Furthermore, the simplified workup procedure, which avoids complex quenching of reactive hydrides, reduces labor hours and solvent consumption, leading to substantial cost savings in the overall production cycle.
- Enhanced Supply Chain Reliability: Sourcing stable solid methylating agents and standard industrial chemicals is far more reliable than managing the supply of unstable or tightly regulated toxic substances. This stability ensures that production schedules are not compromised by delays in obtaining specialized reagents, thereby reducing lead time for high-purity pharmaceutical intermediates. The robustness of the process also means that multiple suppliers can potentially provide the necessary inputs, fostering a competitive procurement environment that further drives down costs.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic heavy metals or cyanide byproducts make this process exceptionally easy to scale from pilot plants to commercial tonnage production. Waste streams are significantly less hazardous, simplifying effluent treatment and reducing the environmental footprint of the manufacturing site. This alignment with green chemistry principles not only satisfies increasingly strict environmental regulations but also enhances the corporate sustainability profile, which is a key metric for modern supply chain evaluations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and experimental data. Understanding these details is crucial for R&D teams evaluating the feasibility of adopting this route for their specific pipeline projects. The answers reflect the proven capabilities of the method to handle diverse substitution patterns while maintaining high efficiency and safety standards.
Q: Why is the new phase transfer catalysis method safer than the traditional reductive amination route?
A: The traditional route relies on sodium cyanoborohydride, a highly flammable and toxic reducing agent, and formaldehyde gas. The new method utilizes solid methylating agents like methyl p-toluenesulfonate under mild alkaline conditions, eliminating fire hazards and toxic gas exposure.
Q: What represents the key yield improvement in this synthesis protocol?
A: By protecting the carboxylic acid as a methyl ester prior to N-methylation, the reaction avoids side reactions and solubility issues common in direct aqueous alkylation, consistently delivering yields above 80% across various substitution patterns.
Q: Is this process suitable for large-scale manufacturing of API intermediates?
A: Yes, the process uses inexpensive reagents like potassium hydroxide and tetrabutylammonium bromide, operates at moderate temperatures (70-90°C), and avoids hazardous cryogenic or high-pressure steps, making it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dimethylamino Phenyl Acetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving needs of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this patented process are fully realized at an industrial level. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of dimethylamino phenyl acetic acid intermediate performs consistently in your downstream synthesis applications.
We invite you to collaborate with us to leverage this superior technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can support your supply chain goals and drive innovation in your drug development programs.
