Revolutionizing Montelukast Intermediate Production: A Safe, Scalable, and Cyanide-Free Synthetic Route
The pharmaceutical industry is constantly seeking robust synthetic pathways that balance efficiency with safety, particularly for high-volume asthma medications like Montelukast. Patent CN103288695A introduces a groundbreaking preparation method for 1-mercaptomethylcyclopropyl acetic acid, a critical intermediate in the synthesis of this blockbuster drug. Historically, the production of this molecule has been plagued by the use of hazardous reagents such as sodium cyanide and explosive diazomethane, which create significant bottlenecks in regulatory approval and environmental compliance. This new technology fundamentally shifts the paradigm by utilizing 1,4-butanediol as a cheap, low-toxicity starting material, navigating through a series of etherification, oxidation, and cyclopropanation steps to achieve the target structure. For R&D Directors and Supply Chain Heads, this patent represents not just a chemical curiosity, but a viable industrial solution that mitigates risk while enhancing yield. The method promises to resolve the chronic issues of heavy environmental pollution and low yield associated with legacy processes, offering a cleaner, more sustainable manufacturing footprint that aligns with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for 1-mercaptomethylcyclopropyl acetic acid have long been dependent on the use of sodium cyanide for nucleophilic substitution, a practice that is increasingly untenable in modern pharmaceutical manufacturing. The reliance on cyanide introduces severe safety hazards, requiring specialized containment facilities and complex waste treatment protocols that drastically inflate operational costs. Furthermore, existing methods often involve multi-step sequences with poor atom economy, leading to significant material loss and difficult purification challenges that compromise overall yield. The use of explosive reagents like diazomethane in some alternative routes further exacerbates safety concerns, making scale-up a dangerous endeavor that many facilities are unwilling to undertake. These legacy processes create a fragile supply chain where production continuity is constantly threatened by regulatory scrutiny and the inherent dangers of handling highly toxic substances. Consequently, manufacturers face substantial liability risks and potential production stoppages, making the search for a safer alternative not just an optimization goal, but a strategic necessity for business continuity.
The Novel Approach
The patented method described in CN103288695A offers a transformative alternative by completely eliminating the need for sodium cyanide and diazomethane, replacing them with a safer, more controllable sequence starting from 1,4-butanediol. This novel approach leverages a strategic protection-deprotection strategy where the starting diol is first converted into a stable intermediate through etherification or esterification, setting the stage for subsequent transformations without the risk of over-oxidation.  . By avoiding toxic cyanide salts, the process significantly reduces the burden on waste treatment systems and lowers the barrier for regulatory approval in strict jurisdictions. The route is designed with industrial scalability in mind, utilizing reagents that are commercially available and easy to handle, thereby simplifying the procurement process and reducing raw material costs. This shift from hazardous chemistry to a benign, high-yield pathway ensures that production can be scaled from pilot batches to multi-ton commercial runs without the exponential increase in safety overhead typically associated with cyanide-based chemistry.
. By avoiding toxic cyanide salts, the process significantly reduces the burden on waste treatment systems and lowers the barrier for regulatory approval in strict jurisdictions. The route is designed with industrial scalability in mind, utilizing reagents that are commercially available and easy to handle, thereby simplifying the procurement process and reducing raw material costs. This shift from hazardous chemistry to a benign, high-yield pathway ensures that production can be scaled from pilot batches to multi-ton commercial runs without the exponential increase in safety overhead typically associated with cyanide-based chemistry.
Mechanistic Insights into Corey-Chaykovsky and Simmons-Smith Cyclopropanation
A pivotal moment in this synthetic route is the formation of the cyclopropane ring, a structural motif essential for the biological activity of the final Montelukast molecule. The patent details the use of either Corey-Chaykovsky or Simmons-Smith reactions to convert the alpha,beta-unsaturated aldehyde intermediate into the corresponding cyclopropyl derivative with high stereochemical control. In the Corey-Chaykovsky variant, a sulfur ylide reagent reacts with the electron-deficient alkene to form the three-membered ring under mild alkaline conditions, avoiding the harsh conditions that might degrade sensitive functional groups. Alternatively, the Simmons-Smith reaction utilizes a zinc-copper couple and methylene iodide to achieve the same cyclopropanation, offering flexibility depending on the specific substrate requirements and available infrastructure. 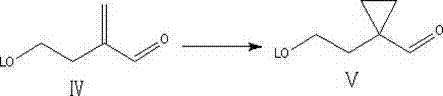 . This mechanistic versatility is crucial for process chemists who need to adapt the synthesis to different reactor configurations or raw material availabilities. The ability to form the cyclopropane ring efficiently without generating toxic byproducts ensures that the impurity profile of the intermediate remains clean, simplifying downstream processing and reducing the need for extensive chromatographic purification steps that are costly and time-consuming.
. This mechanistic versatility is crucial for process chemists who need to adapt the synthesis to different reactor configurations or raw material availabilities. The ability to form the cyclopropane ring efficiently without generating toxic byproducts ensures that the impurity profile of the intermediate remains clean, simplifying downstream processing and reducing the need for extensive chromatographic purification steps that are costly and time-consuming.
Following the ring formation, the synthesis proceeds through a carefully orchestrated sequence of reduction, halogenation, and substitution reactions that preserve the integrity of the cyclopropane ring while installing the necessary mercapto group. The reduction of the aldehyde to an alcohol is performed using mild metal hydrides, ensuring that the strained ring system is not opened or isomerized during the process. Subsequent halogenation activates the primary alcohol for nucleophilic substitution, where a thiourea reagent is employed to introduce the sulfur atom in a controlled manner. This substitution strategy is superior to direct thiolation methods which often suffer from poor selectivity and the formation of disulfide impurities. The final hydrolysis step releases the free thiol and the carboxylic acid simultaneously, delivering the target 1-mercaptomethylcyclopropyl acetic acid in high purity. The entire sequence is designed to minimize side reactions, ensuring that the impurity spectrum is predictable and manageable, which is a key requirement for GMP manufacturing of pharmaceutical intermediates.
How to Synthesize 1-Mercaptomethylcyclopropyl Acetic Acid Efficiently
Implementing this synthesis requires a precise understanding of the reaction conditions, particularly the temperature controls and reagent stoichiometry outlined in the patent examples. The process begins with the protection of 1,4-butanediol, followed by oxidation to the aldehyde, and then the critical cyclopropanation step which dictates the overall success of the route. Operators must maintain strict temperature ranges, such as keeping the oxidation step between -25 to -5°C to prevent over-oxidation to the carboxylic acid prematurely. The detailed standardized synthesis steps involve specific workup procedures, including extraction and distillation under reduced pressure, which are essential for isolating the intermediates in high purity. For a comprehensive breakdown of the exact reagent quantities, reaction times, and workup protocols required to replicate this high-yield process, please refer to the technical guide below.
- Protect 1,4-butanediol via etherification or esterification to form Intermediate II, ensuring stability for subsequent oxidation.
- Perform oxidation to aldehyde Intermediate III, followed by alkylation with formaldehyde to generate the alpha,beta-unsaturated Intermediate IV.
- Execute cyclopropanation using Corey-Chaykovsky or Simmons-Smith reagents, followed by reduction, halogenation, and thiourea substitution to yield the final acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this cyanide-free synthetic route offers profound strategic advantages that extend beyond simple chemical efficiency. By removing sodium cyanide from the bill of materials, manufacturers eliminate the need for specialized hazardous material handling licenses and the associated high costs of toxic waste disposal. This simplification of the safety profile allows for production in a wider range of facilities, increasing the potential supply base and reducing the risk of single-source bottlenecks. Furthermore, the use of 1,4-butanediol as a starting material leverages a commodity chemical with a stable and robust global supply chain, insulating the production process from the volatility often seen with specialized fine chemical reagents. The overall simplification of the process flow, characterized by fewer purification steps and higher yields, translates directly into reduced manufacturing cycle times and lower operational expenditures. These factors combine to create a more resilient supply chain capable of meeting the demanding volume requirements of the global asthma medication market without compromising on safety or quality standards.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like sodium cyanide and diazomethane drastically reduces the raw material costs and the overhead associated with safety compliance. Without the need for complex cyanide destruction units or specialized containment infrastructure, the capital expenditure required for setting up production lines is significantly lowered. Additionally, the high yield and simplified purification process mean that less raw material is wasted per kilogram of final product, improving the overall material efficiency and reducing the cost of goods sold. The avoidance of column chromatography in favor of distillation and crystallization further reduces solvent consumption and processing time, leading to substantial operational savings that can be passed down the supply chain.
- Enhanced Supply Chain Reliability: Relying on commodity starting materials like 1,4-butanediol ensures a stable supply of raw inputs, as these chemicals are produced in massive volumes for various industries and are less prone to shortage than specialized reagents. The removal of toxic cyanide from the process also reduces the regulatory burden, minimizing the risk of production shutdowns due to environmental inspections or safety violations. This stability is crucial for long-term supply agreements with major pharmaceutical companies who require guaranteed continuity of supply for their blockbuster drugs. The robust nature of the chemistry allows for consistent batch-to-batch quality, reducing the rejection rate and ensuring that delivery schedules are met reliably without unexpected delays caused by failed quality control tests.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial large-scale production, with reaction conditions that are easily manageable in standard stainless steel reactors without the need for exotic materials of construction. The absence of heavy metal catalysts and toxic cyanide waste simplifies the effluent treatment process, making it easier to meet stringent environmental discharge standards in regions with strict regulations. This environmental compatibility future-proofs the manufacturing site against tightening global environmental laws, ensuring long-term operational viability. The scalability is further supported by the high yield at each step, which means that scaling up from pilot to commercial scale does not result in the typical drop in efficiency often seen in complex organic syntheses, allowing for seamless capacity expansion as market demand grows.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route for 1-mercaptomethylcyclopropyl acetic acid. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation, providing clarity on safety, purity, and scalability. Understanding these aspects is essential for technical teams evaluating the feasibility of adopting this new method for commercial production. The insights provided here aim to bridge the gap between laboratory-scale success and industrial-scale implementation, ensuring that all stakeholders have a clear understanding of the process capabilities.
Q: Why is the cyanide-free route critical for Montelukast intermediate manufacturing?
A: Traditional methods rely on highly toxic sodium cyanide, posing severe environmental and safety risks. The patented cyanide-free route eliminates these hazards, ensuring regulatory compliance and safer workplace conditions.
Q: How does the new synthetic route improve product purity?
A: By avoiding complex cyanide-based side reactions and utilizing mild oxidation and substitution conditions, the new method simplifies purification, allowing for high-purity isolation without extensive column chromatography.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process uses readily available starting materials like 1,4-butanediol and operates under mild conditions, making it highly scalable for industrial manufacturing with consistent yield and quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Mercaptomethylcyclopropyl Acetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting safer, more efficient synthetic routes for high-value pharmaceutical intermediates like 1-mercaptomethylcyclopropyl acetic acid. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to plant is seamless and efficient. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the exacting standards required for API synthesis. We are committed to leveraging this cyanide-free technology to provide our clients with a supply chain that is not only cost-effective but also environmentally responsible and safe. Our technical team is ready to assist in optimizing this route for your specific production needs, ensuring maximum yield and minimal environmental impact.
We invite you to collaborate with us to explore how this innovative synthesis can enhance your supply chain resilience and reduce your overall manufacturing costs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your current production volumes and requirements. We encourage you to reach out for specific COA data and route feasibility assessments to verify the compatibility of this method with your existing quality systems. Let us help you secure a sustainable and reliable source of this critical intermediate, ensuring that your production of Montelukast remains uninterrupted and competitive in the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
