Advanced Biotransformation of Sulfur-Substituted Podophyllum Derivatives for Commercial Pharmaceutical Applications
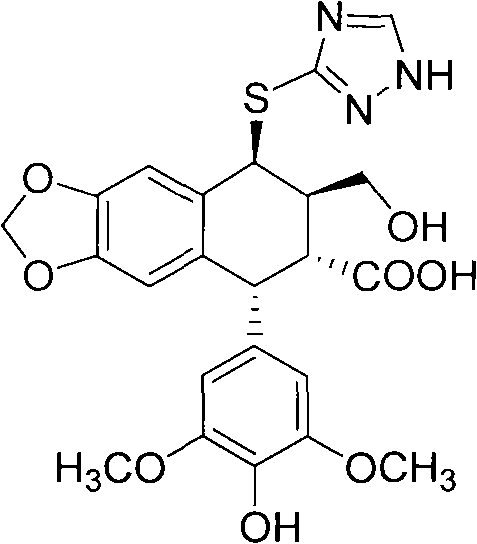
The pharmaceutical industry is constantly seeking sustainable and efficient pathways to produce complex antitumor intermediates, and patent CN102757443A presents a groundbreaking approach to synthesizing sulfur-substituted podophyllum derivatives. This technology leverages advanced microbial biotransformation to modify the molecular structure of podophyllotoxin or 4'-norepipodophyllotoxin, resulting in novel compounds with potent anticancer activity against cell lines such as BGC823, HepG2, and A549. Unlike traditional chemical synthesis which often struggles with harsh conditions and toxic residues, this biocatalytic method utilizes specific endophytic fungi to introduce sulfur-containing heterocycles under mild physiological conditions. The core innovation lies in the ability to perform regioselective modifications at the C-4 position of the podophyllum skeleton, creating valuable intermediates represented by Formula (I) and Formula (II). 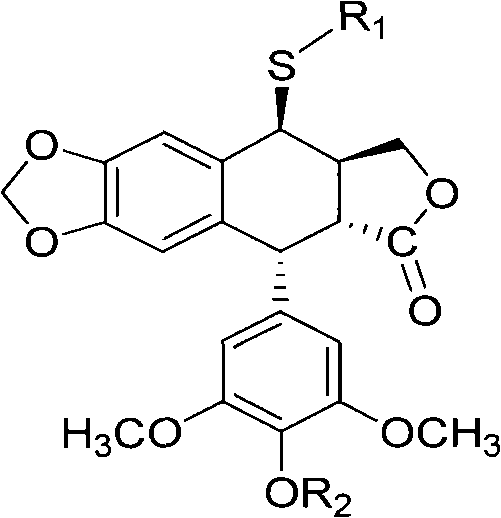
 For procurement and R&D teams, this represents a shift towards greener chemistry that aligns with modern regulatory demands for reduced environmental impact and higher product safety.
For procurement and R&D teams, this represents a shift towards greener chemistry that aligns with modern regulatory demands for reduced environmental impact and higher product safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the structural modification of podophyllum compounds has relied heavily on organic chemical synthesis, a process fraught with significant industrial drawbacks that hinder large-scale production. Conventional routes typically require aggressive reagents and extreme temperatures to form carbon-sulfur bonds, leading to poor yields and the generation of complex impurity profiles that are difficult to separate. Furthermore, the use of transition metal catalysts in traditional thiolation reactions introduces the risk of heavy metal contamination, necessitating expensive and time-consuming purification steps to meet pharmaceutical grade standards. The high E-factor associated with these chemical processes results in substantial organic waste, driving up disposal costs and creating environmental compliance burdens for manufacturers. Additionally, the scalability of chemical synthesis for these sensitive natural product derivatives is often limited by the instability of intermediates under harsh reaction conditions, making consistent commercial supply challenging to maintain.
The Novel Approach
In stark contrast, the biotransformation method disclosed in the patent utilizes the metabolic machinery of fungi like Penicillium purpurogenum to achieve the same structural modifications with remarkable efficiency and selectivity. This biological approach operates at ambient temperatures and neutral pH levels, effectively eliminating the thermal degradation issues common in chemical synthesis while preserving the delicate stereochemistry of the podophyllum core. By employing whole-cell biocatalysis, the process avoids the need for isolated enzymes or toxic chemical reagents, thereby simplifying the operational workflow and reducing the overall cost of goods. The inherent specificity of the microbial enzymes ensures that the sulfur substitution occurs precisely at the desired position, minimizing the formation of unwanted isomers and significantly streamlining the downstream purification process. This green chemistry paradigm not only enhances the sustainability of the manufacturing process but also provides a robust platform for the reliable production of high-purity pharmaceutical intermediates.
Mechanistic Insights into Fungal Biotransformation and Sulfur Incorporation
The mechanistic foundation of this technology rests on the unique metabolic capabilities of specific fungal strains to recognize and modify exogenous podophyllum substrates. When podophyllotoxin or its demethylated analogs are introduced into the fermentation broth, the fungal mycelium absorbs these compounds and subjects them to enzymatic transformations that facilitate the nucleophilic attack of sulfur-containing heterocycles. The process likely involves cytochrome P450 monooxygenases or transferases that activate the C-4 position of the substrate, allowing for the coupling of reagents such as 2-mercapto-1,3,4-thiadiazole or 3-mercapto-1,2,4-triazole.  This enzymatic catalysis ensures high regioselectivity, preventing random substitution on the aromatic rings which is a common side reaction in non-enzymatic chemical synthesis. The metabolic pathway is carefully regulated by optimizing the fermentation medium composition, including carbon sources like glucose and nitrogen sources like yeast extract, to maximize the expression of the relevant biosynthetic enzymes.
This enzymatic catalysis ensures high regioselectivity, preventing random substitution on the aromatic rings which is a common side reaction in non-enzymatic chemical synthesis. The metabolic pathway is carefully regulated by optimizing the fermentation medium composition, including carbon sources like glucose and nitrogen sources like yeast extract, to maximize the expression of the relevant biosynthetic enzymes.
From an impurity control perspective, the biological nature of the reaction inherently limits the types of byproducts formed, as the enzymes are highly specific for their substrates and reaction conditions. Unlike chemical synthesis which might produce a broad spectrum of halogenated or oxidized impurities due to reagent aggressiveness, the biotransformation pathway yields a cleaner crude product profile. The subsequent purification strategy involving silica gel column chromatography and gel filtration is designed to remove residual fungal metabolites and unreacted starting materials, ensuring the final product meets stringent purity specifications. This dual approach of enzymatic specificity followed by physical separation provides a robust mechanism for controlling the杂质谱 (impurity profile), which is critical for regulatory approval of antitumor drug candidates. The ability to consistently produce batches with low levels of genotoxic impurities makes this method particularly attractive for the manufacture of clinical-grade intermediates.
How to Synthesize Sulfur-Substituted Podophyllum Derivatives Efficiently
The synthesis of these valuable antitumor intermediates begins with the preparation of secondary liquid seeds of the selected fungal strains, followed by inoculation into a optimized fermentation medium containing the podophyllum substrate. The process requires precise control over fermentation parameters such as temperature, shaking speed, and pH to maintain high cell viability and enzymatic activity throughout the transformation period. After the bioconversion is complete, the broth undergoes a series of extraction and chromatographic steps to isolate the target sulfur-substituted compounds in high purity. For detailed operational parameters and specific media formulations, please refer to the standardized synthesis guide below.
- Cultivate specific endophytic fungi such as Penicillium purpurogenum in optimized liquid fermentation media containing glucose and yeast extract.
- Introduce podophyllotoxin or 4'-demethylepipodophyllotoxin substrates into the fermentation system along with sulfur-containing heterocycles like mercapto-triazoles.
- Execute separation and purification using silica gel column chromatography followed by gel filtration to isolate high-purity sulfur-substituted derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this biotransformation technology offers substantial strategic advantages in terms of cost stability and supply reliability. The shift from chemical synthesis to fermentation-based production fundamentally alters the cost structure by removing the dependency on expensive and volatile chemical reagents and catalysts. Since the process utilizes renewable biological resources and operates under mild conditions, the energy consumption and waste treatment costs are drastically simplified, leading to significant cost reduction in API manufacturing. Furthermore, the scalability of fermentation processes is well-established in the industry, allowing for seamless expansion from laboratory shake-flasks to industrial-scale bioreactors without the need for complex re-engineering of the production line.
- Cost Reduction in Manufacturing: The elimination of heavy metal catalysts and harsh organic solvents removes the need for costly scavenging steps and specialized waste disposal protocols, directly lowering the operational expenditure. By utilizing inexpensive fermentation media components and achieving high transformation ratios, the overall cost of raw materials is optimized, providing a more competitive pricing structure for the final intermediates. The simplified downstream processing also reduces labor and equipment time, contributing to a leaner and more cost-effective manufacturing model that enhances profit margins for downstream drug developers.
- Enhanced Supply Chain Reliability: Fermentation-based production is less susceptible to the supply chain disruptions often associated with specialty chemical reagents, as the biological catalysts can be maintained and propagated in-house. The robustness of the fungal strains ensures consistent batch-to-batch performance, minimizing the risk of production failures that could delay project timelines. This reliability is crucial for maintaining continuous supply chains for critical antitumor medications, ensuring that pharmaceutical partners can meet their own production schedules without interruption.
- Scalability and Environmental Compliance: The green nature of the biotransformation process aligns perfectly with increasingly strict environmental regulations, reducing the regulatory burden and potential fines associated with hazardous waste generation. The process is inherently scalable, allowing manufacturers to ramp up production volumes from 100 kgs to 100 MT annually to meet growing market demand without compromising on quality or sustainability goals. This scalability ensures that the supply chain can adapt quickly to market fluctuations, providing a secure source of high-quality intermediates for long-term drug development projects.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these sulfur-substituted podophyllum derivatives. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance for industry professionals.
Q: What are the primary advantages of biotransformation over chemical synthesis for podophyllum derivatives?
A: Biotransformation offers significantly milder reaction conditions, eliminates the need for toxic heavy metal catalysts, reduces organic solvent waste (low E-factor), and provides better control over stereochemistry compared to traditional harsh chemical methods.
Q: Which specific microbial strains are utilized in this patented process?
A: The process primarily utilizes Penicillium purpurogenum (CCTCC NO:M 2010262) for the initial conversion and Gibberella fujikuroi (CCTCC NO:M2010038) for further structural modifications, ensuring high transformation ratios.
Q: How does this method impact the purity profile of the final intermediate?
A: The enzymatic specificity of the fungal strains minimizes the formation of regio-isomeric byproducts common in chemical synthesis, while the subsequent multi-step chromatographic purification ensures stringent purity specifications suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfur-Substituted Podophyllum Derivatives Supplier
As a leader in the fine chemical sector, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications for complex pharmaceutical intermediates like those described in CN102757443A. We understand the critical importance of quality consistency in antitumor drug development and are committed to delivering products that meet the highest international standards.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project requirements. By partnering with us, you gain access to specific COA data and route feasibility assessments that can accelerate your development timeline. Let us help you leverage this innovative biotransformation technology to bring your next-generation antitumor therapies to market faster and more economically.
