Advanced Stereoselective Synthesis of Substituted Piperidine Derivatives for Commercial Scale-up
The pharmaceutical industry continuously seeks robust and scalable methods for producing high-purity intermediates, particularly those targeting complex neurological pathways. Patent CN1038932C discloses a novel stereoselective preparation method for substituted piperidine derivatives, which serve as potent Substance P receptor antagonists. These compounds are critical in the development of therapeutics for pain, inflammation, anxiety, and various gastrointestinal disorders. The technical breakthrough lies in the ability to maintain absolute stereochemistry throughout the synthetic sequence, ensuring the biological efficacy of the final active pharmaceutical ingredient. By leveraging specific hydrogenation and reductive amination protocols, this process offers a reliable pathway for manufacturing complex heterocyclic structures that were previously challenging to produce with high enantiomeric excess. 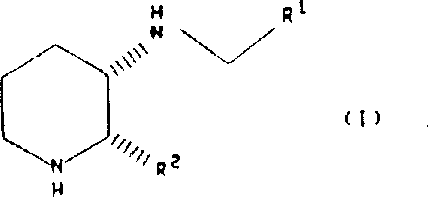
The significance of this technology extends beyond mere chemical synthesis; it addresses the critical need for supply chain stability in the production of neuroactive pharmaceutical intermediates. Traditional methods often struggle with racemization or require expensive chiral resolution steps that drastically increase manufacturing costs and lead times. In contrast, the methodology outlined in CN1038932C utilizes readily available starting materials and standard catalytic hydrogenation equipment, making it highly adaptable for commercial scale-up. For procurement managers and supply chain heads, this represents a strategic opportunity to secure a consistent source of high-quality intermediates without the volatility associated with proprietary chiral catalysts or obscure reagents. The process is designed to be operationally simple, reducing the technical barriers to entry for large-scale production facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted piperidine derivatives with specific stereochemical requirements has been fraught with inefficiencies and technical hurdles. Conventional routes often rely on non-selective reduction methods that produce racemic mixtures, necessitating downstream resolution processes that can discard up to half of the produced material. This not only inflates the cost of goods sold but also creates significant waste management challenges, conflicting with modern environmental compliance standards. Furthermore, traditional methods may require harsh reaction conditions, such as extreme temperatures or highly reactive reducing agents like lithium aluminum hydride, which pose safety risks and complicate process safety management in large reactors. The lack of stereocontrol in these older methodologies often results in impurity profiles that are difficult to purge, potentially compromising the safety and efficacy of the final drug product.
The Novel Approach
The approach detailed in patent CN1038932C overcomes these limitations by employing a stereoselective hydrogenation strategy that preserves the chiral integrity of the molecule from the starting material to the final product. By utilizing specific metal-containing catalysts, such as platinum on carbon or palladium on carbon, under controlled hydrogen pressure, the process selectively reduces the pyridine ring without affecting other sensitive functional groups. This selectivity is paramount for maintaining the structural complexity required for Substance P receptor binding. Additionally, the subsequent reductive amination steps are optimized to proceed under mild conditions using reagents like sodium triacetoxyborohydride, which offers superior chemoselectivity compared to traditional borohydrides. This novel approach streamlines the synthetic route, minimizes waste, and enhances the overall safety profile of the manufacturing process, making it an ideal candidate for modern green chemistry initiatives.
Mechanistic Insights into Stereoselective Hydrogenation and Reductive Amination
The core of this synthetic strategy involves the catalytic hydrogenation of substituted pyridine derivatives to form the corresponding piperidine intermediates. This transformation is mechanistically driven by the adsorption of hydrogen and the substrate onto the surface of the metal catalyst, facilitating the addition of hydrogen atoms across the unsaturated bonds of the pyridine ring. The patent specifies that this reduction can be achieved using various catalysts, including platinum, palladium, nickel, and rhodium, with platinum on carbon being particularly preferred for its high activity and selectivity. The reaction is typically conducted in inert solvents such as methanol, ethanol, or acetic acid, often in the presence of stoichiometric amounts of acid to form the salt directly. This step is crucial as it establishes the cis-configuration of the piperidine ring, which is retained throughout the subsequent synthetic transformations, ensuring the final product possesses the desired stereochemistry for biological activity.
Following the formation of the piperidine intermediate, the process proceeds via a reductive amination mechanism to introduce the diverse side chains defined by the R1 substituent. This involves the condensation of the amine group on the piperidine ring with a substituted aldehyde to form an imine intermediate, which is subsequently reduced in situ. The use of sodium triacetoxyborohydride as the reducing agent is a key mechanistic feature, as it allows the reaction to proceed efficiently at room temperature in solvents like acetic acid or dichloromethane. This reagent is mild enough to avoid reducing other functional groups that might be present on the aromatic rings, such as esters or nitro groups, thereby expanding the scope of compatible substrates. 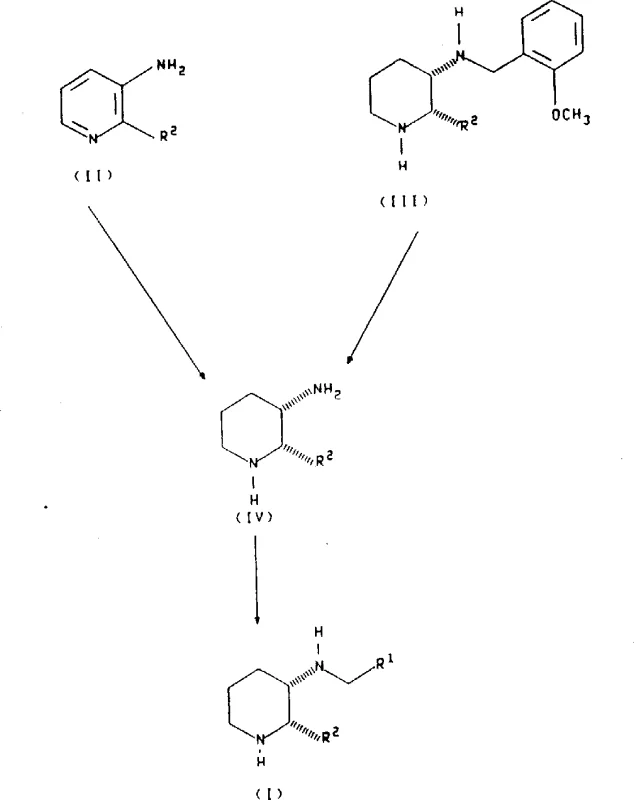
How to Synthesize Substituted Piperidine Efficiently
The synthesis of these high-value pharmaceutical intermediates requires precise control over reaction parameters to ensure consistent quality and yield. The process begins with the selection of appropriate starting pyridine derivatives, which are subjected to catalytic hydrogenation to generate the key piperidine scaffold. Following isolation or in-situ processing, the piperidine intermediate is reacted with a specific aldehyde component in the presence of a mild reducing agent. Detailed standard operating procedures regarding temperature control, pressure management, and work-up protocols are essential for replicating the stereoselectivity described in the patent. For a comprehensive guide on the specific stoichiometry, solvent choices, and purification techniques required to implement this route effectively, please refer to the standardized synthesis steps provided below.
- Reduce pyridine derivatives to piperidine intermediates using catalytic hydrogenation.
- React the piperidine intermediate with substituted aldehydes to form imines.
- Perform reductive amination using sodium triacetoxyborohydride to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this stereoselective synthesis route offers substantial benefits for procurement and supply chain management within the pharmaceutical sector. The reliance on commercially available starting materials, as explicitly noted in the patent data, significantly mitigates the risk of raw material shortages that often plague complex synthetic routes. This availability ensures a more resilient supply chain, allowing manufacturers to source precursors from multiple vendors without compromising on quality or specification. Furthermore, the use of standard catalytic hydrogenation equipment means that the process can be implemented in existing manufacturing facilities without the need for capital-intensive retrofitting or specialized reactor designs. This compatibility with standard infrastructure accelerates the timeline from process development to commercial production, enabling faster time-to-market for new drug candidates.
- Cost Reduction in Manufacturing: The elimination of complex chiral resolution steps and the use of efficient catalytic hydrogenation significantly lower the overall cost of manufacturing. By avoiding the waste associated with racemic synthesis and resolution, the process improves atom economy and reduces the consumption of expensive reagents. The mild reaction conditions also contribute to lower energy consumption, as there is no need for extreme heating or cooling protocols. Additionally, the ability to isolate products as stable salts simplifies downstream processing and packaging, further driving down operational expenses. These cumulative efficiencies translate into a more competitive cost structure for the final pharmaceutical intermediate, providing a distinct economic advantage in a price-sensitive market.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain reliability by reducing dependency on single-source specialty reagents. The catalysts used, such as palladium on carbon, are widely available and can be recovered and recycled in many industrial settings, further securing the supply of critical materials. The process tolerance to various substituents allows for flexibility in sourcing aldehyde precursors, meaning that if one supplier faces disruption, alternatives can be qualified with minimal process re-validation. This flexibility is crucial for maintaining continuous production schedules and meeting the rigorous delivery commitments required by global pharmaceutical clients. It ensures that the supply of these critical intermediates remains uninterrupted even in the face of market volatility.
- Scalability and Environmental Compliance: Scalability is a inherent strength of this methodology, as catalytic hydrogenation and reductive amination are well-established unit operations in the fine chemical industry. The process generates minimal hazardous waste compared to stoichiometric reduction methods, aligning with increasingly stringent environmental regulations. The use of common solvents like alcohols and acetic acid simplifies waste treatment and solvent recovery systems. Moreover, the high selectivity of the reaction reduces the formation of by-products, minimizing the burden on purification systems and reducing the overall environmental footprint of the manufacturing process. This compliance with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturing organization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the stereoselective preparation of substituted piperidine derivatives. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. They are intended to clarify the feasibility, scope, and advantages of this synthetic route for potential partners and stakeholders. Understanding these details is essential for evaluating the integration of this technology into existing supply chains and development pipelines.
Q: What is the primary advantage of this stereoselective method?
A: The method ensures high stereochemical purity by maintaining the absolute configuration of the piperidine ring during transformation, crucial for Substance P receptor antagonist activity.
Q: Are the starting materials commercially available?
A: Yes, the patent specifies that many starting aldehydes and pyridine derivatives are commercially available or easily synthesized from common precursors.
Q: What catalysts are preferred for the hydrogenation step?
A: Platinum on carbon and palladium on carbon are the preferred metal-containing catalysts for the selective reduction of the pyridine ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Piperidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity pharmaceutical intermediates in the development of next-generation therapeutics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the stereoselective methods described in CN1038932C can be translated from the laboratory to the plant floor with precision. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of substituted piperidine derivatives meets the exacting standards required for clinical and commercial applications. Our commitment to technical excellence ensures that your supply chain is supported by a partner who understands the nuances of complex heterocyclic chemistry.
We invite you to collaborate with us to optimize your supply chain for these vital neuroactive intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate our capability to deliver consistent quality at scale. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply of high-purity substituted piperidines, backed by a commitment to innovation and operational excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
