Advanced Manufacturing Strategies for Novel Antidiabetic Pharmaceutical Intermediates
The pharmaceutical landscape for metabolic disorder treatments continues to evolve, driven by the demand for more efficient manufacturing processes of active pharmaceutical ingredients (APIs) and their precursors. Patent CN1323300A represents a significant technological breakthrough in the synthesis of novel antidiabetic compounds having the structural formula (1). This intellectual property outlines a robust, convergent synthetic route that addresses critical bottlenecks found in earlier methodologies, specifically focusing on stereochemical control and process efficiency. The core innovation lies in the strategic timing of chiral resolution, shifting from late-stage purification to intermediate-level optimization. This shift is paramount for industrial applications where yield retention and cost management are critical decision factors for procurement and R&D leadership. By establishing a pathway that minimizes the degradation of optical purity during subsequent chemical transformations, this method ensures a consistent supply of high-quality intermediates essential for downstream drug formulation.
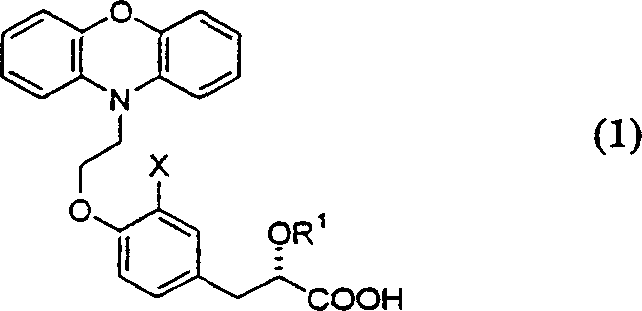
For global supply chain directors, the implications of this patent extend beyond mere chemical novelty; it signals a move towards more sustainable and economically viable production models. The ability to produce complex chiral molecules with reduced step counts and improved overall throughput directly correlates to reduced lead times and enhanced supply security. As the industry faces increasing pressure to lower the cost of goods sold (COGS) while maintaining stringent regulatory standards, adopting such improved synthetic methodologies becomes a competitive necessity. This report analyzes the technical nuances of this invention, providing actionable insights for stakeholders involved in the sourcing and manufacturing of high-purity pharmaceutical intermediates. Understanding these process improvements allows partners to make informed decisions regarding technology transfer and long-term supply agreements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those disclosed in US Patent Application No. 09/012585, typically relied on a linear synthesis where chiral resolution was performed at the very end of the process. As illustrated in Scheme-1 of the background data, this approach involves converting an aldehyde precursor through multiple steps including reduction, hydrolysis, and amide formation before attempting to isolate the desired enantiomer. The fundamental flaw in this strategy is the accumulation of impurities and the inevitable loss of valuable chiral material during the final resolution step. When resolution occurs late, any racemization that happens during previous synthetic steps results in a maximum theoretical yield loss of 50% at the most expensive stage of production. Furthermore, handling large quantities of racemic mixtures through complex multi-step sequences increases solvent consumption, waste generation, and operational hazards, making the process less attractive for large-scale commercial manufacturing.
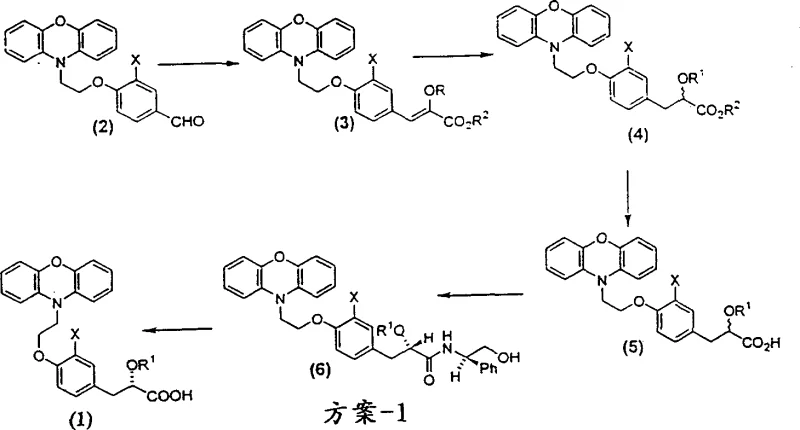
The Novel Approach
In stark contrast, the improved method detailed in CN1323300A introduces a paradigm shift by implementing chiral resolution at an earlier intermediate stage, specifically targeting compounds of formula (10) or (11). By resolving the stereochemistry before introducing the complex phenoxazine moiety, the process ensures that only the desired enantiomer proceeds through the remaining synthetic steps. This convergent strategy significantly mitigates the risk of carrying impurities forward and reduces the burden on final purification processes. The patent highlights that this modification not only simplifies the operational workflow but also dramatically improves the overall economic profile of the synthesis. The ability to recycle the unwanted enantiomer from the mother liquor back into the racemic pool for re-processing further enhances atom economy. This approach transforms the manufacturing landscape from a wasteful linear process into a efficient, circular system that maximizes resource utilization and minimizes environmental impact.
Mechanistic Insights into Convergent Chiral Synthesis
The core of this improved synthesis relies on a sophisticated understanding of stereoselective reactions and protecting group chemistry. The process initiates with the Darzens condensation of a protected p-hydroxybenzaldehyde with an alkyl haloacetate in the presence of a strong base. This reaction generates a glycidic ester, which serves as a versatile scaffold for constructing the chiral center. Subsequent ring-opening of the epoxide functionality, often achieved via catalytic hydrogenation using reagents like Raney Nickel or Pd/C, yields a racemic hydroxy ester. The critical mechanistic advantage arises in the subsequent resolution step, where chiral amines such as R(+)-α-methylbenzylamine or S(+)-phenylglycinol are employed to form diastereomeric salts. These salts exhibit distinct solubility profiles, allowing for the physical separation of the desired (S)-enantiomer with high optical purity. This early establishment of chirality is crucial because it prevents the propagation of stereochemical errors through the subsequent alkylation and etherification steps.
Furthermore, the patent describes an alternative chiral pool strategy utilizing L-Tyrosine as a starting material, as depicted in Scheme-6. This route leverages the inherent chirality of the natural amino acid, effectively bypassing the need for synthetic resolution in certain variations of the process. The transformation involves selective benzylation of the phenolic hydroxyl group followed by diazotization to convert the amino group into a hydroxyl functionality with retention or inversion of configuration depending on specific conditions. This mechanistic pathway underscores the versatility of the invention, offering manufacturers multiple entry points based on raw material availability and cost considerations. The final coupling with phenoxazine mesylate is executed under mild basic conditions, ensuring the integrity of the established chiral center is maintained throughout the final bond formation. Such mechanistic robustness is essential for ensuring batch-to-batch consistency in a GMP manufacturing environment.
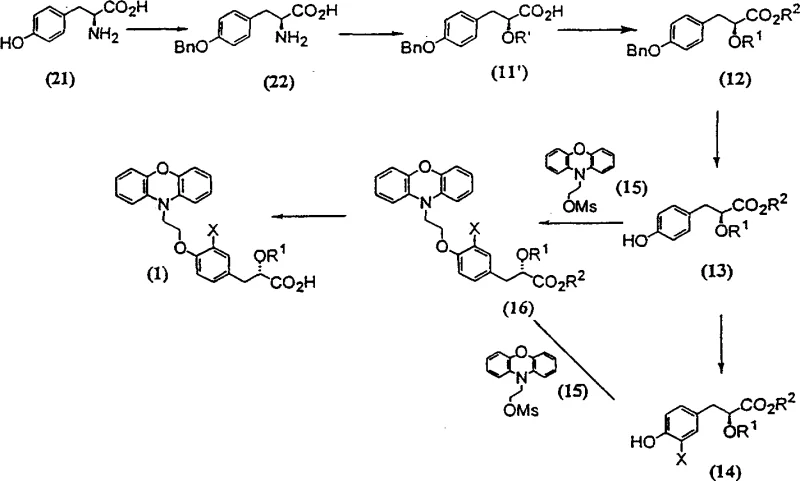
How to Synthesize Antidiabetic Intermediate Efficiently
Implementing this improved synthesis requires precise control over reaction parameters and a deep understanding of the kinetic profiles of each step. The process is designed to be scalable, moving seamlessly from laboratory benchtop to pilot plant and full commercial production. Key operational phases include the initial protection of the phenol, the stereoselective construction of the propionic acid side chain, and the final assembly of the phenoxyacetic acid core. Operators must pay close attention to temperature controls during the Darzens condensation and the exothermic alkylation steps to prevent side reactions. The resolution step demands careful selection of the chiral resolving agent and solvent system to maximize the recovery of the desired isomer. Detailed standard operating procedures (SOPs) are essential to maintain the high purity specifications required for pharmaceutical intermediates. For a comprehensive guide on the specific reaction conditions and workup procedures, please refer to the standardized synthesis protocol below.
- Perform Darzens condensation on protected p-hydroxybenzaldehyde to form glycidic esters, followed by epoxide ring opening.
- Execute chiral resolution at the hydroxy acid intermediate stage using chiral amines to ensure high optical purity before final coupling.
- Couple the resolved chiral phenol intermediate with phenoxazine mesylate and perform final hydrolysis to obtain the target carboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this improved synthesis method offers substantial strategic benefits for procurement managers and supply chain heads. The primary driver for cost reduction lies in the significant improvement in overall yield and the reduction of processing time. By resolving intermediates early, the process avoids the massive material losses associated with late-stage purification of complex molecules. This efficiency translates directly into lower raw material consumption per kilogram of finished product, effectively reducing the variable costs of manufacturing. Additionally, the simplified workflow reduces the demand for specialized equipment and extensive purification columns, lowering capital expenditure requirements for production facilities. The ability to recycle mother liquors further enhances the economic viability, creating a more sustainable cost structure that can withstand market fluctuations in raw material pricing.
- Cost Reduction in Manufacturing: The elimination of late-stage resolution steps removes a major bottleneck that traditionally drives up processing costs. By focusing purification efforts on simpler intermediates, the process reduces solvent usage and energy consumption associated with distillation and crystallization. This streamlined approach minimizes waste disposal costs and lowers the overall environmental footprint of the manufacturing operation. Consequently, suppliers can offer more competitive pricing structures without compromising on quality margins, providing a distinct advantage in tender negotiations for long-term supply contracts.
- Enhanced Supply Chain Reliability: The versatility of the synthetic route, which allows for starting materials like p-hydroxybenzaldehyde or L-Tyrosine, diversifies the supply base and reduces dependency on single-source vendors. This flexibility ensures continuity of supply even when specific raw materials face market shortages. Furthermore, the robustness of the chemical steps means fewer batch failures and less variability in production timelines. For supply chain planners, this predictability is invaluable, allowing for more accurate inventory management and reduced safety stock levels, ultimately freeing up working capital.
- Scalability and Environmental Compliance: The process is explicitly designed for commercial scale-up, utilizing standard chemical engineering unit operations that are well-understood and easily regulated. The reduction in hazardous reagents and the minimization of waste streams align with increasingly strict global environmental regulations. This compliance reduces the risk of regulatory shutdowns and simplifies the permitting process for new manufacturing lines. As the industry moves towards greener chemistry, adopting such efficient and waste-minimizing processes positions companies as leaders in sustainable pharmaceutical manufacturing, enhancing brand reputation and stakeholder value.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the technical specifications and advantageous effects described in the patent documentation. They serve to clarify the operational feasibility and strategic value of adopting this improved method for the production of antidiabetic intermediates. Understanding these details helps stakeholders assess the fit of this technology within their existing manufacturing portfolios and supply chain networks.
Q: What is the primary advantage of the improved synthesis method in CN1323300A?
A: The primary advantage is the implementation of early-stage chiral resolution. Unlike conventional methods that resolve the final product, this process resolves intermediates (Formula 10/11), significantly reducing material loss and improving overall yield to approximately 25%.
Q: Can this process be scaled for commercial production of antidiabetic intermediates?
A: Yes, the patent explicitly describes the method as commercially viable and safe for scaled-up reactions. It utilizes standard unit operations like hydrogenation, alkylation, and crystallization, which are readily adaptable to large-scale manufacturing environments.
Q: How does the chiral pool approach using L-Tyrosine impact the synthesis?
A: Utilizing L-Tyrosine as a starting material provides a natural chiral source, potentially bypassing the need for synthetic resolution steps in certain pathways. This enhances supply chain reliability by leveraging abundant natural amino acids.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Antidiabetic Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and reliable synthesis routes in the modern pharmaceutical supply chain. Our team of expert chemists and engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to market. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging advanced technologies like the convergent synthesis described in CN1323300A, we optimize our manufacturing processes to provide cost-effective solutions without compromising on quality or delivery performance.
We invite you to collaborate with us to explore how these technical advancements can benefit your specific drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality needs. Please contact us today to request specific COA data and route feasibility assessments for your next project. Let us be your partner in navigating the complexities of fine chemical manufacturing and securing a stable supply of critical antidiabetic intermediates for your global operations.
