Advanced Manufacturing of Brivaracetam Intermediates via Optimized Chiral Auxiliary Routes for Global Supply Chains
Advanced Manufacturing of Brivaracetam Intermediates via Optimized Chiral Auxiliary Routes for Global Supply Chains
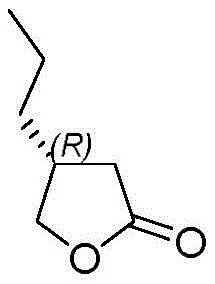
The pharmaceutical landscape for antiepileptic drugs continues to evolve, with Brivaracetam standing out as a third-generation therapy offering superior tolerability and efficacy for partial onset seizures. At the heart of manufacturing this critical active pharmaceutical ingredient lies the challenge of producing its key chiral building block, (R)-3-propyl-γ-butyrolactone, with high optical purity and economic efficiency. Patent CN113336726B, published in late 2022, introduces a transformative synthetic methodology that addresses long-standing bottlenecks in the production of this vital pharmaceutical intermediate. This technical insight report analyzes the patented route, which leverages the stereochemical control of Evans-type chiral auxiliaries to achieve robust asymmetric induction without relying on expensive biocatalysts or toxic heavy metals.
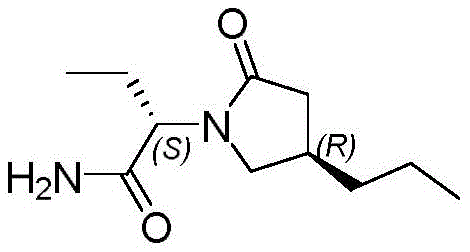
The strategic importance of this intermediate cannot be overstated, as it constitutes the chiral core of the final drug molecule. Traditional methods have often struggled with balancing cost, safety, and stereoselectivity, leading to supply chain vulnerabilities for global pharmaceutical manufacturers. The new approach detailed in the patent utilizes methyl 4-chloro-4-oxobutyrate as a readily accessible starting material, coupling it with (R)-4-benzyl-2-oxazolidinone to set the stage for highly selective alkylation. This shift represents a significant move towards more predictable and scalable chemistry, providing a reliable foundation for the commercial scale-up of complex pharmaceutical intermediates required for next-generation neurological treatments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (R)-3-propyl-γ-butyrolactone has been plagued by significant technical and economic hurdles that hinder efficient industrial production. One prevalent prior art method relies on the use of (R)-epichlorohydrin reacting with malonate esters, followed by Grignard addition and decarboxylation; however, this pathway is characterized by harsh reaction conditions and notoriously low overall yields that erode profit margins. Another common approach involves enzymatic kinetic resolution, which, while green in theory, suffers from the high cost of specialized enzymes, narrow substrate tolerance, and the inherent 50% maximum theoretical yield limitation of resolution processes that discard half the material. Furthermore, alternative routes utilizing transition metal catalysts such as rhodium complexes often require toxic solvents like benzene and involve cumbersome purification steps to remove trace heavy metals to meet stringent regulatory limits for API manufacturing.
The Novel Approach
In stark contrast to these legacy technologies, the novel methodology described in patent CN113336726B offers a streamlined, four-step sequence that maximizes atom economy and operational safety. By employing a chiral auxiliary strategy, the process bypasses the need for expensive chiral columns or difficult enzymatic separations, instead relying on well-understood stoichiometric organic transformations. The route begins with the formation of a chiral imide, which acts as a rigid template to direct the subsequent introduction of the propyl group with high diastereoselectivity. This approach not only simplifies the purification workflow but also utilizes commodity chemicals that are stable and easy to handle, effectively mitigating the supply chain risks associated with sourcing specialized biocatalysts or precious metal catalysts.
Mechanistic Insights into Chiral Auxiliary-Mediated Asymmetric Alkylation
The core innovation of this synthesis lies in the precise stereochemical control exerted by the (R)-4-benzyl-2-oxazolidinone moiety during the alkylation step. When the chiral imide intermediate is treated with a strong base such as n-butyllithium at cryogenic temperatures, it forms a rigid enolate species where the benzyl group of the auxiliary shields one face of the molecule. This steric hindrance forces the incoming electrophile, 1-halopropane, to attack exclusively from the less hindered face, thereby establishing the desired (R)-configuration at the alpha-carbon with exceptional fidelity. The robustness of this mechanistic pathway ensures that the optical purity is locked in early in the synthesis, preventing the erosion of enantiomeric excess in downstream processing steps which is a common failure mode in less controlled catalytic systems.
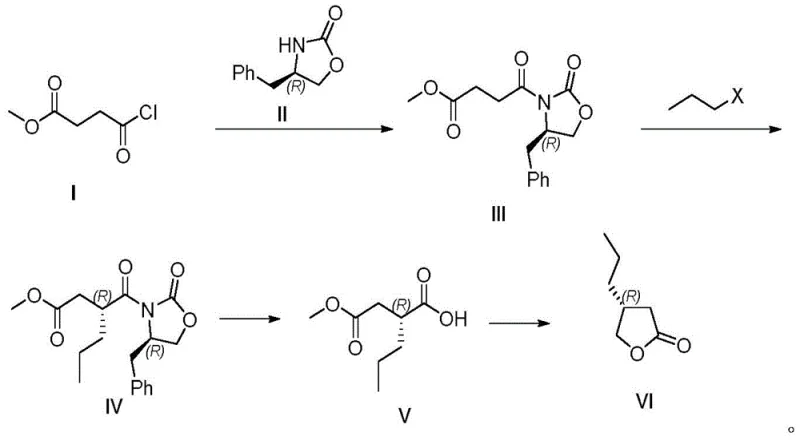
Following the establishment of chirality, the removal of the auxiliary and subsequent cyclization are engineered to preserve the stereochemical integrity while minimizing waste. The cleavage step utilizes a mild oxidative hydrolysis system comprising lithium hydroxide and hydrogen peroxide, which cleanly releases the chiral carboxylic acid without racemization. The final transformation involves a mixed anhydride formation followed by reduction with sodium borohydride and acid-catalyzed lactonization. This cascade effectively converts the linear acid into the cyclic lactone in a single pot operation, reducing solvent usage and processing time. The meticulous control over reaction parameters, such as maintaining temperatures between -78°C and 0°C during critical steps, ensures that side reactions are suppressed, leading to a high-purity product suitable for direct use in API synthesis.
How to Synthesize (R)-3-propyl-γ-butyrolactone Efficiently
The execution of this synthetic route requires careful attention to temperature control and reagent stoichiometry to maximize yield and selectivity. The process is designed to be telescoped where possible, reducing the number of isolation steps and improving overall throughput. Operators must ensure strict anhydrous conditions during the enolate formation and alkylation phases to prevent protonation of the reactive intermediate. The following guide outlines the standardized operational protocol derived from the patent examples, serving as a baseline for process optimization and technology transfer activities within a GMP environment.
- Couple methyl 4-chloro-4-oxobutyrate with (R)-4-benzyl-2-oxazolidinone using an organic base to form the imide intermediate.
- Perform asymmetric alkylation using 1-halopropane and a strong base like n-butyllithium at low temperatures to establish the R-configuration.
- Cleave the chiral auxiliary using lithium hydroxide and hydrogen peroxide to yield the chiral carboxylic acid.
- Reduce the carboxylic acid derivative with sodium borohydride followed by acid-catalyzed cyclization to form the final lactone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this chiral auxiliary-based route presents a compelling value proposition centered on cost stability and operational reliability. Unlike enzymatic routes that depend on the fluctuating availability and pricing of biocatalysts, this method relies on standard organic reagents that are sourced from a broad global supplier base, insulating the manufacturing process from single-source bottlenecks. The elimination of precious metal catalysts such as rhodium or copper not only reduces the direct material cost but also removes the substantial downstream expense associated with metal scavenging and validation testing, which are mandatory for pharmaceutical compliance. Furthermore, the avoidance of toxic solvents like benzene simplifies environmental health and safety protocols, reducing the overhead costs related to waste disposal and worker protection.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven by the use of inexpensive starting materials like methyl 4-chloro-4-oxobutyrate and the high efficiency of the reaction steps which minimize material loss. By achieving high yields in the alkylation and cyclization steps, the process significantly reduces the cost of goods sold compared to low-yielding resolution methods that discard half the starting material. Additionally, the ability to recover and potentially recycle the chiral auxiliary further enhances the cost-effectiveness of the campaign, making it a financially sustainable option for long-term production.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved as the synthesis does not rely on specialized biological agents or rare earth metals that are prone to geopolitical supply disruptions. The reagents used, including standard halides and borohydrides, are commodity chemicals with established logistics networks, ensuring consistent lead times and availability. This stability allows for more accurate production planning and inventory management, reducing the risk of stockouts that could delay the manufacture of the final antiepileptic medication.
- Scalability and Environmental Compliance: From an environmental perspective, the process generates less hazardous waste compared to heavy metal-catalyzed alternatives, facilitating easier compliance with increasingly strict environmental regulations. The reaction conditions are amenable to scale-up in standard stainless steel reactors without requiring exotic equipment for high-pressure hydrogenation or cryogenic enzymatic fermentation. This operational simplicity translates to faster technology transfer from lab to plant and a shorter timeline to reach commercial production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications of adopting this route for industrial manufacturing. Understanding these nuances is critical for R&D teams evaluating process feasibility and procurement teams assessing vendor capabilities.
Q: What are the advantages of using chiral auxiliaries over enzymatic resolution for this intermediate?
A: Chiral auxiliary routes avoid the high costs and stability issues associated with specialized enzymes, offering more robust reaction conditions and easier scalability for industrial manufacturing.
Q: How does this method improve impurity control compared to metal-catalyzed routes?
A: By eliminating transition metal catalysts like rhodium or copper, this method removes the need for complex heavy metal scavenging steps, resulting in a cleaner impurity profile and simpler purification.
Q: Is the starting material methyl 4-chloro-4-oxobutyrate commercially available?
A: Yes, methyl 4-chloro-4-oxobutyrate is a commodity chemical that is readily available from bulk suppliers, ensuring a stable and cost-effective supply chain for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-3-propyl-γ-butyrolactone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced therapeutics like Brivaracetam depends on a robust and high-quality supply of critical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (R)-3-propyl-γ-butyrolactone meets the exacting standards required for global pharmaceutical registration, providing our partners with the confidence needed to navigate complex regulatory landscapes.
We invite you to engage with our technical procurement team to discuss how this optimized synthetic route can be integrated into your supply chain to drive efficiency and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic impact of switching to this methodology. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your specific volume requirements and quality targets.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
