Advanced Manufacturing of 1,5-Diazido-3-Nitroazapentane for High-Energy Propellant Applications
Introduction to Next-Generation Energetic Plasticizer Synthesis
The development of high-performance energetic materials is critical for advancing propulsion technologies, particularly in the realm of solid and liquid propellants where burning rate and flame temperature control are paramount. Patent CN115385822A introduces a transformative synthetic methodology for producing 1,5-diazido-3-nitroazapentane (DIANP), a potent azidonitroamine energetic plasticizer known for its ability to enhance burning rates while reducing flame temperatures and molecular weight of combustion products. This innovation addresses long-standing inefficiencies in the manufacturing of energetic plasticizers by replacing hazardous and low-yielding reagents with a robust magnesium oxide-buffered nitration system. By shifting away from traditional acetic anhydride-based nitration, this process not only mitigates safety risks associated with volatile anhydrides but also significantly elevates the purity and yield of the critical nitro-intermediate. For R&D directors and procurement specialists in the energetic materials sector, this patent represents a pivotal shift towards more sustainable and economically viable production protocols for high-energy density compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
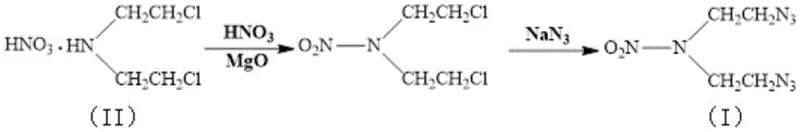
Historically, the synthesis of DIANP has relied heavily on routes that utilize bis(2-chloroethyl)amine hydrochloride as the primary feedstock, necessitating a nitration step mediated by a nitric acid and acetic anhydride system. As illustrated in prior art such as Chinese patent 201410457712.6, this conventional approach suffers from inherent thermodynamic and kinetic limitations that cap the reaction yield at approximately 66% to 67% for the nitro-intermediate stage. Furthermore, the reliance on acetic anhydride introduces significant handling hazards and generates substantial quantities of acidic waste streams that require complex neutralization and disposal procedures, thereby inflating operational expenditures. The subsequent azidation step in these legacy processes often employs phase transfer catalysts in aqueous solutions, which, while functional, typically achieve yields ranging only from 74% to 80% with product purities hovering around 97.5%. These cumulative inefficiencies result in a compounded loss of material throughput and increased environmental burden, making the conventional route less attractive for large-scale commercial manufacturing where margin compression is a constant concern.
The Novel Approach
In stark contrast to the legacy methodologies, the novel approach detailed in the patent utilizes bis(2-chloroethyl)amine nitrate directly as the starting material, bypassing the need for in-situ salt formation and leveraging a magnesium oxide-buffered nitration environment. This strategic modification allows for precise control over the acidity of the reaction medium, preventing the degradation of the sensitive amine backbone that often plagues strong acid nitrations. The process operates under mild thermal conditions, maintaining temperatures between 40-55°C during the nitration phase, which drastically reduces the formation of oxidative by-products. Following the isolation of the high-purity intermediate, the subsequent azidation is conducted in a specialized biphasic solvent system comprising 2-heptanone and water, eliminating the need for expensive phase transfer catalysts. This streamlined workflow not only simplifies the downstream processing requirements but also drives the overall yield of the final DIANP product to impressive levels exceeding 88%, representing a substantial leap forward in process efficiency and material conservation for energetic material manufacturers.
Mechanistic Insights into MgO-Buffered Nitration and Biphasic Azidation
The core chemical innovation of this synthesis lies in the utilization of magnesium oxide as a heterogeneous base within the concentrated nitric acid matrix, creating a buffered nitration system that moderates the reactivity of the nitrating species. Mechanistically, the magnesium oxide reacts with the nitric acid to form magnesium nitrate in situ, which serves to consume excess protons and stabilize the pH environment around the amine substrate. This buffering action is crucial because it prevents the over-protonation of the amine nitrogen, which would otherwise deactivate the molecule towards electrophilic attack by the nitronium ion, while simultaneously protecting the chloroethyl side chains from acid-catalyzed hydrolysis or elimination reactions. The result is a highly selective transformation where the nitro group is installed exclusively at the central nitrogen atom with minimal collateral damage to the molecular scaffold, explaining the observed jump in intermediate purity to over 98%. This level of selectivity is rarely achieved in unbuffered strong acid systems, where competitive degradation pathways often dominate.
Following the nitration, the azidation mechanism benefits significantly from the unique solvation properties of the 2-heptanone and water mixture. In this biphasic system, the organic intermediate exhibits high solubility in the 2-heptanone phase, while the sodium azide remains dissolved in the aqueous phase. The reaction proceeds at the interface or through limited mass transfer of the azide anion into the organic phase, facilitated by the elevated reaction temperature of 65-85°C. This setup effectively mimics the function of a phase transfer catalyst without requiring the addition of quaternary ammonium salts, which can be difficult to remove from the final product and may act as contaminants in sensitive energetic formulations. The thermal energy provided ensures sufficient collision frequency between the nucleophilic azide and the electrophilic chloroethyl carbons, driving the substitution reaction to near-completion and ensuring that the residual chloride content in the final API or intermediate is minimized, a critical parameter for meeting stringent military and aerospace specifications.
How to Synthesize 1,5-Diazido-3-Nitroazapentane Efficiently
Implementing this synthesis protocol requires careful attention to stoichiometry and thermal management to replicate the high yields reported in the patent literature. The process begins with the preparation of the nitrating mixture, where magnesium oxide is dispersed into concentrated nitric acid prior to the addition of the amine salt, ensuring the buffer capacity is established before the exothermic nitration begins. Operators must maintain strict temperature control during the addition phase, keeping the reactor between 20-30°C to prevent runaway reactions, followed by a controlled ramp to 40-55°C to drive the conversion to completion. Once the intermediate is isolated via standard extraction and distillation techniques, the second stage involves mixing the nitro-intermediate with sodium azide in the specific 2-heptanone/water ratio, followed by a prolonged heating period to ensure full conversion. For a comprehensive understanding of the precise operational parameters and safety protocols required for scale-up, please refer to the standardized synthesis guide below.
- Prepare the nitration system by dissolving magnesium oxide in concentrated nitric acid, then slowly add bis(2-chloroethyl)amine nitrate at 20-30°C and react at 40-55°C to form the nitro-intermediate.
- Isolate the 1,5-dichloroethyl-3-nitroazapentane intermediate via ice-water quenching and dichloromethane extraction.
- Perform the azidation reaction by heating the intermediate with sodium azide in a 2-heptanone and water solvent system at 65-85°C to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this MgO-buffered synthesis route offers profound advantages for supply chain managers and procurement officers tasked with optimizing the cost structure of energetic material production. The elimination of acetic anhydride, a reagent subject to price volatility and regulatory scrutiny due to its potential dual-use nature, simplifies the sourcing strategy and reduces the administrative burden associated with hazardous material compliance. Furthermore, the switch to commodity chemicals like magnesium oxide and 2-heptanone creates a more resilient supply chain that is less susceptible to disruptions in the specialty chemical market. The significant improvement in reaction yields translates directly into reduced raw material consumption per kilogram of finished product, effectively lowering the variable cost of goods sold without compromising on quality. This efficiency gain is particularly valuable in the high-volume manufacturing of propellant additives, where even marginal improvements in yield can result in substantial annual savings.
- Cost Reduction in Manufacturing: The transition to this novel synthetic route eliminates the need for expensive phase transfer catalysts and acetic anhydride, replacing them with cost-effective and readily available alternatives like magnesium oxide and 2-heptanone. By removing these high-cost reagents, the direct material cost of the synthesis is significantly reduced, while the simplified workup procedure decreases the consumption of solvents and utilities required for purification. Additionally, the higher reaction yields mean that less starting material is wasted, maximizing the return on investment for every batch produced and allowing for more competitive pricing strategies in the global energetic materials market.
- Enhanced Supply Chain Reliability: Sourcing magnesium oxide and 2-heptanone is inherently more stable than relying on acetic anhydride, which often faces supply constraints due to its widespread use in other industries and strict regulatory controls. By diversifying the reagent portfolio towards bulk commodities, manufacturers can mitigate the risk of production stoppages caused by raw material shortages. The robustness of the reaction conditions also implies a wider operating window, reducing the likelihood of batch failures due to minor process deviations, thereby ensuring a consistent and reliable flow of finished product to meet demanding delivery schedules for defense and aerospace contracts.
- Scalability and Environmental Compliance: The process design inherently supports scalability, as the exothermic nature of the nitration is effectively managed by the magnesium oxide buffer, reducing the cooling load requirements on large-scale reactors. From an environmental standpoint, the reduction in acidic waste streams and the avoidance of hazardous anhydrides simplify the effluent treatment process, lowering the costs associated with waste disposal and environmental compliance. This greener profile not only aligns with modern sustainability goals but also facilitates easier permitting for facility expansions, enabling manufacturers to rapidly scale production from pilot plants to multi-ton commercial facilities without encountering significant regulatory hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis method, derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is essential for process engineers looking to adapt this technology for their own manufacturing lines, as it clarifies the specific roles of key reagents and the expected performance metrics. These insights help bridge the gap between laboratory-scale success and industrial-scale viability, ensuring that stakeholders have a clear picture of the operational benefits.
Q: Why is Magnesium Oxide used instead of Acetic Anhydride in this nitration process?
A: Magnesium Oxide acts as a buffering agent that neutralizes excess acidity generated during nitration without introducing water prematurely. This contrasts with the conventional Acetic Anhydride method, which often results in lower yields (66-67%) and generates significant acidic waste. The MgO system stabilizes the reaction environment, leading to higher purity intermediates (98.2-98.5%) and improved overall safety by avoiding hazardous anhydride reagents.
Q: What are the advantages of using a 2-heptanone and water solvent system for azidation?
A: The biphasic solvent system comprising 2-heptanone and water facilitates efficient phase transfer of the azide ion while maintaining a controlled reaction temperature between 65-85°C. This specific solvent combination enhances the solubility of the organic intermediate while allowing the inorganic sodium azide to react effectively at the interface or within the aqueous phase, resulting in superior conversion rates (88.7-90% yield) compared to traditional phase transfer catalyst methods.
Q: How does this synthesis method impact the purity profile of the final energetic plasticizer?
A: The optimized reaction conditions, specifically the controlled temperature ramping and the use of high-purity starting materials like bis(2-chloroethyl)amine nitrate, minimize the formation of side-products and oligomers. The patent data indicates a final product purity of 98.8-99.6%, which is critical for energetic applications where impurity profiles can affect the stability and burning rate of the final propellant formulation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Diazido-3-Nitroazapentane Supplier
As the demand for high-performance energetic plasticizers continues to grow, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthesis technologies and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, allowing us to seamlessly transition innovative patent methodologies like the MgO-buffered nitration route into reliable supply chains. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1,5-diazido-3-nitroazapentane meets the exacting standards required for propellant and pyrotechnic applications, providing our partners with the confidence needed for critical mission profiles.
We invite procurement leaders and technical directors to engage with our Customized Cost-Saving Analysis service, where we evaluate your current supply chain against this optimized synthetic route to quantify potential efficiencies. Please contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your volume requirements, and let us demonstrate how our advanced manufacturing expertise can drive value and reliability in your energetic materials portfolio.
