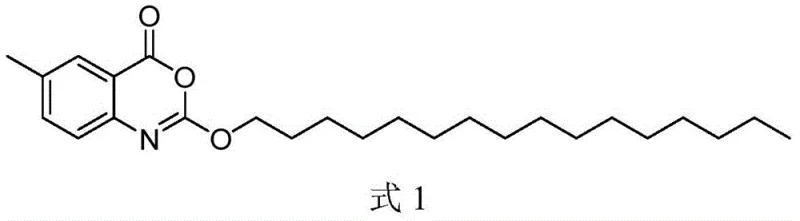
Advanced Manufacturing of Cetilistat via Novel Triphosgene Cyclization Route
Introduction to Advanced Cetilistat Manufacturing
The global demand for effective anti-obesity therapeutics has driven intense research into the scalable production of lipase inhibitors like Cetilistat. Patent CN110903259A introduces a groundbreaking methodology that fundamentally restructures the synthetic pathway for this critical pharmaceutical intermediate. By utilizing 2-amino-5-methylbenzoic acid as the primary starting material, this invention circumvents the historical bottlenecks of low yield and complex purification that have plagued earlier generations of synthesis technology. The core innovation lies in a strategic three-step sequence that leverages triphosgene-mediated cyclization and specific sulfonate activation, ensuring a robust and reproducible manufacturing protocol. This approach not only addresses the chemical inefficiencies of prior art but also aligns perfectly with modern green chemistry principles by operating under mild conditions without the need for hazardous high-pressure equipment or toxic transition metals.
For pharmaceutical developers and supply chain strategists, the implications of this patent are profound, offering a reliable Cetilistat supplier pathway that guarantees consistency and quality. The traditional reliance on one-pot reactions often resulted in tangled impurity profiles that were costly and time-consuming to resolve. In contrast, this novel process isolates key intermediates, allowing for rigorous quality control at each stage before proceeding to the final coupling. The result is a final product with purity exceeding 99 percent, a specification that is critical for regulatory approval in major markets. By shifting the paradigm from brute-force chemistry to precise, step-wise construction, this technology represents a significant leap forward in the industrial preparation of metabolic disease treatments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cetilistat has been fraught with significant technical hurdles that hindered its widespread commercial adoption. Early methods, such as those disclosed in CN1359378A, attempted to achieve the target molecule through a direct one-step reaction between 2-amino-5-methylbenzoic acid and n-hexadecyl chloroformate. While conceptually simple, this approach failed miserably in practice, requiring a massive excess of reagents to drive conversion, which inevitably led to a total yield of merely 15 percent. Furthermore, the reaction mixture was contaminated with substantial amounts of unreacted hexadecanol and other byproducts, making the subsequent separation and purification processes extremely difficult and economically unviable. Another prominent route, detailed in CN1785967A, relied on a palladium-catalyzed carbonylation step that necessitated high temperatures of 115 degrees Celsius and high pressures of 8 bar. This not only increased the capital expenditure for specialized equipment but also introduced the persistent risk of palladium residue, a critical failure point for API manufacturing.
The Novel Approach
The methodology presented in CN110903259A offers a decisive break from these inefficient traditions by decoupling the synthesis into three distinct, highly optimized stages. Instead of forcing a difficult one-pot cyclization, the new route first constructs the benzoxazine core independently using triphosgene, a safer and more manageable phosgene equivalent. This ensures that the heterocyclic ring is formed with high fidelity before the long alkyl chain is introduced. Subsequently, the long-chain alcohol is activated as a sulfonate ester rather than a reactive chloroformate, providing a stable yet reactive electrophile for the final coupling step. This strategic separation of concerns eliminates the cross-contamination issues seen in older methods and allows each intermediate to be purified to near-perfect standards. The result is a streamlined process that operates at atmospheric pressure and moderate temperatures, drastically reducing energy consumption and safety risks while boosting the overall yield to over 52 percent.
Mechanistic Insights into Triphosgene-Mediated Cyclization and Alkylation
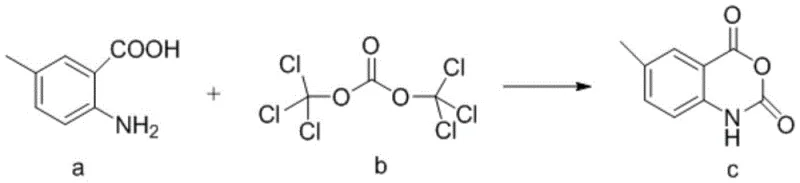
The heart of this synthetic breakthrough lies in the clever utilization of triphosgene to activate the carboxylic acid and amine functionalities of the starting material simultaneously. In the first critical step, 2-amino-5-methylbenzoic acid reacts with triphosgene in the presence of an organic base such as pyridine or triethylamine. This reaction generates an unstable acyl chloride intermediate in situ, which immediately undergoes intramolecular nucleophilic attack by the adjacent amino group. This cyclization event forms the rigid 6-methyl-2,4-dihydro-1H-3,1-benzoxazine-2,4-dione structure, known as intermediate c. The use of triphosgene is particularly advantageous because it releases phosgene equivalents in a controlled manner, minimizing the formation of polymeric side products and ensuring a clean conversion to the cyclic anhydride-like structure. This intermediate serves as the perfect nucleophilic partner for the subsequent alkylation, possessing the necessary electronic configuration to react selectively at the oxygen atom.
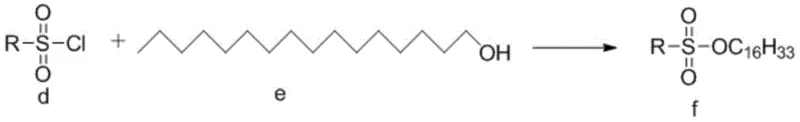
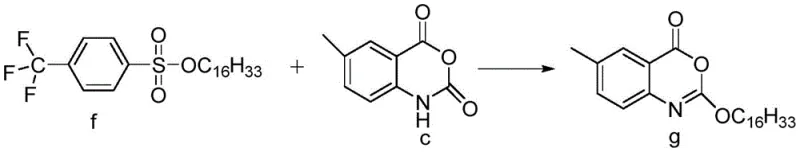
Parallel to the core formation, the long hydrophobic tail is prepared through a specialized sulfonation process. N-hexadecanol is reacted with a specific sulfonyl chloride, such as p-trifluoromethylbenzenesulfonyl chloride, to generate the sulfonate ester intermediate f. This activation strategy is superior to traditional chloroformate methods because sulfonates are excellent leaving groups that are more stable to hydrolysis during storage and handling. In the final convergence step, the nitrogen-containing heterocycle of intermediate c attacks the electrophilic carbon of the sulfonate chain in intermediate f. This SN2-type alkylation occurs smoothly under mild basic conditions, displacing the sulfonate group and forging the critical ether linkage that defines the Cetilistat molecule. The entire sequence avoids the use of transition metals, thereby eliminating the need for expensive scavenging resins to remove trace catalysts from the final drug substance.
How to Synthesize Cetilistat Efficiently
The execution of this synthesis requires precise control over stoichiometry and temperature to maximize the yield of each discrete step. The process begins with the careful addition of triphosgene to a solution of the amino acid in acetonitrile, maintaining a temperature of roughly 52 degrees Celsius to facilitate cyclization without degradation. Following the isolation of the benzoxazine core, the focus shifts to the preparation of the sulfonate tail, where reflux conditions in dichloromethane ensure complete conversion of the long-chain alcohol. The final coupling is performed under ice-bath conditions initially to control exothermicity, followed by gentle heating to drive the reaction to completion. For a comprehensive guide on the specific molar ratios, solvent choices, and workup procedures detailed in the patent, please refer to the standardized synthesis steps provided below.
- Synthesize intermediate c (6-methyl-2,4-dihydro-1H-3,1-benzoxazine-2,4-dione) by reacting 2-amino-5-methylbenzoic acid with triphosgene under organic base catalysis.
- Prepare intermediate f (sulfonate) by reacting specific sulfonyl chloride with n-hexadecanol in the presence of a base.
- Perform the final alkylation reaction between intermediate c and intermediate f using an organic base to yield Cetilistat.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this novel synthetic route offers transformative benefits that directly impact the bottom line and operational reliability. The most significant advantage is the complete elimination of precious metal catalysts, specifically palladium, which was a requirement in previous high-yield attempts. Removing this dependency not only eradicates the cost associated with purchasing expensive catalysts but also removes the entire downstream unit operation dedicated to metal scavenging and validation. This simplification of the purification train leads to substantial cost savings in both materials and processing time, while simultaneously de-risking the supply chain from fluctuations in the price of rare earth metals. Furthermore, the avoidance of high-pressure reactors means that production can be scaled up using standard glass-lined steel vessels available in most multipurpose chemical plants, significantly lowering the barrier to entry for contract manufacturing organizations.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by its high atom economy and the use of commodity chemicals. By achieving a total yield that is significantly higher than legacy methods, the amount of raw material required per kilogram of final product is drastically reduced. Additionally, the mild reaction conditions operate at atmospheric pressure and moderate temperatures, which translates to lower energy consumption for heating and cooling utilities. The absence of complex high-pressure equipment maintenance and the removal of costly metal catalyst recovery steps further contribute to a leaner, more cost-effective manufacturing profile that enhances margin potential for bulk producers.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of robust, commercially available starting materials like 2-amino-5-methylbenzoic acid and n-hexadecanol. Unlike routes that depend on specialized or unstable reagents, the intermediates in this process, particularly the sulfonate ester, possess sufficient stability for safe storage and transport. This stability allows for a flexible production schedule where intermediates can be stockpiled during periods of low demand and rapidly converted to the final API when needed. The simplified purification process also reduces the lead time for batch release, enabling faster response to market demands and ensuring a steady flow of high-quality material to downstream formulation partners.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly lower than that of traditional methods, aligning with increasingly strict global regulations on pharmaceutical manufacturing. The process generates fewer hazardous byproducts and avoids the use of toxic heavy metals, simplifying waste treatment and disposal protocols. The high purity of the crude product reduces the volume of solvents required for recrystallization, minimizing volatile organic compound emissions. These factors combined make the process highly scalable from pilot plant to commercial tonnage without encountering the safety or environmental bottlenecks that often stall the expansion of more hazardous chemical pathways.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. They are derived from a detailed analysis of the experimental data and comparative examples provided in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this method for large-scale production.
Q: How does this new process improve upon traditional Cetilistat synthesis yields?
A: Traditional methods often suffer from low total yields around 15-27% due to side reactions and difficult purification. This novel route achieves a total yield exceeding 52% with individual step yields over 82%, significantly reducing raw material waste.
Q: Does this synthesis method involve heavy metal catalysts?
A: No, unlike previous routes that required expensive palladium catalysts under high pressure, this process is entirely metal-free. This eliminates the risk of heavy metal residue in the final API, simplifying quality control and regulatory compliance.
Q: What are the purity specifications achievable with this route?
A: The process is designed to minimize side products and impurities such as residual hexadecanol esters. Through optimized recrystallization steps, the final Cetilistat product consistently achieves a purity greater than 99%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cetilistat Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant manufacturing pathways for high-value pharmaceutical intermediates like Cetilistat. Our technical team has thoroughly analyzed the innovations within CN110903259A and integrated similar best practices into our own production capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel route are fully realized in a GMP-compliant environment. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the >99 percent purity standard required for global regulatory submissions.
We invite procurement leaders and R&D directors to collaborate with us to leverage this advanced technology for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this metal-free, high-yield process for your organization. We encourage you to contact our technical procurement team today to request specific COA data from our recent pilot runs and to discuss route feasibility assessments tailored to your volume requirements. Let us help you secure a sustainable and cost-effective supply of this vital anti-obesity intermediate.
