Advanced Synthesis of Cytidine Diphosphate: A Scalable Route for High-Purity Pharmaceutical Intermediates
Advanced Synthesis of Cytidine Diphosphate: A Scalable Route for High-Purity Pharmaceutical Intermediates
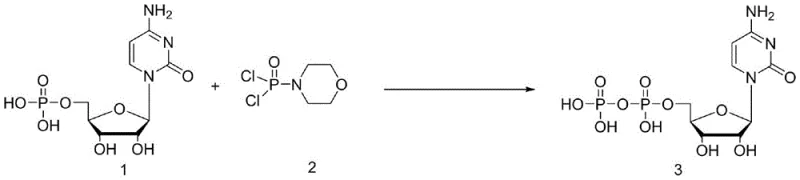
The landscape of nucleoside synthesis is undergoing a significant transformation driven by the urgent need for more efficient, cost-effective, and environmentally sustainable manufacturing processes. A pivotal development in this field is detailed in patent CN113999270B, which discloses a robust method for synthesizing Cytidine Diphosphate (CDP), a critical intermediate in the production of polyinosinic-polycytidylic acid and various antiviral therapeutics. This technology represents a departure from complex, multi-step biosynthetic or chemically intensive routes, offering a streamlined pathway that achieves high purity through direct condensation. For R&D directors and procurement strategists alike, understanding the nuances of this patented approach is essential for securing a competitive edge in the supply of high-value pharmaceutical intermediates. The method utilizes cytidylic acid and dichlorophosphoryl morpholine as key starting materials, reacting under mild alkali catalysis to form the diphosphate bond directly.
The implications of this technological breakthrough extend far beyond the laboratory bench, addressing critical pain points in the global supply chain for nucleotide derivatives. By reducing the synthetic sequence to merely two primary operational steps—condensation followed by hydrolysis and crystallization—the process minimizes material loss and significantly shortens production cycles. This efficiency is paramount for manufacturers aiming to meet the escalating demand for immunomodulators and antiviral agents without compromising on quality standards. Furthermore, the avoidance of hazardous coupling reagents and complex purification columns aligns perfectly with modern green chemistry principles, reducing the environmental footprint of production facilities. As we delve deeper into the technical specifics, it becomes clear that this methodology offers a viable solution for the commercial scale-up of complex nucleoside diphosphates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cytidine Diphosphate has been plagued by inefficiencies inherent in both biosynthetic and traditional chemical pathways. Biosynthetic methods, while theoretically elegant, often suffer from low titers and complex reaction mixtures where separating the target diphosphate from monophosphate precursors and other metabolic byproducts proves exceptionally difficult and costly. On the chemical synthesis front, conventional strategies frequently rely on the activation of the phosphate group using reagents like DCC (dicyclohexylcarbodiimide) or the preparation of activated nucleoside derivatives such as 5'-OTs (tosylates). These approaches introduce significant downstream processing challenges; for instance, the removal of dicyclohexylurea (DCU), a byproduct of DCC coupling, is notoriously difficult and can lead to persistent impurities in the final active pharmaceutical ingredient. Additionally, multi-step protection and deprotection sequences required in older chemical routes increase the overall cost of goods sold (COGS) and generate substantial chemical waste, rendering them less attractive for large-scale industrial application.
The Novel Approach
In stark contrast to these legacy methods, the novel approach outlined in the patent data utilizes a direct condensation strategy that dramatically simplifies the molecular construction of the diphosphate backbone. By employing dichlorophosphoryl morpholine as a phosphorylating agent, the reaction bypasses the need for pre-activating the nucleoside with leaving groups like tosylates or forming unstable morpholidates. This direct coupling, facilitated by a simple inorganic or organic base, allows for the formation of the phosphoanhydride bond in a single pot with high atom economy. The subsequent hydrolysis step cleaves the morpholine moiety cleanly, yielding the free acid form of the diphosphate. This streamlined workflow not only reduces the number of unit operations but also eliminates the need for ion-exchange chromatography, a bottleneck in many nucleotide purifications. Instead, the process relies on a sophisticated recrystallization protocol using alcohol-water mixtures to achieve purity levels exceeding 95%, demonstrating that simplicity in design can lead to superiority in outcome.
Mechanistic Insights into Alkali-Catalyzed Phosphorylation
The core of this synthetic innovation lies in the precise control of the phosphorylation mechanism under alkali catalysis. The reaction initiates with the nucleophilic attack of the phosphate oxygen of cytidylic acid on the phosphorus center of dichlorophosphoryl morpholine. The presence of a base, such as potassium carbonate or triethylamine, serves a dual purpose: it neutralizes the hydrogen chloride generated during the substitution reaction, driving the equilibrium forward, and it potentially enhances the nucleophilicity of the phosphate group by forming a transient phosphate salt. This mechanistic pathway is highly favorable because dichlorophosphoryl morpholine is a highly reactive electrophile, yet the morpholine group acts as a stable leaving group that can be subsequently hydrolyzed under controlled aqueous conditions. The choice of solvent, typically polar aprotic solvents like acetonitrile or dioxane, is critical to maintaining the solubility of the ionic intermediates while preventing premature hydrolysis of the sensitive phosphochloridate species before the coupling is complete.
Impurity control is intrinsically built into this mechanism through the physical properties of the byproducts. Unlike organic coupling byproducts that co-elute with the product, the salts generated (such as potassium chloride or triethylamine hydrochloride) are largely insoluble in the organic reaction medium or can be easily filtered off prior to the hydrolysis step. Furthermore, the hydrolysis of the acyl chloride intermediate is conducted at low temperatures (around 0°C) to prevent the degradation of the sensitive pyrimidine base or the cleavage of the newly formed diphosphate bond. The final purification via recrystallization exploits the differential solubility of the Cytidine Diphosphate in ethanol-water mixtures versus potential mono-phosphate contaminants or unreacted starting materials. This rigorous control over reaction parameters ensures that the final product meets stringent HPLC purity specifications, often exceeding 96%, which is crucial for its application in sensitive biological assays and therapeutic formulations.
How to Synthesize Cytidine Diphosphate Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control to maximize yield and minimize side reactions. The process is designed to be operationally simple, making it accessible for manufacturing teams looking to transition from lab-scale optimization to pilot production. The following guide outlines the critical phases of the synthesis, emphasizing the specific conditions that lead to the high yields reported in the patent literature. Operators should note that the selection of the base and solvent can be tuned based on availability and cost, providing flexibility in the supply chain without compromising the integrity of the final product.
- Condense cytidylic acid with dichlorophosphoryl morpholine in an organic solvent (e.g., acetonitrile) using an alkali base like potassium carbonate at 0-50°C.
- Filter off salt byproducts and concentrate the filtrate to an oily residue.
- Hydrolyze the acyl chloride intermediate with water, then induce crystallization using an alcohol/water mixed system to obtain high-purity Cytidine Diphosphate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method translates into tangible strategic advantages that go beyond mere technical feasibility. The elimination of expensive coupling reagents like DCC and the removal of ion-exchange chromatography steps result in a drastically simplified cost structure. Traditional nucleotide synthesis often incurs high costs due to the consumption of stoichiometric amounts of activating agents and the extensive resin usage required for purification. By replacing these with inexpensive inorganic bases and simple crystallization techniques, the variable cost per kilogram of the final intermediate is significantly reduced. Moreover, the use of common commodity chemicals such as acetonitrile, methanol, and potassium carbonate ensures that raw material sourcing is stable and resilient against market volatility, securing the continuity of supply for long-term production contracts.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the reduction in both material and processing costs. By avoiding the use of DCC, manufacturers eliminate the expense associated with purchasing this high-cost reagent and the subsequent waste disposal fees related to its urea byproduct. Furthermore, the ability to purify the product via crystallization rather than chromatography represents a massive saving in both time and consumables, as chromatographic resins are capital-intensive and have limited lifecycles. This lean manufacturing approach allows for a more competitive pricing model for the final Cytidine Diphosphate, enabling downstream drug manufacturers to optimize their own COGS while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available, non-proprietary raw materials. Unlike specialized enzymatic substrates or custom-synthesized protecting groups that may have single-source suppliers, reagents like dichlorophosphoryl morpholine and cytidylic acid are produced by multiple chemical vendors globally. This diversification of the supplier base mitigates the risk of shortages and allows procurement teams to negotiate better terms. Additionally, the robustness of the reaction conditions, which tolerate a range of temperatures and solvent grades, reduces the likelihood of batch failures due to minor variations in raw material quality, ensuring consistent output and reliable delivery schedules to customers.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this route is exceptionally well-suited for industrial expansion. The process generates solid inorganic waste (salts) that can often be recycled or disposed of with lower environmental impact compared to the complex organic waste streams of traditional methods. The patent data confirms successful scale-up from gram quantities to multi-kilogram batches (e.g., 2kg scale in Example 7) without loss of yield or purity, demonstrating excellent linear scalability. This predictability allows engineering teams to design production facilities with confidence, knowing that the process kinetics and heat transfer requirements observed at the bench scale will translate effectively to reactor vessels, facilitating a smoother technology transfer and faster time-to-market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy techniques. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is this method superior to traditional DCC-mediated coupling for CDP synthesis?
A: Traditional methods often rely on DCC (dicyclohexylcarbodiimide), which generates dicyclohexylurea (DCU) as a byproduct that is notoriously difficult to remove completely, contaminating the final API. This novel method avoids DCC entirely, utilizing dichlorophosphoryl morpholine which generates easily removable inorganic salts, significantly simplifying purification and improving final product purity.
Q: How does this process achieve high purity without ion exchange chromatography?
A: The process leverages a specific recrystallization technique using an ethanol/water mixed solvent system. By carefully controlling the temperature and solvent ratios during the crystallization phase, impurities remain in the mother liquor while the target Cytidine Diphosphate precipitates as a high-purity solid, eliminating the need for expensive and time-consuming ion exchange column separation.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates successful scale-up from gram scale to kilogram scale (Example 7 utilizes 2kg of starting material). The use of common, low-cost reagents like potassium carbonate and acetonitrile, combined with simple filtration and crystallization unit operations, makes it highly adaptable for commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cytidine Diphosphate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity profiles demonstrated in patent CN113999270B can be reliably replicated at an industrial level. We understand that for critical intermediates like Cytidine Diphosphate, consistency is key; therefore, our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain needs. Whether you require a Customized Cost-Saving Analysis to quantify the economic benefits of switching to this route or need to verify specific technical parameters, our technical procurement team is ready to assist. We encourage you to reach out to request specific COA data and route feasibility assessments tailored to your project requirements. By partnering with NINGBO INNO PHARMCHEM, you secure not just a supplier, but a strategic ally committed to driving efficiency and innovation in the production of high-value pharmaceutical intermediates.
