Advanced Palladium-Pyridine Catalysis for Scalable Benzodiazepine Pharmaceutical Intermediates Manufacturing
Advanced Palladium-Pyridine Catalysis for Scalable Benzodiazepine Pharmaceutical Intermediates Manufacturing
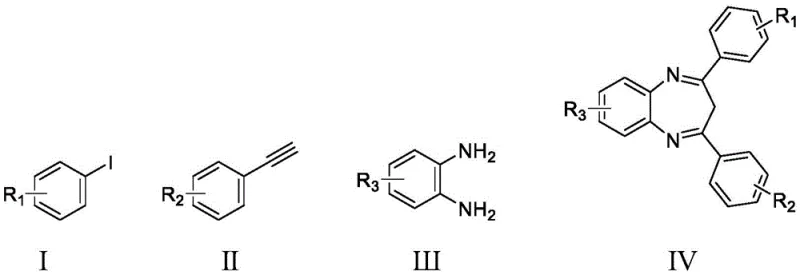
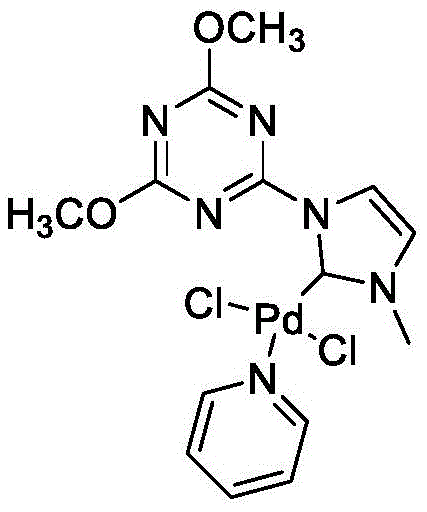
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for bioactive heterocycles, particularly benzodiazepines, which serve as critical scaffolds for anticonvulsants, anxiolytics, and sedatives. A groundbreaking approach detailed in patent CN112812070A introduces a highly efficient method for preparing these compounds via palladium-pyridine catalysis. This innovative technology leverages a four-component coupling reaction involving iodobenzene derivatives, phenylacetylene derivatives, o-phenylenediamine derivatives, and carbon monoxide. By utilizing a specialized palladium pyridine complex, the process achieves exceptional catalytic activity with remarkably low metal loading, addressing key pain points in modern drug substance manufacturing regarding cost and environmental impact.
This novel synthetic pathway represents a significant leap forward for reliable pharmaceutical intermediate suppliers aiming to optimize their production portfolios. The method operates under mild reaction conditions, typically between 60-100°C and under moderate CO pressure of 3-6 atm, eliminating the need for extreme thermal or pressure parameters often associated with traditional carbonylation reactions. The versatility of this chemistry allows for the introduction of diverse functional groups, enabling the rapid generation of structural analogs essential for medicinal chemistry campaigns. For procurement and supply chain leaders, this translates to a more resilient sourcing strategy for complex heterocyclic building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzodiazepine cores has relied on several established but often problematic methodologies. Traditional routes frequently involve the condensation of 1,3-diketones with o-phenylenediamine or the reaction of nitriles and unsaturated functional groups with diamines. These classical approaches often necessitate the use of harsh reaction media, including strong mineral acids or bases, which can lead to significant safety hazards and generate substantial amounts of corrosive waste streams. Furthermore, many existing protocols require microwave assistance or elevated temperatures that are difficult to control on a multi-ton scale, posing risks for thermal runaway and limiting the feasibility of commercial scale-up.
Another critical drawback of conventional methods is the inefficiency regarding catalyst usage and step economy. Many transition-metal catalyzed alternatives reported in literature suffer from high catalyst loading requirements, driving up the cost of goods sold (COGS) due to the price of precious metals like palladium. Additionally, the purification of final products from these reactions can be arduous, often requiring extensive chromatographic separation to remove metal residues and side products. This complexity not only extends the production cycle time but also reduces the overall yield, making it challenging to achieve the high purity standards demanded by regulatory bodies for active pharmaceutical ingredients (APIs).
The Novel Approach
In stark contrast, the technology disclosed in CN112812070A offers a streamlined, one-pot solution that fundamentally reshapes the economic and operational landscape of benzodiazepine synthesis. By employing a unique palladium pyridine catalyst, the new method facilitates a direct four-component coupling that constructs the seven-membered diazepine ring with high precision. The reaction conditions are notably mild, proceeding efficiently at temperatures ranging from 60-100°C, which significantly lowers energy consumption compared to high-temperature reflux methods. The use of carbon monoxide as a carbonyl source ensures high atom economy, incorporating the carbonyl group directly into the scaffold without the need for pre-functionalized acyl chlorides or other activated species.
The operational simplicity of this novel approach is a major advantage for industrial application. The reaction system demonstrates remarkable stability towards air, reducing the stringent requirement for inert atmosphere handling that typically complicates organometallic processes. Moreover, the catalyst loading is exceptionally low, ranging from 0.01 to 0.02 mol% relative to the iodobenzene substrate. This drastic reduction in precious metal usage not only lowers raw material costs but also simplifies downstream processing, as there is less metal residue to remove from the final product. The result is a green, efficient, and cost-effective pathway that aligns perfectly with the goals of sustainable chemical manufacturing.
Mechanistic Insights into Palladium-Pyridine Catalyzed Carbonylation
The core of this technological breakthrough lies in the unique structure and reactivity of the palladium pyridine catalyst. Unlike standard palladium salts which may aggregate or deactivate under reaction conditions, this specialized complex features a coordinated pyridine ligand environment that stabilizes the active palladium species. This stabilization is crucial for maintaining high turnover numbers (TON) throughout the reaction cycle. The mechanism likely involves the oxidative addition of the aryl iodide to the palladium center, followed by coordination and insertion of the alkyne and carbon monoxide. The presence of the pyridine ligand modulates the electronic properties of the metal center, facilitating these migratory insertion steps while preventing the formation of inactive palladium black.
Furthermore, the catalyst exhibits excellent tolerance to various functional groups, which is essential for the synthesis of diverse pharmaceutical intermediates. The reaction proceeds through a cyclization step where the o-phenylenediamine component attacks the activated intermediate, closing the seven-membered ring to form the benzodiazepine core. The mild basicity of triethylamine used in the system aids in neutralizing the HI byproduct generated during the oxidative addition, driving the equilibrium forward without degrading sensitive functional groups on the substrate. This mechanistic elegance ensures that the reaction yields are consistently high, often exceeding 90% even with sterically hindered or electronically diverse substrates.
Impurity control is another area where this mechanism excels. The high selectivity of the palladium-pyridine system minimizes the formation of homocoupling byproducts or oligomerization of the alkyne, which are common issues in other palladium-catalyzed reactions. The clean reaction profile means that the crude product contains fewer impurities, simplifying the purification workflow. For R&D directors focused on impurity profiling and regulatory compliance, this level of control is invaluable. It reduces the burden on analytical teams to identify and quantify trace impurities, accelerating the timeline from process development to clinical supply.
How to Synthesize Benzodiazepine Compounds Efficiently
The practical implementation of this synthesis route is straightforward and amenable to standard chemical processing equipment. The process begins with the preparation of the active catalyst, followed by the assembly of the reaction mixture in a suitable solvent like toluene. The reaction is then pressurized with carbon monoxide and heated, after which standard workup procedures such as filtration and evaporation are employed. The detailed standardized synthesis steps, including specific molar ratios and purification techniques, are outlined in the guide below to ensure reproducibility and optimal yield.
- Prepare the palladium pyridine catalyst by reacting compound A with palladium dichloride and potassium carbonate in pyridine at 40°C.
- Combine iodobenzene derivative, phenylacetylene derivative, o-phenylenediamine derivative, the prepared catalyst, and triethylamine in toluene.
- Introduce CO gas to 3-6 atm pressure and react at 60-100°C for 6-12 hours, followed by purification via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this palladium-pyridine catalyzed method offers tangible strategic benefits that extend beyond mere technical performance. The primary driver of value is the significant reduction in manufacturing costs achieved through process intensification. By lowering the catalyst loading to trace levels, the direct material cost associated with precious metals is minimized. Additionally, the use of commodity chemicals like iodobenzenes and phenylacetylenes as starting materials ensures a stable and competitive supply base, mitigating the risk of raw material shortages that can plague more exotic synthetic routes.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the ultra-low loading of the palladium catalyst. In traditional cross-coupling reactions, catalyst costs can constitute a significant portion of the total variable cost, especially when scaling to metric ton quantities. By reducing the catalyst requirement to 0.01-0.02 mol%, the process effectively decouples production volume from precious metal expenditure. Furthermore, the elimination of harsh reagents like strong acids reduces the cost of corrosion-resistant equipment and waste neutralization, leading to substantial overall cost savings in API manufacturing.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain continuity by reducing dependency on specialized reagents or complex multi-step sequences. The starting materials, such as substituted iodobenzenes and o-phenylenediamines, are widely available from global chemical suppliers, ensuring consistent availability. The mild reaction conditions also mean that the process can be executed in a broader range of manufacturing facilities without requiring specialized high-pressure or high-temperature reactors, thereby increasing the pool of qualified contract manufacturing organizations (CMOs) capable of producing these intermediates.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this method aligns with green chemistry principles, facilitating easier regulatory approval and community acceptance. The high atom economy and reduced waste generation lower the environmental footprint of the manufacturing process. The absence of strong corrosive agents simplifies effluent treatment, reducing the cost and complexity of wastewater management. These factors collectively make the commercial scale-up of complex pharmaceutical intermediates more feasible and sustainable, ensuring long-term viability for high-volume production campaigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and stakeholders.
Q: What are the primary advantages of this palladium-pyridine catalytic method over traditional benzodiazepine synthesis?
A: This method significantly reduces the required amount of palladium catalyst to trace levels (0.01-0.02 mol%) while maintaining high catalytic activity and air stability. Unlike conventional methods that often require harsh strong acids, strong bases, or microwave assistance, this process operates under mild conditions (60-100°C) with excellent atom economy.
Q: What represents the substrate scope for this four-component coupling reaction?
A: The reaction demonstrates broad substrate applicability, tolerating various substituents on the iodobenzene, phenylacetylene, and o-phenylenediamine components. Electron-donating groups like methyl and methoxy, as well as electron-withdrawing halogens such as fluorine, chlorine, and bromine, are all compatible, yielding products with high purity and efficiency.
Q: How does this technology impact the cost structure for large-scale pharmaceutical intermediate production?
A: By drastically lowering the loading of the expensive palladium catalyst and utilizing readily available raw materials like iodobenzenes and alkynes, the overall manufacturing cost is substantially reduced. Furthermore, the simplified operation and short reaction times minimize energy consumption and equipment occupancy, enhancing overall process economics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzodiazepine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-pyridine catalyzed synthesis for the global pharmaceutical market. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including safe CO handling and precise temperature control, while our rigorous QC labs guarantee that every batch meets stringent purity specifications required for clinical and commercial applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this route can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the supply of high-purity pharmaceutical intermediates.
