Advanced Catalyst-Free Synthesis of High-Purity 2,6-Dichloro Benzoxazole for Commercial Herbicide Production
The production of high-purity heterocyclic intermediates remains a critical bottleneck in the global agrochemical supply chain, particularly for the synthesis of phenoxy carboxylic acid herbicides such as fenoxaprop. Patent CN102558086B introduces a transformative synthesizing method for preparing high-purity 2,6-dichloro benzoxazole, addressing long-standing issues regarding safety, purity, and process scalability. This innovative approach replaces hazardous traditional chlorinating agents with a controlled, catalyst-free system utilizing di(trichloromethyl) carbonate and a finishing agent of trichloromethyl chloroformate. By implementing a precise stage-wise heating protocol, the process achieves a molar yield exceeding 98 percent and a mass content of over 99 percent directly from crystallization. For R&D directors and procurement managers seeking a reliable agrochemical intermediate supplier, this technology represents a significant leap forward in process intensification and risk mitigation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2,6-dichloro benzoxazole has been plagued by severe safety hazards and inefficient purification steps that drive up operational costs. One prevalent legacy route utilizes phosphorus pentachloride (PCl5) as the chlorinating agent, which causes serious corrosion to reactor equipment, making it difficult to detect structural integrity failures until a catastrophic breach occurs. If moisture inadvertently enters a corroded system containing PCl5, a vigorous and explosive reaction ensues, creating unacceptable safety liabilities for manufacturing plants. Furthermore, alternative routes employing chlorine gas suffer from poor regioselectivity, leading to addition reactions on the phenyl ring that generate numerous by-products and result in low molar yields of only 80-82 percent. Another common method relies on phosgene, which, while effective, is a severely toxic chemical subject to strict national controls on storage and transportation, effectively disqualifying manufacturers without specialized phosgene resources from entering the market.
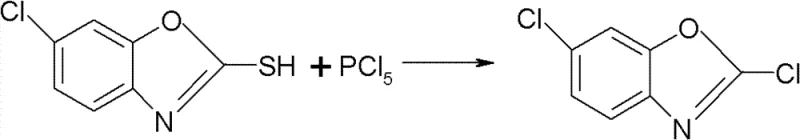
The Novel Approach
The patented methodology fundamentally reengineers the reaction landscape by eliminating the need for corrosive inorganic chlorinating agents and toxic gaseous phosgene. Instead, it employs di(trichloromethyl) carbonate, a solid phosgene substitute that offers superior handling safety and higher purity with fewer inherent impurities compared to raw phosgene or PCl5. A key differentiator of this novel approach is the abandonment of amine catalysts like DMF, which are known to persist in the final product and complicate downstream purification. By combining this safer reagent profile with a unique stage-heating technique and a final "polishing" step using a small amount of trichloromethyl chloroformate, the reaction proceeds to near-completion without generating the complex impurity profiles seen in older methods. This allows for the direct isolation of the target molecule via simple cooling and crystallization, bypassing the energy-intensive high-vacuum rectification required by previous BTC-based routes.
Mechanistic Insights into Catalyst-Free Stage Heating Chlorination
The core innovation of this synthesis lies in the kinetic control of the decomposition of di(trichloromethyl) carbonate through a meticulously managed temperature ramping profile. Rather than applying rapid heat which could lead to uncontrolled exothermic decomposition and the sudden release of large volumes of phosgene, the process heats the reaction mixture from an initial 45-55°C to a final range of 80-110°C at a slow, controlled rate of 0.2-1°C per minute. This gradual thermal input ensures that the chlorinating species are generated in situ at a rate that matches the consumption by the 2-sulfhydryl-6-chlorobenzoxazole substrate, thereby maintaining a low concentration of reactive intermediates and minimizing side reactions. The absence of a catalyst further simplifies the mechanistic pathway, preventing the formation of stable catalyst-substrate complexes that often trap impurities or require harsh conditions to break down.
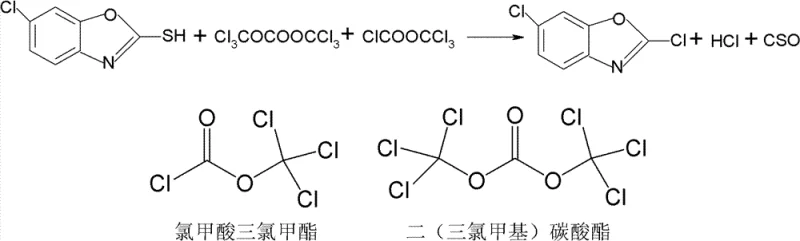
Impurity control is achieved through a dual-mechanism strategy involving both the primary reagent and a secondary finishing agent. The primary reaction with di(trichloromethyl) carbonate converts the bulk of the thiol group, but to ensure absolute conversion and prevent the persistence of unreacted starting material, a small stoichiometric amount of trichloromethyl chloroformate is introduced in the final stage. This secondary reagent acts as a scavenger for any remaining thiol groups, driving the reaction equilibrium completely toward the desired dichloro product. Because the process avoids DMF, which typically forms difficult-to-remove adducts and degrades at high temperatures to form dimethylamine impurities, the resulting crude solid is exceptionally clean. The subsequent removal of a portion of the solvent followed by cooling to 0-5°C induces rapid crystallization of the pure product, leaving soluble impurities in the mother liquor which can be recycled, thus enhancing the overall atom economy of the process.
How to Synthesize 2,6-Dichloro Benzoxazole Efficiently
The operational protocol for this synthesis is designed for robustness and ease of scale-up, requiring standard glass-lined or stainless steel reactors capable of precise temperature control. The process begins by suspending the thiol precursor in a hydrocarbon solvent such as toluene or xylene, followed by the addition of the solid carbonate reagent. The critical phase involves the slow, stage-wise heating cycle which must be strictly adhered to in order to manage gas evolution and reaction kinetics. Once the primary conversion is achieved, the finishing agent is added dropwise, and after a short holding period, the solvent volume is reduced under vacuum to supersaturate the solution before cooling.
- Mix 2-sulfhydryl-6-chlorobenzoxazole with di(trichloromethyl) carbonate in a solvent like toluene and heat gradually to 50°C.
- Perform stage-wise heating to 105-110°C at a controlled rate, maintaining the temperature for 0.5 to 2 hours to allow slow decomposition.
- Add a small amount of trichloromethyl chloroformate, react for another hour, then remove solvent and crystallize the product at 0°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers profound strategic benefits that extend beyond simple yield improvements. The elimination of phosphorus pentachloride removes the massive capital expenditure associated with frequent reactor replacements due to corrosion, while also mitigating the risk of unplanned downtime caused by equipment failure. Furthermore, the shift away from gaseous phosgene alleviates the stringent regulatory burdens and security costs associated with storing and transporting Schedule 1 chemicals, allowing for a more flexible and resilient supplier network. The ability to produce the intermediate as a high-purity solid directly from the reactor significantly reduces the utility consumption associated with high-vacuum distillation, leading to substantial cost savings in energy and processing time.
- Cost Reduction in Manufacturing: The catalyst-free nature of this process eliminates the expensive and complex steps required to remove residual metal or amine catalysts from the final product. In traditional methods, the presence of DMF necessitates high-energy distillation or extensive washing procedures to meet purity specifications, whereas this new route allows for direct crystallization. This simplification of the downstream processing train drastically reduces the consumption of solvents, energy, and labor hours, translating into a significantly lower cost of goods sold (COGS) for the final agrochemical active ingredient.
- Enhanced Supply Chain Reliability: By utilizing solid reagents like di(trichloromethyl) carbonate instead of hazardous gases or corrosive solids, the logistics of raw material procurement become far more stable and less prone to regulatory disruption. Suppliers of solid carbonyl sources are more numerous and operate under less restrictive transport protocols than phosgene producers, ensuring a continuous flow of materials even during periods of heightened safety inspections. This reliability is crucial for maintaining uninterrupted production schedules for critical herbicide intermediates in a volatile global market.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of heavy metal or persistent organic pollutants in the waste stream simplify environmental compliance and waste treatment protocols. The process generates fewer hazardous by-products and allows for the recycling of mother liquors, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing site. This ease of scalability ensures that production can be ramped from pilot batches to multi-ton commercial runs without encountering the thermal runaway or purification bottlenecks typical of older chlorination technologies.
Frequently Asked Questions (FAQ)
The following technical queries address common concerns regarding the implementation and quality assurance of this advanced synthesis method. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the technology's viability for your specific application requirements. Understanding these nuances is essential for technical teams assessing the feasibility of integrating this intermediate into their existing herbicide production lines.
Q: Why is the new synthesis method safer than using Phosphorus Pentachloride (PCl5)?
A: The conventional PCl5 route poses severe corrosion risks to reactors and potential explosion hazards if moisture enters the system. The new method utilizes di(trichloromethyl) carbonate and trichloromethyl chloroformate under mild conditions, eliminating these safety critical failures.
Q: How does this process achieve >99% purity without complex purification?
A: By avoiding catalysts like DMF which create difficult-to-remove impurities, and utilizing a stage-heating protocol that ensures complete reaction of the starting material, the product precipitates directly as a high-purity solid upon cooling, bypassing the need for energy-intensive high-vacuum distillation.
Q: What are the supply chain advantages of using Di(trichloromethyl) Carbonate over Phosgene?
A: Phosgene is a highly toxic gas with strict storage and transportation regulations that limit supplier options. Di(trichloromethyl) carbonate is a solid substitute that is safer to handle and transport, ensuring more reliable supply continuity and reducing logistical compliance costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,6-Dichloro Benzoxazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the consistent supply of high-purity intermediates is the backbone of efficient agrochemical manufacturing. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and risk-free. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2,6-dichloro benzoxazole meets the exacting standards required for downstream herbicide synthesis. Our commitment to quality assurance ensures that our clients receive materials that facilitate high-yield final couplings without the need for additional purification steps.
We invite global partners to leverage our technical expertise to optimize their supply chains and reduce overall production costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals in the agrochemical sector.
