Advanced Enzymatic Resolution Technology for Commercial Scale Production of Chiral Amines
Introduction to High-Efficiency Chiral Amine Synthesis
The production of optically active amines serves as a cornerstone for the modern pharmaceutical and agrochemical industries, where stereochemical purity dictates biological efficacy. Patent CN1220702A introduces a transformative methodology for synthesizing these critical intermediates through an enzymatic kinetic resolution process that significantly outperforms traditional chemical resolution techniques. This innovation addresses the longstanding industrial challenge of achieving high optical purity without compromising on reaction kinetics or economic feasibility. By leveraging specific hydrolases, particularly lipases from Candida antarctica, the process enables the enantioselective acylation of racemic amines under mild conditions. The technical breakthrough lies in the unexpected finding that esters lacking electron-rich heteroatoms adjacent to the carbonyl carbon can effectively drive this resolution, contrary to prior teachings which suggested such structures were ineffective. This discovery opens a vast chemical space for substrate variation, allowing manufacturers to utilize readily available and cost-effective acylating agents.
Furthermore, the robustness of this catalytic system allows for operation at relatively high substrate concentrations, directly translating to improved volumetric productivity in commercial reactors. For R&D directors and process chemists, this represents a pivotal shift away from the inefficient, highly diluted protocols of the past towards a more intensified manufacturing paradigm. The ability to isolate both (S)- and (R)-enantiomers with excellent optical purity provides a versatile platform for developing diverse drug candidates and crop protection agents. As global demand for chiral intermediates escalates, adopting such efficient biocatalytic routes becomes not just a technical preference but a strategic necessity for maintaining competitive advantage in the fine chemicals sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the enzymatic resolution of amines has been plagued by significant operational inefficiencies that hindered large-scale adoption. Prior art, such as that described in DE-A 4332738, relied heavily on esters possessing electron-rich heteroatoms in the acid moiety adjacent to the carbonyl group to achieve enantioselectivity. While these methods could yield optically pure products, they suffered from critically low space-time yields due to the requirement for extremely long reaction times. To mitigate side reactions and maintain enzyme activity, these processes necessitated highly dilute solutions, which drastically increased solvent consumption and downstream processing costs. Additionally, the high enzyme-to-substrate ratios required in these older protocols made the biocatalyst a major cost driver, rendering the overall process economically unattractive for commodity-level production. The purification and concentration steps were also cumbersome, often requiring multiple attempts to isolate the product without degrading the sensitive enzyme or the product itself.
The Novel Approach
The methodology disclosed in the present patent fundamentally disrupts these limitations by demonstrating that effective kinetic resolution can be achieved using simple esters that do not possess the previously deemed essential electron-rich heteroatoms. This counter-intuitive finding allows for the use of inexpensive and widely available acylating agents, such as ethyl acetate or ethyl chloroacetate, rather than specialized, costly reagents. Crucially, the reaction can be conducted at significantly higher substrate concentrations, which dramatically improves the space-time yield and reduces the reactor volume required for a given output. The process operates efficiently within a moderate temperature range of 10°C to 60°C, ensuring enzyme stability while maintaining rapid reaction kinetics. This novel approach not only simplifies the supply chain by utilizing common chemical feedstocks but also enhances the overall sustainability of the manufacturing process by reducing solvent waste and energy consumption associated with concentrating dilute streams.
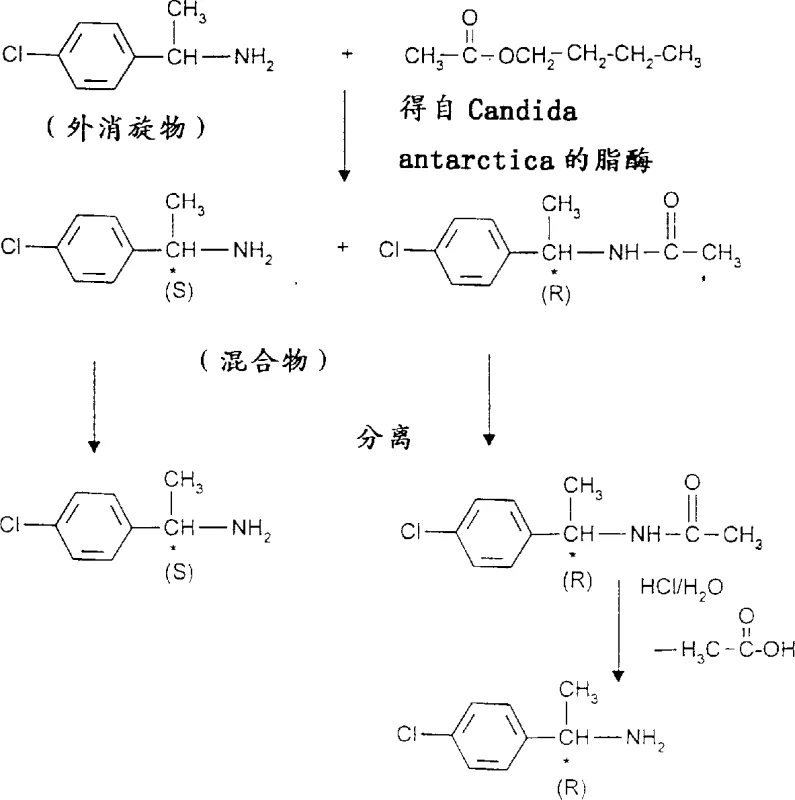
Mechanistic Insights into Lipase-Catalyzed Kinetic Resolution
The core mechanism driving this synthesis is the enantioselective acylation of the racemic amine substrate by the immobilized lipase. In this kinetic resolution, the enzyme distinguishes between the two enantiomers of the starting amine based on their spatial configuration at the chiral center. The lipase from Candida antarctica preferentially catalyzes the acylation of the (R)-enantiomer of the amine when reacted with the chosen ester, converting it into the corresponding acylated amide derivative. Meanwhile, the (S)-enantiomer remains largely unreacted in the reaction mixture. This differential reactivity creates a chemical distinction between the two isomers, transforming a difficult separation of enantiomers into a straightforward separation of chemically distinct species: a free amine and an amide. The structural integrity of the chiral center is preserved throughout this transformation, ensuring that the optical purity established during the enzymatic step is maintained in the final isolated products.
Following the enzymatic conversion, the separation strategy exploits the differences in basicity and solubility between the free (S)-amine and the acylated (R)-amide. Standard acid-base extraction techniques allow for the clean partitioning of the unreacted (S)-amine into the aqueous phase as a salt, while the neutral acylated (R)-amide remains in the organic phase. If the (R)-amine is the desired target, a subsequent hydrolysis step using strong acid or base cleaves the amide bond, regenerating the free amine with inverted or retained configuration depending on the specific pathway, though in this specific patent context, it releases the (R)-amine from the acylated precursor. This mechanistic clarity provides process engineers with predictable control over impurity profiles, as the primary byproduct is simply the alcohol derived from the ester, which is easily removed during workup.

How to Synthesize Optically Active Amines Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize enantiomeric excess and conversion rates. The process begins with the preparation of a reaction mixture containing the racemic amine, the selected ester acylating agent, and the immobilized biocatalyst in a suitable organic solvent such as methyl tert-butyl ether or dimethoxyethane. The choice of ester is critical; while ethyl acetate is effective, esters like ethyl chloroacetate or ethyl cyanoacetate can offer enhanced reactivity and easier downstream handling. The reaction is typically monitored via gas chromatography to determine the optimal endpoint, usually targeting around 50% conversion to maximize the optical purity of both the remaining substrate and the formed product. Once the desired conversion is reached, the solid biocatalyst is removed by simple filtration, allowing for potential reuse of the enzyme in subsequent batches, which further drives down operational costs.
- React racemic amines with specific esters (e.g., ethyl chloroacetate) in the presence of immobilized lipase from Candida antarctica (Novozym 435) at 10-60°C.
- Separate the resulting mixture into the unreacted (S)-amine and the acylated (R)-amine using standard extraction or distillation techniques.
- Optionally hydrolyze the acylated (R)-amine using acid or base treatment to release the free (R)-amine with high optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers compelling advantages that directly impact the bottom line and operational resilience. The ability to use commodity esters instead of specialized acylating agents significantly reduces raw material costs and mitigates supply risk associated with niche chemicals. Furthermore, the high stability of the Candida antarctica lipase allows for extended catalyst life and potential recycling, as evidenced by experimental data showing minimal activity loss over multiple cycles. This durability translates to a lower frequency of enzyme replenishment orders and reduced inventory holding costs for biocatalysts. The process operates under atmospheric pressure and moderate temperatures, eliminating the need for expensive high-pressure reactors or complex cooling infrastructure, thereby lowering capital expenditure requirements for new production lines.
- Cost Reduction in Manufacturing: The elimination of expensive, specialized acylating components in favor of readily available esters drastically lowers the bill of materials for each production batch. Additionally, the ability to run reactions at higher concentrations reduces the volume of solvents required, which subsequently lowers the costs associated with solvent purchase, recovery, and waste disposal. The simplified workup procedure, relying on standard filtration and extraction rather than complex chromatographic separations, further reduces labor and utility costs per kilogram of product. These cumulative efficiencies result in a substantially more cost-competitive manufacturing process compared to traditional resolution methods.
- Enhanced Supply Chain Reliability: By utilizing robust, commercially available enzymes like Novozym 435 and common chemical feedstocks, the supply chain becomes less vulnerable to disruptions caused by single-source suppliers of exotic reagents. The process tolerance for varying substrate structures means that the same production line can be adapted to manufacture a wide range of chiral amine intermediates, providing flexibility to respond to shifting market demands. The high yields and optical purities achieved reduce the need for reprocessing or off-spec material management, ensuring a consistent and reliable flow of high-quality intermediates to downstream customers.
- Scalability and Environmental Compliance: The benign nature of the reagents and the absence of heavy metal catalysts align perfectly with modern green chemistry principles and stringent environmental regulations. The aqueous waste streams generated are easier to treat compared to those from transition metal-catalyzed processes, reducing the burden on wastewater treatment facilities. The scalability of the process is proven by the successful demonstration of gram-to-kilolab scale reactions with consistent results, indicating a low risk profile for commercial scale-up. This environmental and operational compatibility facilitates faster regulatory approvals and smoother technology transfer to large-scale manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic resolution technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation, providing a reliable basis for feasibility assessments. Understanding these details is crucial for project managers evaluating the integration of this process into existing manufacturing portfolios. The clarity provided here aims to accelerate decision-making and reduce the time required for initial technical due diligence.
Q: What represents the primary advantage of this enzymatic process over prior art?
A: Unlike previous methods requiring long reaction times and highly dilute solutions, this process utilizes esters without electron-rich heteroatoms adjacent to the carbonyl, allowing for higher substrate concentrations, shorter reaction times, and improved space-time yields.
Q: Which biocatalyst is preferred for this resolution technique?
A: The patent specifically highlights the use of lipase obtained from Candida antarctica, commercially available as Novozym 435, due to its stability at elevated temperatures and high enantioselectivity.
Q: Can this method produce both (R) and (S) enantiomers?
A: Yes, the process yields the (S)-amine directly from the reaction mixture, while the (R)-amine is obtained after separating and hydrolyzing the acylated (R)-amide derivative, allowing access to both optical isomers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Amines Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity chiral intermediates play in the development of next-generation therapeutics and agrochemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of patents like CN1220702A are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of optically active amine meets the exacting standards required by global regulatory bodies. Our commitment to quality assurance ensures that our clients receive materials that facilitate smooth downstream synthesis without unexpected impurities or delays.
We invite you to collaborate with us to leverage this advanced enzymatic technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and target specifications. Please contact us to request specific COA data and route feasibility assessments for your desired chiral amine intermediates. By partnering with us, you gain access to a supply chain that prioritizes efficiency, quality, and long-term reliability, empowering your R&D and production teams to focus on innovation while we handle the complexities of fine chemical manufacturing.
