Advanced Synthesis of 2,3-Thiomorpholine Diketone-2-Oxime Derivatives for Pharmaceutical Applications
The pharmaceutical landscape for gastrointestinal therapeutics continues to evolve, driven by the persistent global demand for effective treatments against gastric and duodenal ulcers. Patent CN1016689B introduces a groundbreaking methodology for the preparation of 2,3-thiomorpholine diketone-2-oxime derivatives, a class of compounds distinguished by their potent cytoprotective properties and ability to inhibit acid secretion. This technology represents a significant leap forward from earlier iterations of heterocyclic chemistry, offering a robust pathway to high-value active pharmaceutical ingredients (APIs). The core innovation lies in the precise functionalization of the thiomorpholine ring, specifically targeting the nitrogen atom to create derivatives with superior pharmacological profiles compared to historical agrochemical analogues.
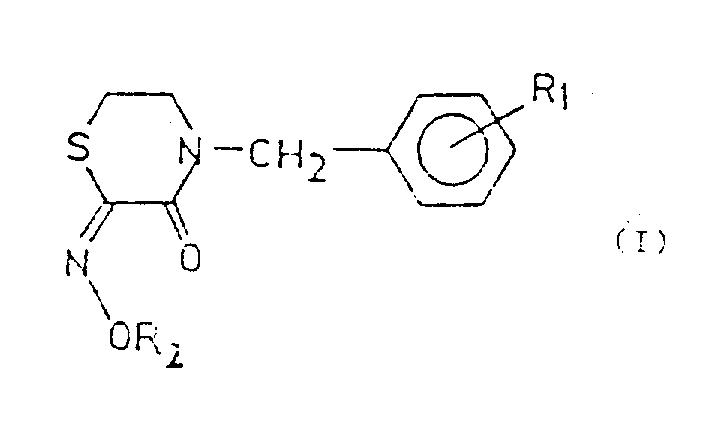
For R&D directors and process chemists, the structural versatility offered by this patent is paramount. The general Formula I encompasses a wide array of substituents where R1 can be hydrogen, halogen, or various alkyl and alkoxy groups, while R2 allows for extensive modification including ester, cyano, amino, and acyl groups. This modularity enables the fine-tuning of physicochemical properties such as solubility and metabolic stability, which are critical factors in drug development. By establishing a reliable pharmaceutical intermediates supplier network capable of executing this specific chemistry, organizations can secure a steady pipeline of novel candidates for ulcer therapy, addressing a market need that remains substantial despite the availability of older proton pump inhibitors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thiomorpholine derivatives was largely dominated by methodologies intended for agricultural applications, as evidenced by patents from major chemical corporations like Union Carbide and DuPont. These conventional routes often failed to address the specific stereochemical and electronic requirements necessary for mammalian therapeutic efficacy. A critical limitation in prior art was the connectivity of the benzyl group; in many agrochemical precursors, the benzyl moiety was not linked directly to the nitrogen atom on the thiomorpholine ring, resulting in compounds with negligible cytoprotective activity. Furthermore, traditional methods frequently relied on harsh reaction conditions or expensive transition metal catalysts that complicated downstream purification and introduced toxic heavy metal impurities, posing significant challenges for cost reduction in API manufacturing and regulatory compliance.
The Novel Approach
The methodology disclosed in CN1016689B overcomes these historical barriers by introducing a targeted alkylation and acylation strategy specifically designed for medicinal chemistry. This novel approach utilizes a readily accessible intermediate, the 2,3-thiomorpholine diketone-2-oxime (Formula II), as a versatile scaffold. By reacting this core with specific electrophiles (Formula III or IV) under mild basic conditions, the process achieves high regioselectivity at the nitrogen center. This ensures that the resulting derivatives possess the correct structural orientation required for binding to biological targets involved in ulcer formation. The shift from agrochemical-focused synthesis to pharma-grade precision not only enhances biological activity but also streamlines the production workflow, eliminating the need for complex rearrangement steps that typically plague older synthetic routes.
Mechanistic Insights into Base-Catalyzed N-Alkylation and Acylation
At the heart of this synthesis is a nucleophilic substitution mechanism facilitated by inorganic or organic bases. The process begins with the deprotonation of the oxime nitrogen in the Formula II intermediate, generating a highly reactive nucleophile. 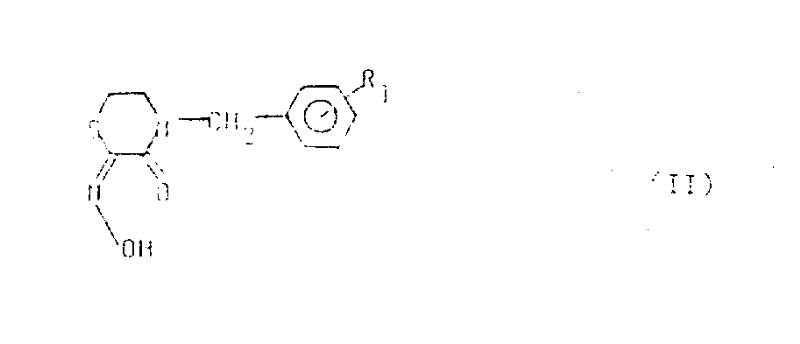 This anionic species then attacks the electrophilic carbon of the alkylating agent (Formula III), where the leaving group X is typically a halogen, mesyloxy, or tosyloxy group. The choice of solvent plays a crucial role in stabilizing the transition state; polar aprotic solvents like dimethylformamide (DMF) or dimethyl sulfoxide (DMSO) are particularly effective in enhancing the nucleophilicity of the oxime anion, thereby accelerating the reaction rate without requiring excessive thermal energy. This mechanistic efficiency is key to achieving the high yields reported in the patent examples.
This anionic species then attacks the electrophilic carbon of the alkylating agent (Formula III), where the leaving group X is typically a halogen, mesyloxy, or tosyloxy group. The choice of solvent plays a crucial role in stabilizing the transition state; polar aprotic solvents like dimethylformamide (DMF) or dimethyl sulfoxide (DMSO) are particularly effective in enhancing the nucleophilicity of the oxime anion, thereby accelerating the reaction rate without requiring excessive thermal energy. This mechanistic efficiency is key to achieving the high yields reported in the patent examples.
Impurity control is another critical aspect of this mechanism, particularly for high-purity pharmaceutical intermediates. The reaction conditions are optimized to minimize side reactions such as O-alkylation, which can occur competitively with N-alkylation. By carefully selecting the base—ranging from potassium hydroxide to triethylamine—and controlling the temperature between 0°C and 100°C, the process favors the thermodynamic N-substituted product. Post-reaction workup involves simple filtration to remove inorganic salts followed by evaporation, which effectively separates the organic product from the reaction matrix. This simplicity in purification is a major advantage, as it reduces the reliance on resource-intensive chromatographic separations, thereby lowering the overall environmental footprint and production costs associated with commercial scale-up of complex heterocyclic compounds.
How to Synthesize 2,3-Thiomorpholine Diketone-2-Oxime Efficiently
The synthesis of these valuable derivatives follows a logical two-stage progression that balances chemical efficiency with operational simplicity. The first stage involves the preparation of the core oxime scaffold from a nitromethylene thiazolidine precursor using alkaline hydrolysis, a reaction that proceeds with exceptional yield (up to 91%) under reflux conditions. The second stage involves the functionalization of this core using the alkylation or acylation protocols described above. Detailed standardized synthetic steps for this process are provided in the guide below, ensuring reproducibility and quality control for manufacturing teams.
- Preparation of the Core Intermediate: Synthesize the 2,3-thiomorpholine diketone-2-oxime core (Formula II) by reacting 2-Nitromethylene-3-(phenylmethyl)thiazolidine with an alkaline reagent such as sodium hydroxide in an aqueous-alcoholic medium under reflux.
- Alkylation or Acylation Reaction: React the Formula II intermediate with the appropriate electrophile (Formula III for alkylation/acylation or Formula IV for formylation) in an inert organic solvent like acetone or DMF, utilizing an inorganic or organic base.
- Purification and Isolation: Filter off inorganic salts, evaporate the solvent, and purify the resulting crude product through recrystallization or column chromatography to achieve high-purity specifications suitable for pharmaceutical use.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond mere chemical novelty. The process is designed with industrial scalability in mind, utilizing commodity chemicals and solvents that are globally available and cost-effective. By eliminating the need for exotic catalysts or cryogenic conditions, the manufacturing process becomes significantly more robust against supply chain disruptions. This resilience is crucial for maintaining reducing lead time for high-purity pharmaceutical intermediates, ensuring that production schedules remain uninterrupted even in volatile market conditions. The ability to source raw materials locally further insulates the supply chain from geopolitical risks and freight cost fluctuations.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by its reliance on inexpensive reagents and straightforward unit operations. The use of common bases like sodium hydroxide or triethylamine, coupled with solvents such as acetone and ethanol, drastically reduces the raw material expenditure compared to processes requiring precious metal catalysts. Furthermore, the simplified workup procedure, which often requires only filtration and evaporation rather than complex distillation or extraction sequences, leads to substantial savings in utility consumption and labor hours. These efficiencies compound at scale, resulting in a significantly lower cost of goods sold (COGS) for the final active ingredient.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, particularly the thiazolidine derivatives and various alkyl halides, are produced by a wide network of chemical manufacturers worldwide. This diversity of supply sources mitigates the risk of single-supplier dependency, a common vulnerability in the pharmaceutical supply chain. Additionally, the stability of the intermediates allows for flexible inventory management; the core oxime can be stockpiled and functionalized on demand, enabling a responsive 'just-in-time' manufacturing model that aligns perfectly with fluctuating market demand for ulcer therapies.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is inherently greener than many alternatives. The absence of heavy metals simplifies waste treatment and ensures that the final product meets stringent residual solvent and impurity guidelines set by regulatory bodies like the FDA and EMA. The reaction generates minimal hazardous waste, primarily consisting of inorganic salts that are easy to dispose of or recycle. This alignment with green chemistry principles not only reduces disposal costs but also enhances the corporate sustainability profile, a factor increasingly weighted in vendor selection by top-tier pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these thiomorpholine derivatives. The answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for decision-makers evaluating this technology for their pipelines.
Q: What are the primary therapeutic applications of these thiomorpholine derivatives?
A: According to patent CN1016689B, these compounds exhibit significant cytoprotective effects and inhibit acid secretion, making them highly effective intermediates for treating gastric and duodenal ulcers.
Q: How does this synthesis method improve upon conventional agrochemical routes?
A: Unlike prior art focused on agrochemicals where the benzyl group was not linked to the ring nitrogen, this novel process specifically targets the nitrogen atom on the thiomorpholine ring, optimizing the molecular structure for mammalian pharmacological activity.
Q: What solvents and conditions are optimal for scaling this reaction?
A: The process utilizes common industrial solvents such as acetone, ethanol, and dimethylformamide (DMF) at temperatures ranging from 0°C to 100°C, ensuring excellent scalability and safety for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Thiomorpholine Diketone-2-Oxime Supplier
As the global demand for effective gastrointestinal therapeutics continues to rise, securing a manufacturing partner with deep technical expertise is essential. NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to ensure that every batch of 2,3-thiomorpholine diketone-2-oxime derivatives meets stringent purity specifications. We understand the critical nature of these intermediates in the drug development lifecycle and are committed to delivering consistent quality that accelerates your time-to-market.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced synthesis capabilities can support your next breakthrough in ulcer therapy.
