Advanced Asymmetric Synthesis of Pseudophrynaminol: Technical Breakthroughs for Commercial Scale-up
Advanced Asymmetric Synthesis of Pseudophrynaminol: Technical Breakthroughs for Commercial Scale-up
The pharmaceutical landscape is increasingly demanding high-purity chiral intermediates, particularly for neurological applications where enantiomeric purity dictates biological efficacy. Patent CN1070647A presents a groundbreaking methodology for the preparation of optically active pseudophrynaminol, a potent alkaloid with potential therapeutic applications in treating senile dementia and myasthenia gravis. Unlike traditional extraction methods from Australian frog skin which are unsustainable and yield insufficient quantities for clinical trials, this synthetic route offers a robust, scalable alternative. The core innovation lies in the strategic introduction of a multi-functional group (MRG) that serves simultaneously as a protecting group, a solubility enhancer, and a chiral inducer. This technical insight report analyzes the mechanistic superiority of this approach, providing R&D directors and procurement leaders with a clear understanding of its value proposition as a reliable pharmaceutical intermediate supplier solution. By leveraging this asymmetric synthesis technology, manufacturers can bypass the limitations of racemic synthesis, ensuring that the final active pharmaceutical ingredient (API) meets the stringent stereochemical requirements necessary for modern drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pseudophrynaminol and its analogues has been plagued by significant chemical inefficiencies that hinder commercial viability. Prior art, such as the methods disclosed by Miguel O. Mitchell and Pier Giorgio Cozzi, predominantly yields racemic mixtures, containing equal parts of levo and dextro forms. This is a critical drawback because the biological activities of these enantiomers differ substantially; a racemic mixture obscures the specific therapeutic potential of the active isomer and complicates toxicological profiling. Furthermore, conventional routes often involve lengthy synthetic sequences with multiple protection and deprotection steps, leading to cumulative yield losses and increased waste generation. The reliance on resolution techniques to separate enantiomers from a racemic bulk is inherently inefficient, theoretically capping the maximum yield at 50% for the desired isomer unless dynamic kinetic resolution is employed, which adds further complexity and cost. For supply chain heads, these inefficiencies translate into higher raw material consumption, extended production lead times, and a larger environmental footprint, making the cost reduction in API intermediate manufacturing a challenging objective with legacy technologies.
The Novel Approach
The methodology outlined in CN1070647A fundamentally restructures the synthetic pathway to prioritize stereocontrol from the outset. By utilizing a chiral urea derivative, specifically N-(S)-(-)-1-phenylethyl urea, the process introduces chirality early in the synthesis, converting the problem of enantiomer separation into a more manageable diastereomer separation. This strategic shift allows for the selective alkylation of the 2-oxindole scaffold, where the chiral auxiliary directs the incoming alkyl group to a specific face of the molecule. The result is a mixture of diastereomers that possess distinct physical properties, enabling their separation via standard silica gel column chromatography with high efficiency. This approach eliminates the theoretical 50% yield ceiling associated with racemic resolution, potentially doubling the output of the desired optically active material from the same amount of starting inputs. Additionally, the reaction conditions are notably mild, operating effectively within a temperature range of -40°C to solvent reflux, which reduces energy consumption and minimizes the degradation of sensitive intermediates. For procurement managers, this translates to a more predictable supply of high-purity alkaloid intermediates with significantly reduced processing costs.
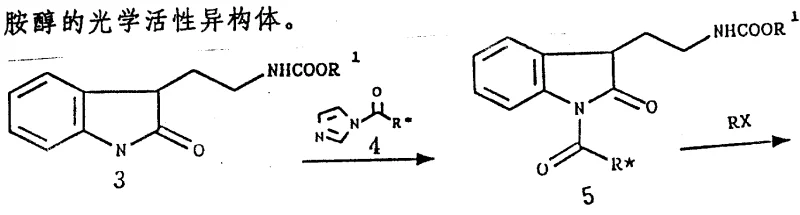
Mechanistic Insights into Chiral Auxiliary-Mediated Asymmetric Alkylation
The heart of this technological advancement is the sophisticated use of the multi-role group (MRG) which functions as a chiral auxiliary. In the initial step, the substituted 2-oxindole (Compound 3) reacts with the chiral urea (Compound 4) in the presence of a strong base such as sodium hydride or potassium hydride. This reaction forms a carbamoyl intermediate (Compound 5) where the chiral information from the phenylethyl group is transferred to the indole nitrogen. The steric bulk of the chiral group creates a differentiated environment around the reactive center, effectively shielding one face of the molecule from electrophilic attack. When the alkylating agent, such as bromomethyl crotonate, is introduced, it approaches the intermediate preferentially from the less hindered side. This diastereoselective alkylation is the critical control point that determines the optical purity of the final product. The patent specifies that various chiral auxiliaries can be employed, including derivatives with phenyl or naphthyl groups, allowing for fine-tuning of the stereoselectivity based on specific steric requirements. This level of control is essential for R&D directors who need to ensure that the impurity profile of the intermediate remains within tight specifications to facilitate regulatory approval for downstream drug substances.
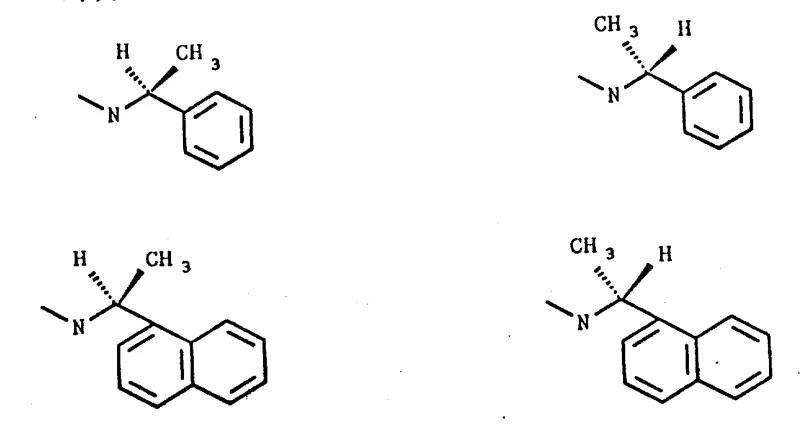
Following the alkylation, the resulting diastereomers (Compound 6 and 7) are separated using flash silica gel chromatography. The efficiency of this separation is bolstered by the distinct polarity differences induced by the spatial arrangement of the chiral auxiliary and the newly added alkyl chain. Once isolated, the pure diastereomers undergo a reduction step, typically utilizing Lithium Aluminium Hydride (LiAlH4) or catalytic hydrogenation. This reduction cleaves the carbonyl groups and removes the chiral auxiliary, releasing the free optically active pseudophrynaminol. The removal of the auxiliary is clean and quantitative, ensuring that no trace of the chiral inducer remains in the final product, which is vital for meeting stringent purity specifications. The patent data indicates that this route can achieve high yields, with the alkylation step reporting yields up to 98% and the final reduction step achieving 76%. The ability to produce both levorotatory and dextrorotatory forms by simply switching the enantiomer of the starting chiral auxiliary provides unparalleled flexibility. This mechanistic robustness ensures that the commercial scale-up of complex pharmaceutical intermediates can proceed with consistent quality and minimal batch-to-batch variation.
How to Synthesize Pseudophrynaminol Efficiently
The synthesis of pseudophrynaminol via this patented route involves a sequence of highly optimized chemical transformations designed for maximum efficiency and stereocontrol. The process begins with the preparation of the 2-oxindole scaffold from readily available tryptamine, followed by the critical asymmetric induction step using chiral ureas. The subsequent alkylation and reduction steps are engineered to preserve the optical integrity of the molecule while maximizing throughput. For process chemists and manufacturing teams, understanding the precise order of reagent addition and temperature control is vital to replicating the high yields reported in the patent literature. The detailed standardized synthesis steps see the guide below.
- Preparation of substituted 2-oxindole intermediate via carbamation of tryptamine derivatives followed by acid-catalyzed cyclization.
- Asymmetric induction by reacting the 2-oxindole with a chiral N-(S)-(-)-1-phenylethyl urea derivative in the presence of a strong base like sodium hydride.
- Diastereoselective alkylation using bromomethyl crotonate, followed by chromatographic separation of diastereomers and final reduction with Lithium Aluminium Hydride.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this asymmetric synthesis route offers profound advantages for supply chain stability and cost management. Traditional methods that rely on natural extraction or racemic synthesis are fraught with supply risks and inefficiency. Natural extraction is limited by the availability of biological sources and suffers from low throughput, while racemic synthesis wastes half of the produced material. In contrast, this novel approach utilizes synthetic starting materials that are commercially available in bulk, ensuring a continuous and reliable supply chain. The elimination of transition metal catalysts in favor of organic bases and hydride reagents simplifies the purification process, removing the need for expensive and time-consuming heavy metal scavenging steps. This reduction in processing complexity directly correlates to lower manufacturing costs and shorter production cycles. For procurement managers, this means a more stable pricing structure and the ability to secure long-term contracts without the volatility associated with scarce natural resources or complex catalytic systems.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis route significantly lowers the cost of goods sold (COGS) by reducing the number of unit operations required. By avoiding the need for chiral chromatography to separate enantiomers and instead relying on diastereomer separation, the process utilizes standard, cost-effective silica gel media. Furthermore, the high yields reported in the alkylation and reduction steps minimize raw material waste, ensuring that a greater proportion of input costs are converted into saleable product. The absence of precious metal catalysts also removes a major cost driver and supply risk, as the price and availability of metals like palladium or platinum can fluctuate wildly. This qualitative improvement in process efficiency allows for substantial cost savings that can be passed down the supply chain, making the final API more competitive in the global market.
- Enhanced Supply Chain Reliability: Dependence on natural sources for alkaloids creates a fragile supply chain vulnerable to environmental and geopolitical disruptions. This synthetic route decouples production from biological constraints, enabling manufacturing in controlled industrial environments year-round. The use of robust chemical reagents such as sodium hydride and lithium aluminium hydride, which are staples of the fine chemical industry, ensures that raw material sourcing is straightforward and reliable. Additionally, the ability to synthesize both enantiomers on demand provides flexibility to meet varying market needs without retooling the entire production line. This adaptability reduces lead time for high-purity pharmaceutical intermediates, allowing downstream drug manufacturers to accelerate their own development timelines and respond more quickly to clinical trial demands.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are amenable to large-scale production, with temperatures and pressures that fall within standard industrial operating ranges. The solvent systems employed, such as tetrahydrofuran (THF) and dichloromethane, are well-understood and can be efficiently recovered and recycled, minimizing environmental impact. The reduction in waste generation, achieved through higher yields and fewer purification steps, aligns with modern green chemistry principles and regulatory expectations. This ease of scale-up ensures that the transition from laboratory gram-scale to multi-ton commercial production is smooth and predictable. For supply chain heads, this means the confidence to commit to large volume orders knowing that the manufacturing partner has a proven, scalable pathway that complies with strict environmental and safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and benefits of the process. Understanding these details is crucial for stakeholders evaluating the integration of this intermediate into their supply chain.
Q: How does this patent address the issue of racemic mixtures in pseudophrynaminol synthesis?
A: The patent introduces a chiral multi-role group (MRG) that induces asymmetry during the alkylation step, allowing for the separation of diastereomers and the isolation of pure enantiomers, unlike prior art which produced racemic forms.
Q: What are the key advantages of the reaction conditions described in CN1070647A?
A: The process utilizes mild reaction temperatures ranging from -40°C to reflux and avoids harsh transition metal catalysts, significantly simplifying downstream purification and reducing heavy metal contamination risks.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route is characterized by short synthetic steps, high yields (up to 98% in alkylation), and the use of commercially available reagents, making it highly adaptable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pseudophrynaminol Supplier
The technical potential of the asymmetric synthesis route described in CN1070647A is immense, offering a pathway to high-purity chiral alkaloids that are essential for next-generation neurological therapeutics. NINGBO INNO PHARMCHEM stands ready to leverage this technology, bringing our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to your project. Our facility is equipped with rigorous QC labs and stringent purity specifications to ensure that every batch of pseudophrynaminol meets the exacting standards required for pharmaceutical applications. We understand the critical nature of chiral purity and impurity control, and our team is dedicated to maintaining the integrity of this sophisticated synthesis from raw material intake to final delivery.
We invite you to initiate a conversation about optimizing your supply chain for this critical intermediate. Our technical procurement team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with us, you gain access to specific COA data and route feasibility assessments that will empower your R&D and procurement teams to make informed decisions. Let us help you secure a stable, cost-effective, and high-quality supply of optically active pseudophrynaminol to support your drug development goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
