Advanced Synthesis of 4-Aldehyde-2,2-Difluorobenzodioxole for Scalable Agrochemical Production
Advanced Synthesis of 4-Aldehyde-2,2-Difluorobenzodioxole for Scalable Agrochemical Production
The global demand for high-efficiency, low-toxicity agricultural fungicides has placed significant pressure on the supply chains of key intermediates such as 4-aldehyde-2,2-difluorobenzodioxole (CAS 119895-68-0). As the critical precursor for Fludioxonil, a phenylpyrrole fungicide with broad-spectrum activity, the economic and technical viability of its production is paramount for the agrochemical industry. Patent CN101851225A introduces a transformative synthetic methodology that fundamentally shifts the feedstock paradigm from expensive, scarce phenols to abundant o-cresol. This technical breakthrough addresses long-standing bottlenecks regarding raw material availability, process safety, and environmental compliance, offering a robust pathway for reliable agrochemical intermediate supplier networks to secure long-term production capacity.
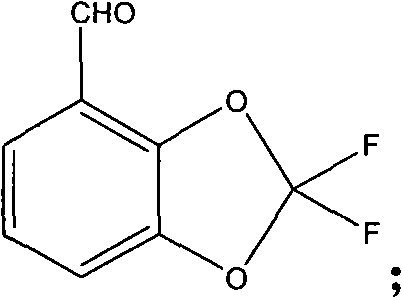
The strategic value of this patent lies in its comprehensive re-engineering of the synthetic route, moving away from multi-step derivatizations of catechol or vanillin which are often plagued by high costs and supply volatility. By leveraging ortho-orientating formylation followed by a telescoped Dakin reaction and cyclization, the process achieves a level of atom economy and operational simplicity that was previously unattainable. For R&D directors and procurement managers alike, understanding the mechanistic nuances of this route is essential for evaluating its potential for cost reduction in agrochemical intermediate manufacturing. The following analysis dissects the technical superiority of this method, highlighting its implications for commercial scale-up of complex heterocyclic compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-aldehyde-2,2-difluorobenzodioxole has relied on pathways that are inherently inefficient and economically burdensome. Traditional routes often commence with catechol, requiring etherification, chlorination, fluorination, and acylation, a sequence that frequently necessitates the use of n-Butyl Lithium in the final acylation step. The reliance on n-Butyl Lithium imposes severe reaction conditions, including cryogenic temperatures and strict anhydrous environments, which are difficult to maintain safely on a large industrial scale. Alternative methods utilizing O-vanillin suffer from raw material constraints, as O-vanillin is typically a byproduct of vanillin synthesis with limited market availability, creating a fragile supply chain susceptible to price fluctuations. Furthermore, routes starting from 3-methylpyrocatechol, while chemically feasible, do not solve the fundamental issue of high raw material costs, as this starting material itself is expensive and not produced in the massive volumes required for commodity agrochemical production.
The Novel Approach
In stark contrast, the methodology disclosed in CN101851225A utilizes o-cresol, a ubiquitous and low-cost petrochemical derivative, as the primary feedstock. This shift alone represents a monumental advantage in cost reduction in agrochemical intermediate manufacturing, decoupling production from the volatile markets of fine phenolic compounds. The novel approach integrates a direct ortho-formylation to generate 3-methylsalicylaldehyde, followed immediately by a Dakin reaction to produce 3-methylpyrocatechol in situ. Crucially, this intermediate is not isolated; it proceeds directly to cyclization with dichloromethane or dibromomethane. This telescoping of steps eliminates multiple work-up and purification stages, drastically reducing solvent usage and processing time. Additionally, the substitution of elemental chlorine gas with phosphorus pentachloride for the chlorination step mitigates significant environmental and safety hazards, aligning the process with modern green chemistry principles and regulatory standards.
Mechanistic Insights into Ortho-Formylation and Direct Oxidation
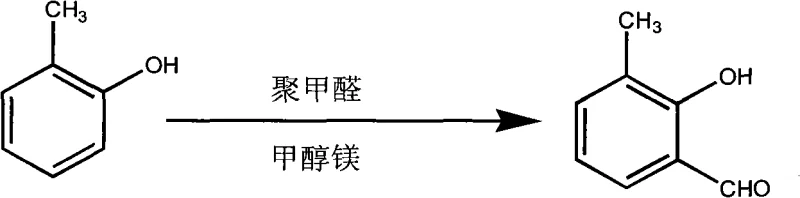
The cornerstone of this synthetic strategy is the efficient functionalization of the o-cresol ring system. The process initiates with the generation of magnesium methylate from magnesium chips and anhydrous methanol, which serves as a base to facilitate the ortho-orientating formylation using paraformaldehyde. This reaction selectively installs the aldehyde functionality adjacent to the hydroxyl group, setting the stage for the subsequent transformation. The resulting 3-methylsalicylaldehyde is then subjected to a Dakin oxidation using alkaline hydrogen peroxide. This step converts the aldehyde group into a second hydroxyl group, effectively generating the 3-methylpyrocatechol moiety required for dioxole ring formation. The elegance of this mechanism lies in its compatibility with the subsequent cyclization, allowing the reaction mixture to be transferred directly to an autoclave for reaction with methylene chloride under basic conditions and phase-transfer catalysis.
Following the formation of the 4-methylbenzodioxole core, the synthesis employs a sophisticated three-step modification of the methyl substituent to achieve the final aldehyde functionality. Unlike conventional routes that might involve radical chlorination followed by hydrolysis, this patent describes a chlorination using phosphorus pentachloride to form a dichloromethyl intermediate, followed by fluorination with hydrogen fluoride-triethylamine complex to install the difluoro group on the dioxole ring. The final, and perhaps most critical, transformation is the oxidation of the methyl group to an aldehyde. This is achieved using sodium persulfate in the presence of catalytic amounts of ferrous sulfate and cupric acetate. This metal-catalyzed oxidation system offers a controlled and selective pathway to the aldehyde, avoiding the over-oxidation to carboxylic acids that often plagues such transformations, thereby ensuring high purity specifications for the final product.

How to Synthesize 4-Aldehyde-2,2-Difluorobenzodioxole Efficiently
The practical implementation of this synthesis requires careful control of reaction parameters to maximize yield and minimize impurity formation. The process begins with the preparation of 3-methylsalicylaldehyde, where temperature control during the addition of paraformaldehyde is critical to prevent polymerization side reactions. Following the Dakin oxidation, the direct cyclization step demands precise management of pressure and temperature within the autoclave to ensure complete conversion of the diol to the dioxole ring. The subsequent chlorination and fluorination steps must be conducted under strictly anhydrous conditions to prevent hydrolysis of the reactive intermediates, particularly the acid chloride species generated during chlorination. Finally, the oxidation step requires a balanced addition of the persulfate oxidant to maintain the reaction exotherm within safe limits while ensuring complete conversion of the methyl group. Detailed standardized operating procedures for these critical unit operations are essential for technology transfer and successful commercialization.
- Perform ortho-orientating formylation on o-cresol using magnesium methylate and paraformaldehyde to obtain 3-methylsalicylaldehyde.
- Convert 3-methylsalicylaldehyde to 3-methylpyrocatechol via Dakin reaction, then react directly with dichloromethane to form 4-methylbenzodioxole without isolation.
- Execute a three-step sequence on 4-methylbenzodioxole involving chlorination with PCl5, fluorination with HF/triethylamine, and oxidation with sodium persulfate to yield the final aldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this o-cresol-based route offers compelling strategic advantages that extend beyond simple unit cost savings. The primary benefit is the stabilization of the supply chain through the use of o-cresol, a commodity chemical produced in vast quantities globally, which eliminates the supply risks associated with niche starting materials like O-vanillin or 3-methylpyrocatechol. This abundance ensures consistent availability of raw materials, reducing lead time for high-purity agrochemical intermediates and protecting downstream production schedules from upstream disruptions. Furthermore, the elimination of hazardous reagents such as chlorine gas and n-Butyl Lithium simplifies the regulatory compliance landscape, reducing the administrative burden and insurance costs associated with handling highly dangerous substances.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the convergence of cheap raw materials and streamlined processing. By utilizing o-cresol, the baseline material cost is significantly lower than that of catechol or vanillin derivatives. Moreover, the telescoping of the Dakin reaction and cyclization into a continuous sequence without intermediate isolation results in substantial cost savings by reducing solvent consumption, energy usage for distillation, and labor hours. The avoidance of expensive organometallic reagents like n-Butyl Lithium further contributes to a leaner cost structure, making the final product highly competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved by decoupling production from the limited supply chains of specialized phenolic compounds. O-cresol is a standard petrochemical derivative with a robust and diversified global supply network, ensuring that production can be scaled up rapidly to meet surging demand without encountering raw material bottlenecks. This reliability is crucial for maintaining the continuity of Fludioxonil production, a key crop protection agent, and allows manufacturers to offer more stable pricing and delivery commitments to their customers.
- Scalability and Environmental Compliance: From an operational perspective, the replacement of gaseous chlorine with solid phosphorus pentachloride simplifies the engineering requirements for the chlorination step, removing the need for complex gas handling and scrubbing systems. This not only lowers capital expenditure for plant setup but also significantly reduces the environmental footprint of the process by minimizing the risk of toxic gas emissions. The direct oxidation of the methyl group to an aldehyde further enhances the environmental profile by avoiding the generation of chlorinated waste streams associated with hydrolysis routes, facilitating easier waste treatment and disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 4-aldehyde-2,2-difluorobenzodioxole, based on the specific innovations detailed in the patent literature. These insights are designed to clarify the operational benefits and chemical rationale behind the o-cresol route, providing decision-makers with the necessary information to evaluate its feasibility for their specific manufacturing contexts. Understanding these details is key to appreciating the full value proposition of this advanced synthetic methodology.
Q: Why is o-cresol preferred over catechol or vanillin derivatives for this synthesis?
A: O-cresol is a significantly cheaper and more readily available commodity chemical compared to catechol or O-vanillin. Using o-cresol eliminates the reliance on expensive byproducts and avoids the severe reaction conditions (such as n-Butyl Lithium) associated with traditional catechol routes, thereby enhancing supply chain stability.
Q: How does this novel route improve environmental safety compared to conventional methods?
A: The process replaces hazardous chlorine gas with phosphorus pentachloride for chlorination, significantly reducing environmental pollution risks. Furthermore, the final conversion of the methyl group to an aldehyde utilizes a direct oxidation method rather than a chlorination-hydrolysis route, simplifying operations and minimizing toxic waste generation.
Q: What are the key operational advantages of the one-pot Dakin and cyclization steps?
A: By performing the Dakin reaction and subsequent cyclization without isolating the intermediate 3-methylpyrocatechol, the process drastically reduces solvent consumption, processing time, and material loss. This telescoped approach streamlines the workflow, leading to substantial cost reductions and higher overall throughput for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Aldehyde-2,2-Difluorobenzodioxole Supplier
The technical advancements outlined in patent CN101851225A represent a significant leap forward in the production of Fludioxonil intermediates, yet translating laboratory success to industrial reality requires a partner with deep process engineering expertise. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this o-cresol route are fully realized in a commercial setting. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of 4-aldehyde-2,2-difluorobenzodioxole meets the exacting standards required for modern agrochemical formulations, providing our partners with a reliable foundation for their own manufacturing success.
We invite global agrochemical leaders to collaborate with us to leverage this cost-effective and environmentally superior synthetic route. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, as well as specific COA data and route feasibility assessments. Let us help you secure a sustainable and competitive supply of this critical intermediate, driving efficiency and profitability in your agrochemical portfolio.
