Industrial Scale-Up of Benvitimod: A Novel Green Catalytic Route for Pharmaceutical Intermediates
Introduction to Advanced Benvitimod Synthesis
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally benign synthetic routes for high-value active pharmaceutical ingredients (APIs) and their intermediates. A significant breakthrough in this domain is detailed in Chinese Patent CN112661611A, which discloses a novel preparation method for benzenediol bioactive substances, specifically targeting Benvitimod (3,5-dihydroxy-4-isopropyl-trans-stilbene) and its derivatives. This patent addresses critical bottlenecks in existing manufacturing processes by introducing a direct phosphonation strategy that bypasses traditional halogenation steps. For R&D directors and procurement specialists, this innovation represents a paradigm shift towards greener chemistry, offering a pathway to reduce reliance on toxic reagents while enhancing overall process efficiency. The method leverages a synergistic catalytic system involving potassium iodide and phase transfer catalysts to activate benzyl alcohol directly, a feat previously difficult to achieve with high selectivity. By integrating this technology, manufacturers can potentially lower the barrier to entry for producing this first-in-class aromatic hydrocarbon receptor agonist, which has shown immense promise in treating autoimmune diseases such as psoriasis.
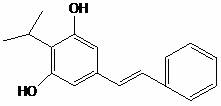
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthetic routes for Benvitimod and related stilbene derivatives have been plagued by significant operational hazards and environmental burdens. As illustrated in various prior art disclosures, such as the route depicted in CN103172497A, traditional methods often rely on a multi-step sequence involving Friedel-Crafts alkylation, reduction, and crucially, halogenation using thionyl chloride or elemental bromine. These halogenating agents are not only highly corrosive and toxic but also generate substantial volumes of acidic wastewater and sulfonated impurities that require complex and costly remediation. Furthermore, the subsequent Wittig olefination in conventional pathways typically demands the use of dangerous strong bases like sodium hydride or potassium tert-butoxide under strictly anhydrous and oxygen-free conditions, usually in solvents like tetrahydrofuran which are difficult to recover on a large scale. The cumulative effect of these harsh conditions is a process with low atom economy, high safety risks, and a cumbersome purification profile that struggles to meet modern Good Manufacturing Practice (GMP) standards for pharmaceutical intermediates.
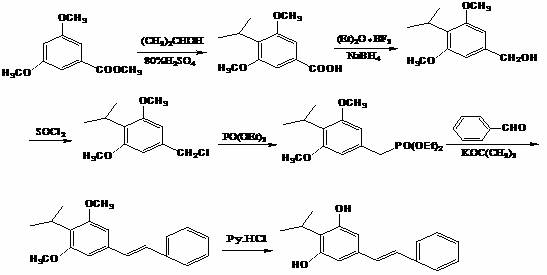
The Novel Approach
In stark contrast, the methodology presented in CN112661611A offers a streamlined and elegant solution that fundamentally redesigns the synthetic logic. The core innovation lies in the direct conversion of 4-isopropyl-3,5-dimethoxybenzyl alcohol into the corresponding phosphonate intermediate without the need for a preceding chlorination or bromination step. This is achieved through a novel catalytic system utilizing potassium iodide and a phase transfer catalyst in a polar aprotic solvent like dimethyl sulfoxide. This direct activation allows for a seamless transition into the Wittig reaction with benzaldehyde using mild inorganic bases such as sodium hydroxide at ambient temperatures. The elimination of the halogenation step removes the need for thionyl chloride and the associated waste streams, while the use of sodium hydroxide replaces hazardous sodium hydride, drastically simplifying the reaction setup and workup. The final product can often be isolated via simple filtration and recrystallization, avoiding the extensive solvent extractions typical of older methods. This approach not only enhances safety but also significantly improves the economic viability of the process by reducing reagent costs and waste disposal fees.
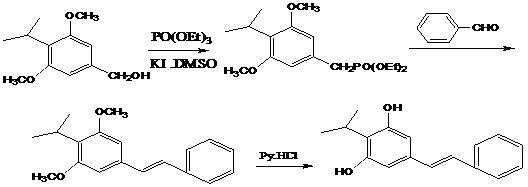
Mechanistic Insights into KI/PTC Catalyzed Direct Phosphonation
The mechanistic elegance of this new route centers on the activation of the benzyl alcohol hydroxyl group by the iodide ion in the presence of a phase transfer catalyst. In traditional Arbuzov reactions, an alkyl halide is required to react with trialkyl phosphite; however, this patent demonstrates that in situ generation of the reactive species can occur directly from the alcohol. The potassium iodide likely facilitates the formation of a transient alkyl iodide or activates the alcohol for nucleophilic attack by the phosphite, while the phase transfer catalyst ensures efficient interaction between the ionic species and the organic substrate in the polar aprotic medium. This catalytic cycle operates effectively at temperatures between 80°C and 130°C, providing the necessary energy to drive the phosphonation to completion without degrading the sensitive methoxy groups on the aromatic ring. Following the formation of the phosphonate, the subsequent Wittig reaction proceeds under remarkably mild conditions. The addition of benzaldehyde and solid sodium hydroxide generates the ylide species in situ, which then condenses with the aldehyde to form the stilbene double bond. The use of a mild base like NaOH is particularly advantageous as it minimizes side reactions such as ester hydrolysis or polymerization, which are common when using stronger, non-nucleophilic bases in protic or semi-protic environments.
From an impurity control perspective, this mechanism offers distinct advantages for ensuring high purity in the final API intermediate. By avoiding the use of thionyl chloride, the process eliminates the formation of sulfonated byproducts and chlorinated impurities that are notoriously difficult to remove and can persist through subsequent steps. Furthermore, the reaction conditions are sufficiently selective to prevent the isomerization of the double bond, ensuring the formation of the desired E-isomer (trans-stilbene) which is critical for biological activity. The workup procedure, which involves simply adding water to precipitate the product or facilitate phase separation, allows for the effective removal of water-soluble catalysts like potassium iodide and the phase transfer agent. This inherent simplicity in the reaction design translates to a cleaner crude product profile, reducing the burden on downstream purification units such as chromatography or repeated recrystallizations, thereby enhancing the overall yield and quality of the high-purity pharmaceutical intermediate.
How to Synthesize Benvitimod Efficiently
The synthesis of Benvitimod via this patented route involves a logical sequence of reactions designed for maximum efficiency and minimal waste. The process begins with the direct phosphonation of the benzyl alcohol precursor, followed immediately by olefination and concluding with a demethylation step to reveal the active phenolic groups. This integrated approach allows for the production of the key stilbene scaffold in high yield with fewer unit operations. For process chemists looking to implement this technology, the detailed standardized synthesis steps are outlined below to ensure reproducibility and safety.
- React 4-isopropyl-3,5-dimethoxybenzyl alcohol with triethyl phosphite in DMSO using KI and a phase transfer catalyst at 80-130°C to form the phosphonate intermediate.
- Perform a Wittig reaction by adding benzaldehyde and sodium hydroxide at 20-30°C, followed by water quenching and filtration to isolate the stilbene derivative.
- Demethylate the product using pyridine hydrochloride at temperatures exceeding 180°C, followed by solvent separation and recrystallization to obtain high-purity Benvitimod.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers compelling strategic advantages that extend beyond mere technical feasibility. The primary benefit lies in the drastic simplification of the supply chain for raw materials. By eliminating the need for specialized and hazardous reagents such as thionyl chloride, elemental bromine, and sodium hydride, manufacturers can source cheaper, more ubiquitous commodities like potassium iodide and sodium hydroxide. This shift reduces the volatility of raw material costs and mitigates the risk of supply disruptions associated with controlled or dangerous chemicals. Additionally, the removal of the halogenation step significantly lowers the environmental compliance burden. Traditional halogenated waste streams require expensive treatment protocols to neutralize acidity and remove heavy metals or halides; avoiding these entirely results in substantial cost savings in waste management and effluent treatment, directly impacting the bottom line of the manufacturing operation.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the consolidation of reaction steps and the substitution of expensive reagents. In conventional routes, the halogenation step often requires stoichiometric amounts of costly activating agents and generates equimolar amounts of acid waste. By bypassing this step entirely, the new method reduces the total mass of reagents required per kilogram of product. Furthermore, the use of sodium hydroxide instead of sodium hydride or potassium tert-butoxide represents a massive reduction in reagent cost, as NaOH is a commodity chemical available at a fraction of the price of specialty bases. The ability to recover and reuse pyridine hydrochloride in the demethylation step further enhances the cost-efficiency profile, creating a closed-loop system that minimizes material loss and maximizes resource utilization in API manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by the robustness of the reaction conditions. Traditional Wittig reactions often require strictly anhydrous solvents and inert atmospheres, necessitating specialized equipment and rigorous quality control of solvents which can delay production schedules. The new method tolerates a broader range of conditions and utilizes solvents like DMSO which are easier to handle and source in bulk quantities. The simplified workup, which often involves filtration rather than complex extractions, reduces the processing time per batch, allowing for faster turnaround and increased throughput. This operational flexibility ensures that production timelines are more predictable and less susceptible to delays caused by equipment maintenance or solvent quality issues, securing a steady flow of high-purity pharmaceutical intermediates to downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor is often hindered by heat transfer issues and safety concerns associated with exothermic reactions involving hazardous reagents. The mild nature of the KI-catalyzed phosphonation and the ambient temperature Wittig reaction make this process inherently safer and easier to scale. The absence of gas evolution (common in hydride reductions) and the use of non-flammable polar solvents reduce the risk of fire and explosion, lowering insurance premiums and safety infrastructure costs. Moreover, the reduction in toxic waste generation aligns perfectly with increasingly stringent global environmental regulations. Manufacturers adopting this green chemistry approach can market their products as sustainably produced, gaining a competitive edge in markets where environmental, social, and governance (ESG) criteria are becoming decisive factors in supplier selection.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this technology resolves historical pain points in Benvitimod production. Understanding these details is crucial for stakeholders evaluating the feasibility of transitioning to this superior manufacturing platform.
Q: How does the new KI-catalyzed method improve safety compared to traditional routes?
A: The novel method eliminates the use of hazardous halogenating agents like thionyl chloride and elemental bromine, as well as dangerous strong bases like sodium hydride, significantly reducing operational risks and waste treatment costs.
Q: What are the key advantages for large-scale manufacturing of Benvitimod?
A: The process combines phosphonation and Wittig steps efficiently, avoids complex anhydrous conditions, allows for the recovery of pyridine hydrochloride, and utilizes inexpensive reagents like sodium hydroxide, making it highly suitable for commercial scale-up.
Q: Can this synthesis route be adapted for related diphenylethane derivatives?
A: Yes, the intermediate E-4-isopropyl-3,5-dimethoxy-stilbene produced by this method can be further reduced and demethylated to synthesize 3,5-dihydroxy-4-isopropyl diphenylethane, demonstrating the versatility of this platform technology.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benvitimod Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising potential of the CN112661611A patent can be fully realized in an industrial setting. Our facilities are equipped with state-of-the-art reactors capable of handling the specific thermal and mixing requirements of the KI-catalyzed phosphonation and subsequent Wittig reactions. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Benvitimod intermediate meets the exacting standards required for pharmaceutical applications, minimizing the risk of batch rejection and ensuring patient safety.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced synthetic technology for your supply chain needs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing processes can deliver high-quality intermediates with superior economic and environmental performance. Let us help you secure a sustainable and cost-effective supply of this critical dermatological API intermediate.
