Industrial Scale Synthesis of 2'-Deoxy-2'-fluorouridine via Novel Triflation-Fluorination Strategy
Industrial Scale Synthesis of 2'-Deoxy-2'-fluorouridine via Novel Triflation-Fluorination Strategy
The pharmaceutical industry continuously demands robust, scalable, and cost-effective routes for critical nucleoside analogs, particularly 2'-deoxy-2'-fluorouridine, which serves as a pivotal intermediate in the synthesis of various antiviral and anticancer agents. Patent CN1795200A discloses a groundbreaking methodology that addresses the longstanding challenges associated with the fluorination of nucleosides, specifically targeting the efficient production of this high-value compound. The core innovation lies in a refined triflation-fluorination sequence that utilizes organic base-hydrofluoric acid complexes, effectively mitigating the severe side reactions and purification bottlenecks inherent in conventional processes. By shifting away from harsh anhydrous hydrofluoric acid conditions and expensive fluorinating reagents like DAST, this technology offers a pathway to high-purity crystalline products suitable for direct commercial application. 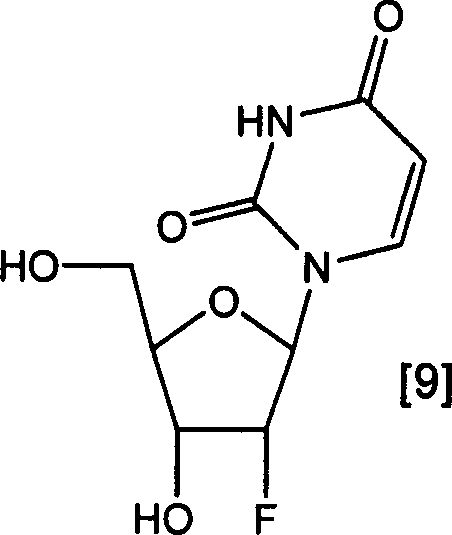
This technical insight report analyzes the mechanistic advantages and commercial implications of this patented route for R&D directors, procurement managers, and supply chain leaders. The ability to produce 2'-deoxy-2'-fluorouridine with exceptional purity through recrystallization rather than column chromatography represents a significant leap forward in process chemistry. For stakeholders in the fine chemical sector, understanding these nuances is critical for evaluating potential suppliers and optimizing manufacturing strategies. The following sections detail how this novel approach overcomes historical limitations, ensuring a reliable supply of high-quality pharmaceutical intermediates while drastically simplifying downstream processing and waste management protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2'-deoxy-2'-fluorouridine has been plagued by significant operational and economic hurdles that hinder industrial scalability. Traditional methods often rely on the ring-opening of 2,2'-anhydrouridine with excessive amounts of highly corrosive anhydrous hydrofluoric acid at elevated temperatures. This approach necessitates specialized reactor materials capable of withstanding extreme corrosion, leading to substantial capital expenditure and maintenance costs. Furthermore, the reaction conditions often result in low throughput due to the requirement for high dilution, and the isolation of the product typically demands labor-intensive column chromatography, which is impractical for multi-kilogram or ton-scale production. Alternative methods utilizing reagents such as DAST (Diethylaminosulfur trifluoride) present their own set of challenges, including high reagent costs, thermal instability, and difficulty in handling large quantities safely. Additionally, prior art involving tetrabutylammonium fluoride (TBAF) for nucleophilic substitution frequently suffers from competitive elimination reactions, yielding unwanted byproducts and reducing overall process efficiency.
The Novel Approach
The methodology outlined in CN1795200A introduces a paradigm shift by employing a two-step sequence involving triflation followed by fluorination with organic base-HF complexes. Instead of using hazardous anhydrous HF or expensive DAST, the process utilizes trifluoromethanesulfonyl fluoride (CF3SO2F) or similar triflating agents in the presence of organic bases like triethylamine or pyridine. This generates a reactive 2'-triflate intermediate which is subsequently subjected to nucleophilic substitution using a salt or complex formed from an organic base and hydrofluoric acid, such as triethylamine-trihydrofluoride. 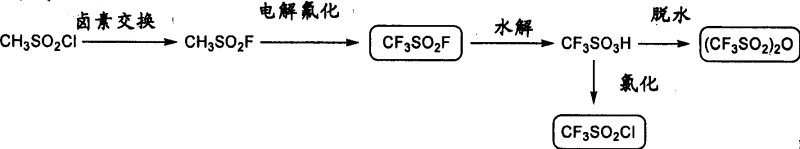
This strategic choice of reagents not only enhances safety by allowing the use of standard glass or stainless steel reactors but also significantly improves atom economy and reaction selectivity. Crucially, the patent describes a unique purification strategy where the crude product is temporarily converted into a 3',5'-diacetyl derivative. This derivative possesses excellent crystallinity, enabling purification via simple recrystallization to achieve high-purity white crystalline powder, thereby completely eliminating the need for column chromatography. This transition from chromatographic purification to crystallization is a major breakthrough for cost reduction in pharmaceutical intermediate manufacturing, offering a streamlined path from raw materials to final API precursors.
Mechanistic Insights into Organic Base-HF Complex Fluorination
The success of this synthetic route hinges on the precise control of nucleophilic substitution dynamics at the 2'-position of the ribose ring. In conventional SN2 reactions using strong bases like TBAF, the trifluoromethanesulfonate (triflate) leaving group is prone to competitive elimination reactions, leading to the formation of unsaturated byproducts where the 1' and 2' carbons form a double bond. The patented process mitigates this by utilizing organic base-HF complexes, which provide fluoride ions with high nucleophilicity but relatively weak basicity. This subtle balance suppresses the elimination pathway, favoring the desired inversion of configuration to yield the 2'-fluoro product with high stereochemical fidelity. The use of tetrahydropyranyl (THP) groups for protecting the 3' and 5' hydroxyl functions further stabilizes the substrate during the triflation step, ensuring that the reaction proceeds cleanly without affecting other sensitive functional groups on the uracil base.
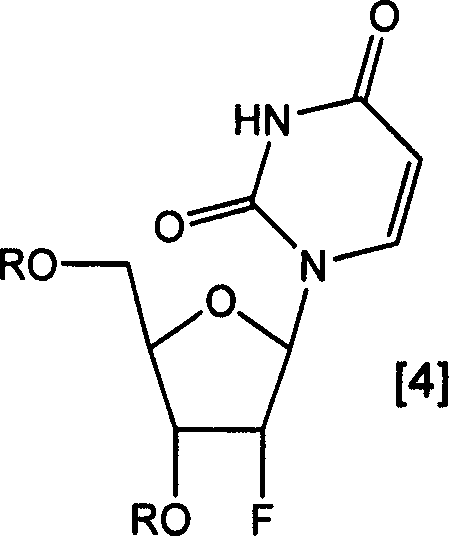
Furthermore, the mechanism allows for a "pot-type" reaction sequence where the triflation and fluorination steps can potentially be telescoped without isolating the unstable triflate intermediate. This reduces solvent usage and processing time, enhancing the overall green chemistry profile of the synthesis. The subsequent deprotection and acetylation steps are carefully optimized to facilitate the formation of the crystalline diacetyl intermediate. By converting the amorphous or oily free nucleoside into a crystalline diacetate, the process leverages differences in solubility to exclude impurities effectively. This mechanistic understanding is vital for R&D teams aiming to replicate or scale this process, as it highlights the importance of reagent selection and temperature control in maximizing yield and minimizing impurity profiles.
How to Synthesize 2'-Deoxy-2'-fluorouridine Efficiently
The synthesis protocol described in the patent offers a comprehensive guide for producing high-purity 2'-deoxy-2'-fluorouridine suitable for pharmaceutical applications. The process begins with the protection of the starting material, followed by activation of the 2'-hydroxyl group via triflation. The subsequent fluorination step utilizes safe and commercially available HF complexes, avoiding the hazards of anhydrous HF. Finally, a clever purification sequence involving acetylation and recrystallization ensures the removal of trace impurities before the final deprotection step yields the target molecule. Detailed standardized synthesis steps see the guide below.
- Protect the 3',5'-hydroxyl groups of 1-beta-D-arabinofuranosyl uracil using a THP group to form the protected intermediate.
- React the protected intermediate with a triflating agent (e.g., CF3SO2F) in the presence of an organic base to form the 2'-triflate ester.
- Perform nucleophilic substitution using an organic base-hydrofluoric acid complex to introduce the fluorine atom, followed by deprotection, acetylation, recrystallization, and final deacetylation to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology translates into tangible operational efficiencies and risk mitigation. The primary advantage lies in the drastic simplification of the purification process. By replacing column chromatography with recrystallization, manufacturers can significantly reduce solvent consumption, waste generation, and processing time. This shift not only lowers the direct cost of goods sold but also enhances the environmental compliance profile of the production facility, aligning with increasingly stringent global regulations on chemical waste. The ability to produce white crystalline powder directly from recrystallization ensures a consistent and high-quality product that meets rigorous pharmaceutical standards without the variability often associated with chromatographic methods.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like DAST and anhydrous HF results in substantial raw material cost savings. Furthermore, the use of organic base-HF complexes allows for the utilization of standard reaction vessels, avoiding the capital investment required for Hastelloy or Teflon-lined reactors needed for anhydrous HF processes. The avoidance of column chromatography removes a major bottleneck in production capacity, allowing for higher throughput and reduced labor costs associated with batch processing and solvent recovery. These factors collectively contribute to a more economically viable manufacturing process that can compete effectively in the global market for nucleoside intermediates.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, such as trifluoromethanesulfonyl fluoride, triethylamine, and acetic anhydride, are commodity chemicals with robust global supply chains. This reduces the risk of supply disruptions that can occur with specialty reagents like DAST, which may have limited suppliers and longer lead times. The stability of the intermediates and the robustness of the reaction conditions ensure consistent production schedules, enabling suppliers to meet tight delivery deadlines for downstream API manufacturers. This reliability is crucial for maintaining continuous production lines in the pharmaceutical sector, where delays can have cascading effects on drug development and commercialization timelines.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop to pilot plant and full commercial production. The "pot-type" reaction capability minimizes the number of unit operations, reducing the footprint of the manufacturing facility and energy consumption. From an environmental perspective, the reduction in solvent usage and the elimination of silica gel waste from chromatography significantly lower the ecological impact of the synthesis. This aligns with the principles of green chemistry and supports corporate sustainability goals, making the supplier a more attractive partner for environmentally conscious pharmaceutical companies seeking to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of 2'-deoxy-2'-fluorouridine based on the patented technology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details helps in making informed decisions regarding process adoption and supplier selection.
Q: How does this method improve purity compared to traditional HF fluorination?
A: Traditional methods often require column chromatography due to side reactions and difficult purification. This patent introduces a recrystallization step of the 3',5'-diacetyl intermediate, allowing for the production of high-purity white crystalline powder without the need for complex chromatographic separation.
Q: What are the safety advantages of using organic base-HF complexes?
A: Unlike anhydrous hydrofluoric acid which is highly corrosive and requires specialized reactor materials, organic base-HF complexes (such as pyridine-HF or triethylamine-HF) are less corrosive, allowing the use of standard glass or stainless steel reactors while maintaining high nucleophilicity for the substitution reaction.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process is designed for industrial scalability. It utilizes readily available reagents like trifluoromethanesulfonyl fluoride and avoids expensive, difficult-to-handle reagents like DAST. The ability to perform pot-type reactions and simple recrystallization significantly enhances throughput and operational feasibility.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2'-Deoxy-2'-fluorouridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our team of expert chemists has extensively evaluated the synthetic route described in CN1795200A and possesses the technical capability to implement this advanced triflation-fluorination strategy at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2'-deoxy-2'-fluorouridine meets the highest industry standards for pharmaceutical intermediates.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality expectations. By leveraging our expertise in nucleoside chemistry and process optimization, we can help you secure a stable source of high-purity materials while minimizing your overall production expenses. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can support your long-term business goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
