Advanced Synthesis of Benzocarbazole Dye Sensitizers for High-Efficiency Solar Cells
Advanced Synthesis of Benzocarbazole Dye Sensitizers for High-Efficiency Solar Cells
The rapid evolution of renewable energy technologies has placed significant demand on the development of high-performance materials for dye-sensitized solar cells (DSSCs). A pivotal advancement in this sector is detailed in patent CN107253946B, which discloses a novel class of benzocarbazole compounds designed to function as superior dye sensitizers. These compounds, specifically designated as Formula I, Formula II, and Formula III, represent a strategic integration of a benzocarbazole electron donor, a diazosulfide bi-aromatic ring bridge, and a cyanoacetic acid acceptor. This unique molecular architecture is engineered to maximize visible light absorption and enhance photoelectric conversion efficiency, addressing critical bottlenecks in current photovoltaic material design. The patent outlines a robust synthetic methodology that allows for the precise construction of these complex conjugated systems, offering a promising pathway for the next generation of optoelectronic devices.
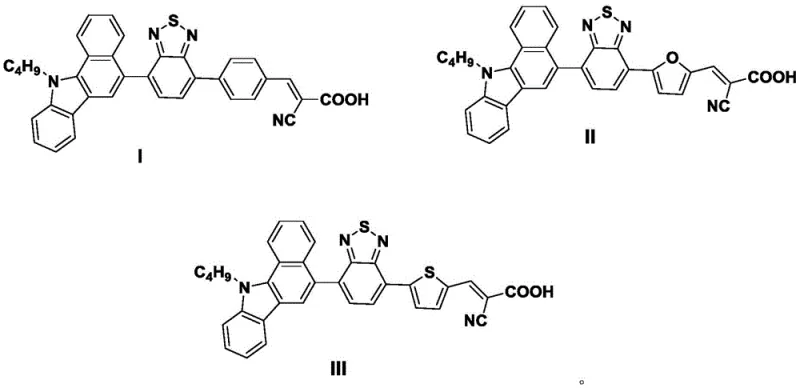
For research and development teams focusing on high-purity OLED material and photovoltaic intermediates, the structural novelty of these compounds cannot be overstated. The introduction of the benzocarbazole moiety provides a rigid, planar framework that facilitates efficient intramolecular charge transfer, a property often lacking in simpler carbazole derivatives. Furthermore, the variation in the pi-bridge—utilizing phenyl, furan, or thiophene rings—allows for fine-tuning of the HOMO-LUMO energy levels, thereby optimizing the match with the semiconductor bandgap. This level of molecular engineering is essential for achieving the reported photoelectric conversion efficiencies of up to 5.20%, positioning these compounds as valuable assets for any reliable electronic chemical supplier aiming to expand their portfolio in the renewable energy sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of D-pi-A type dye sensitizers has been plagued by challenges related to synthetic complexity and thermal stability. Conventional methods often rely on less stable donor groups that degrade under prolonged sunlight irradiation or require harsh reaction conditions that introduce difficult-to-remove impurities. Many existing carbazole-based dyes suffer from limited conjugation lengths, which restricts their absorption range primarily to the UV region, leaving a significant portion of the solar spectrum unharvested. Additionally, traditional synthetic routes frequently involve multi-step protections and deprotections, leading to lower overall yields and increased waste generation. For procurement managers, these inefficiencies translate into higher costs of goods sold and unpredictable supply chains, as the reliance on unstable intermediates can cause batch-to-batch variability that complicates quality control protocols in large-scale manufacturing environments.
The Novel Approach
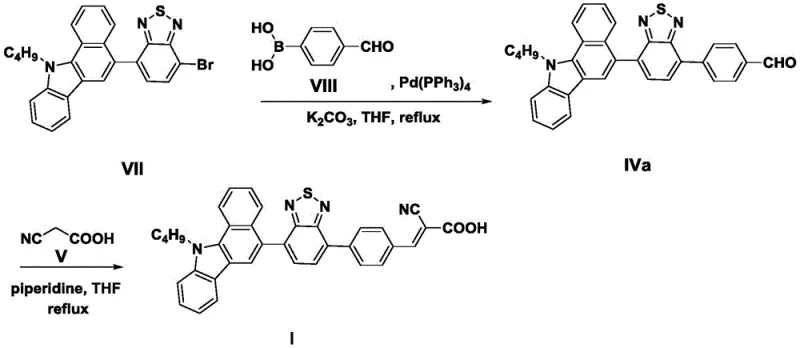
The methodology presented in CN107253946B offers a transformative solution by streamlining the synthesis into a highly efficient two-stage process centered around palladium-catalyzed cross-coupling and base-mediated condensation. By utilizing a pre-functionalized benzocarbazole-quinoxaline bromide (Formula VII) as a common starting point, the process allows for the modular introduction of different pi-bridges via Suzuki coupling with readily available boronic acids (Formulas VIII, IX, X). This modularity significantly reduces the time required to screen and optimize new dye variants. The subsequent Knoevenagel condensation step is performed under relatively mild conditions (60-80°C), avoiding the extreme temperatures that often lead to decomposition in sensitive organic electronics. This approach not only enhances the thermal stability of the final product but also simplifies the purification process, making it an ideal candidate for cost reduction in display & optoelectronic materials manufacturing.
Mechanistic Insights into Pd-Catalyzed Coupling and Knoevenagel Condensation
The core of this synthetic strategy relies on the precise execution of a Suzuki-Miyaura cross-coupling reaction to construct the extended pi-conjugated backbone. In this mechanism, the tetrakis(triphenylphosphine)palladium(0) catalyst facilitates the oxidative addition into the carbon-bromine bond of the benzocarbazole precursor, forming a reactive organopalladium intermediate. This species then undergoes transmetallation with the aryl boronic acid in the presence of a base, typically potassium carbonate, which activates the boron species. The final reductive elimination step releases the coupled aldehyde product (Formula IVa-c) and regenerates the active palladium catalyst. This cycle is crucial for maintaining high turnover numbers and ensuring that residual metal content in the final dye is minimized, a critical parameter for preventing recombination losses in the final solar cell device.
Following the backbone construction, the installation of the electron-accepting anchor group is achieved through a classic Knoevenagel condensation mechanism. In this step, the aldehyde group of the intermediate reacts with the active methylene group of cyanoacetic acid. The presence of a secondary amine base, such as piperidine, serves a dual purpose: it deprotonates the cyanoacetic acid to form a nucleophilic enolate and simultaneously activates the carbonyl carbon of the aldehyde through iminium ion formation. This synergistic activation lowers the energy barrier for the carbon-carbon bond formation, driving the reaction to completion even at moderate temperatures. The resulting double bond extends the conjugation system further, red-shifting the absorption maximum into the visible region. Crucially, the reaction conditions specified (reflux in chloroform or acetonitrile for 8-14 hours) ensure thermodynamic control, favoring the formation of the more stable E-isomer, which is essential for optimal packing on the TiO2 surface.
How to Synthesize Benzocarbazole Dye Sensitizers Efficiently
The synthesis of these high-value intermediates requires strict adherence to the optimized parameters outlined in the patent to ensure reproducibility and high purity. The process begins with the preparation of the aldehyde intermediates via Suzuki coupling, followed by the condensation with cyanoacetic acid. Each step demands careful control of stoichiometry, temperature, and atmosphere to prevent side reactions such as homocoupling or hydrolysis. The detailed standardized synthesis steps below provide a roadmap for scaling this chemistry from gram-scale laboratory experiments to kilogram-level production, ensuring that the critical quality attributes of the dye are maintained throughout the process.
- Perform a Suzuki coupling reaction between the brominated benzocarbazole-quinoxaline precursor and appropriate formyl-boronic acids using Pd(PPh3)4 catalyst in THF with potassium carbonate.
- Purify the resulting aldehyde intermediate (Formula IVa-c) via silica gel column chromatography using dichloromethane and petroleum ether.
- Conduct a Knoevenagel condensation by refluxing the aldehyde intermediate with cyanoacetic acid and piperidine in chloroform or acetonitrile for 8 to 14 hours.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers substantial logistical and economic benefits over traditional dye manufacturing processes. The reliance on commodity chemicals such as cyanoacetic acid, piperidine, and common boronic acids ensures a stable and diversified supply base, reducing the risk of raw material shortages that often plague the fine chemical industry. Furthermore, the use of standard solvents like tetrahydrofuran, chloroform, and dichloromethane means that existing manufacturing infrastructure can be utilized without the need for specialized corrosion-resistant equipment or exotic solvent recovery systems. This compatibility with standard plant operations significantly lowers the barrier to entry for commercial scale-up, allowing for rapid deployment of production capacity to meet market demand.
- Cost Reduction in Manufacturing: The streamlined two-step synthesis eliminates the need for multiple protection and deprotection sequences that are common in complex heterocycle synthesis. By removing these unit operations, manufacturers can achieve significant savings in labor, solvent consumption, and waste disposal costs. The high yields reported in the patent examples, particularly the 87% yield for Compound I, indicate a highly atom-economical process that maximizes the value derived from each kilogram of starting material. Additionally, the avoidance of expensive transition metal catalysts in the final condensation step further reduces the cost of goods, as there is no need for costly metal scavenging resins to meet strict purity specifications for electronic applications.
- Enhanced Supply Chain Reliability: The modular nature of the synthetic route allows for the decoupling of intermediate production from final dye assembly. Aldehyde intermediates (Formula IVa-c) can be synthesized and stockpiled in advance, creating a buffer inventory that insulates the supply chain from fluctuations in the availability of specific boronic acids. This flexibility enables a just-in-time manufacturing model where the final condensation step can be triggered based on real-time customer orders, drastically reducing lead times for high-purity benzocarbazole dye sensitizers. The robustness of the reaction conditions also implies a wider operating window, reducing the likelihood of batch failures due to minor process deviations and ensuring consistent on-time delivery.
- Scalability and Environmental Compliance: The reaction conditions described, specifically the moderate temperatures of 60-80°C and atmospheric pressure operation, are inherently safer and easier to scale than high-pressure or cryogenic alternatives. This thermal profile minimizes energy consumption for heating and cooling, contributing to a lower carbon footprint for the manufacturing process. Moreover, the purification method utilizes standard silica gel chromatography with well-defined eluent systems, which can be easily translated into continuous chromatography or crystallization processes at an industrial scale. The absence of highly toxic reagents or persistent organic pollutants in the waste stream simplifies effluent treatment, ensuring compliance with increasingly stringent environmental regulations governing the production of specialty chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on performance metrics and process feasibility for potential partners and licensees.
Q: What is the photoelectric conversion efficiency of these new benzocarbazole dyes?
A: According to patent CN107253946B, the dye-sensitized solar cells assembled with these compounds achieve photoelectric conversion efficiencies ranging from 4.35% to 5.20%, with the furan-bridged variant showing the highest performance.
Q: What are the key reaction conditions for the final condensation step?
A: The final step involves heating the aldehyde precursor with cyanoacetic acid in an organic solvent like chloroform or acetonitrile at 60-80°C for 8 to 14 hours under the catalysis of a base such as piperidine.
Q: Why is the benzocarbazole structure preferred for DSSC applications?
A: Benzocarbazole offers strong electron-donating ability, excellent planarity, and promotes better delocalization of dye molecules, leading to high responsivity in the visible light region and improved stability under sunlight.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzocarbazole Dye Sensitizer Supplier
As the global demand for efficient renewable energy solutions continues to surge, the ability to source high-quality dye sensitizers is paramount for the success of DSSC manufacturers. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver these advanced benzocarbazole compounds. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch meets the exacting standards required for high-performance optoelectronic applications. We understand that consistency is key in the electronics sector, and our dedicated process chemistry team is committed to maintaining the highest levels of quality assurance throughout the entire manufacturing lifecycle.
We invite industry leaders to collaborate with us to unlock the full potential of these novel materials. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your product development timeline and secure a competitive advantage in the rapidly evolving market for dye-sensitized solar cells.
