Advanced Asymmetric Hydrogenation for High-Purity Pharmaceutical Intermediates and Commercial Scalability
Introduction to Advanced Catalytic Methodologies in Fine Chemical Synthesis
The pharmaceutical industry continuously demands more efficient and stereoselective pathways for the production of complex active pharmaceutical ingredients (APIs) and their precursors. Patent CN1139576C introduces a groundbreaking catalytic method for the manufacture of optically active (R)- or (S)-1-(4-methoxy-benzyl)-1,2,3,4,5,6,7,8-octahydro-isoquinoline adducts, which serve as critical intermediates in the synthesis of medicinal products such as dextromethorphan. This technology represents a significant leap forward in asymmetric synthesis, addressing long-standing challenges regarding stability and selectivity in the hydrogenation of enimine derivatives. By leveraging sophisticated iridium-based catalytic systems coordinated with optically active diphosphine ligands, this process enables the direct transformation of stable enimine salts into high-value chiral amines with exceptional control over stereochemistry. The implications for large-scale manufacturing are profound, offering a route that bypasses the inefficiencies of traditional racemic resolution while maintaining rigorous purity standards required for regulatory compliance in the global pharmaceutical market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of octahydroisoquinoline derivatives relied heavily on non-stereoselective hydrogenation methods followed by cumbersome resolution steps. Conventional approaches often utilized heterogeneous catalysts that produced racemic mixtures, necessitating the separation of the desired (S)-enantiomer from the unwanted (R)-isomer. This resolution process is inherently inefficient, theoretically limiting the maximum yield to 50% unless complex racemization and recycling loops are implemented, which adds significant operational cost and waste. Furthermore, prior art indicates that attempting to hydrogenate the free base form of the enimine substrate is problematic due to its intrinsic chemical instability. The free base tends to undergo disproportionation reactions, yielding a mixture of 1-(4-methoxy-benzyl)-5,6,7,8-tetrahydro-isoquinoline and the desired octahydro product in unpredictable ratios. This lack of stability not only complicates purification but also leads to inconsistent batch quality, posing a severe risk for supply chain reliability and cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
The methodology disclosed in CN1139576C fundamentally alters the reaction landscape by utilizing the enimine substrate in the form of stable acid addition salts rather than the unstable free base. This strategic shift allows for the direct asymmetric hydrogenation of the carbon-nitrogen double bond without the aforementioned disproportionation side reactions. The process employs a homogeneous catalytic system comprising an iridium complex and a chiral diphosphine ligand, optionally supplemented with specific additives to fine-tune reactivity. 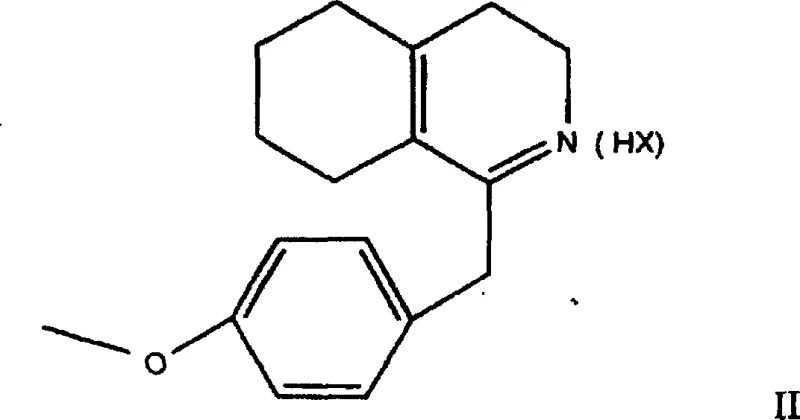 As illustrated in the reaction scheme, the transformation proceeds smoothly under hydrogen pressure to yield the target compound of Formula I with high conversion rates. This approach eliminates the need for post-reaction resolution, effectively doubling the theoretical yield compared to racemic methods and significantly simplifying the downstream processing workflow. The ability to handle the substrate as a stable salt also enhances operational safety and storage stability, making it a superior choice for reliable pharmaceutical intermediate supplier operations.
As illustrated in the reaction scheme, the transformation proceeds smoothly under hydrogen pressure to yield the target compound of Formula I with high conversion rates. This approach eliminates the need for post-reaction resolution, effectively doubling the theoretical yield compared to racemic methods and significantly simplifying the downstream processing workflow. The ability to handle the substrate as a stable salt also enhances operational safety and storage stability, making it a superior choice for reliable pharmaceutical intermediate supplier operations.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
The core of this technological advancement lies in the precise engineering of the catalytic center. The active species is generated from iridium precursors, such as [IrCl(COD)]2, coordinated with bulky, electron-rich chiral diphosphine ligands. These ligands, exemplified by structures like (R,R)-MOD-DIOP or various BPPM derivatives, create a highly defined chiral pocket around the metal center. 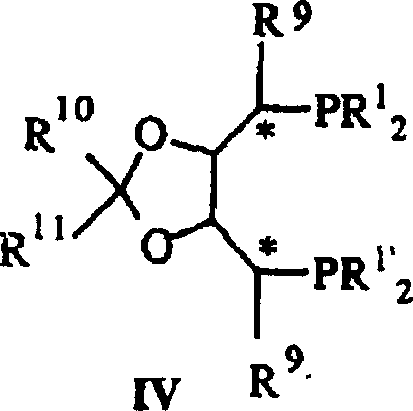 The steric bulk provided by substituents such as tert-butyl or methoxy groups on the phosphine aryl rings dictates the facial selectivity of the hydrogen addition to the C=N bond. When the enimine salt coordinates to the iridium center, the chiral environment forces the hydride transfer to occur from a specific direction, thereby inducing high enantiomeric excess (e.e.) in the product. The patent highlights that the nature of the counterion in the substrate salt (e.g., HBF4, H2SO4, HPF6) and the presence of additives like iodide salts (Bu4N+I-) play pivotal roles. The iodide ions are believed to act as promoters that stabilize the cationic iridium species or facilitate ligand exchange, thereby boosting both the turnover frequency and the stereocontrol of the reaction. This mechanistic understanding allows for the rational selection of ligand-substrate pairs to optimize outcomes for specific commercial applications.
The steric bulk provided by substituents such as tert-butyl or methoxy groups on the phosphine aryl rings dictates the facial selectivity of the hydrogen addition to the C=N bond. When the enimine salt coordinates to the iridium center, the chiral environment forces the hydride transfer to occur from a specific direction, thereby inducing high enantiomeric excess (e.e.) in the product. The patent highlights that the nature of the counterion in the substrate salt (e.g., HBF4, H2SO4, HPF6) and the presence of additives like iodide salts (Bu4N+I-) play pivotal roles. The iodide ions are believed to act as promoters that stabilize the cationic iridium species or facilitate ligand exchange, thereby boosting both the turnover frequency and the stereocontrol of the reaction. This mechanistic understanding allows for the rational selection of ligand-substrate pairs to optimize outcomes for specific commercial applications.
Furthermore, the impurity profile is tightly controlled through the stabilization of the substrate. By avoiding the free base form, the formation of tetrahydro-isoquinoline byproducts is suppressed. The reaction conditions, typically ranging from 20°C to 100°C and 10 to 90 bar of hydrogen pressure, are mild enough to prevent thermal degradation yet vigorous enough to ensure complete conversion. The use of solvent systems such as toluene/methanol mixtures or even supercritical carbon dioxide provides a tunable medium that solubilizes both the organic substrate and the ionic catalyst components. This solubility balance is crucial for maintaining a homogeneous reaction phase, which ensures consistent heat and mass transfer throughout the reactor volume, a key factor in achieving reproducible high-purity results in commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 1-(4-methoxy-benzyl)-octahydroisoquinoline Efficiently
Implementing this synthesis route requires careful attention to the preparation of the catalyst and the handling of the sensitive enimine salts. The process begins with the in situ generation of the active iridium complex under strictly anaerobic conditions to prevent catalyst deactivation by oxygen. The substrate, typically prepared via a Bishler-Napieralsky cyclization followed by salt formation, is introduced into the reactor along with the catalyst solution and necessary additives. The detailed standardized synthesis steps involve precise control over the substrate-to-catalyst ratio, which can range widely from 100 to 50,000 depending on the specific ligand efficiency desired.
- Preparation of the Iridium Catalyst Complex: Dissolve the iridium precursor [IrCl(COD)]2 and the chiral diphosphine ligand (e.g., (R,R)-MOD-DIOP) in an inert solvent like methanol under an oxygen-free atmosphere.
- Substrate Addition and Additive Integration: Introduce the enimine salt substrate (Formula II) along with specific additives such as tetrabutylammonium iodide and a base like triethylamine to the catalyst solution.
- Asymmetric Hydrogenation Reaction: Seal the reactor and conduct hydrogenation at controlled temperatures (20-100°C) and pressures (10-90 bar) to achieve high enantiomeric excess and conversion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the manufacturing workflow. By eliminating the resolution step required in traditional racemic synthesis, the process removes an entire unit operation involving crystallization, filtration, and mother liquor recycling. This reduction in processing steps translates directly into lower capital expenditure for equipment and reduced operational costs for labor and utilities. Moreover, the theoretical yield improvement from a maximum of 50% in resolution processes to near-quantitative yields in this asymmetric method means that less raw material is required to produce the same amount of final API, driving down the cost of goods sold significantly.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal removal steps often associated with heterogeneous catalysis, combined with the high efficiency of the homogeneous iridium system, leads to streamlined production. The ability to recycle the catalyst or operate at high turnover numbers reduces the consumption of precious metals. Additionally, the use of stable salt forms minimizes material loss due to decomposition, ensuring that every kilogram of starting material contributes effectively to the final output, resulting in substantial cost savings in fine chemical manufacturing.
- Enhanced Supply Chain Reliability: The robustness of the salt form of the substrate ensures longer shelf-life and easier logistics compared to unstable free bases. The process tolerates a variety of solvent systems, including greener alternatives like supercritical CO2, which mitigates regulatory risks associated with volatile organic compound (VOC) emissions. This flexibility allows manufacturers to adapt quickly to changing environmental regulations without disrupting production schedules, thereby securing a continuous supply of high-purity pharmaceutical intermediates for downstream customers.
- Scalability and Environmental Compliance: The reaction conditions described, such as moderate temperatures and pressures, are well within the operating parameters of standard industrial hydrogenation reactors. The high substrate-to-catalyst ratios demonstrated in the patent examples indicate that the process is economically viable even at multi-ton scales. Furthermore, the high selectivity reduces the generation of hazardous waste streams associated with byproduct separation, aligning with modern green chemistry principles and facilitating easier waste management and environmental compliance certification.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the operational parameters and expected outcomes for potential licensees or manufacturing partners. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this route into their existing production pipelines.
Q: Why is the salt form of the enimine substrate preferred over the free base in this hydrogenation process?
A: The free base form of the enimine substrate is chemically unstable and prone to disproportionation into tetrahydro- and octahydro-isoquinoline byproducts. Utilizing stable acid addition salts (such as tetrafluoroborate or bisulfate salts) prevents these side reactions, ensuring higher selectivity and yield of the desired optically active product.
Q: What role do additives like iodide salts play in the iridium-catalyzed reaction?
A: Additives such as tetrabutylammonium iodide serve as crucial promoters that significantly enhance both the catalytic activity and the enantioselectivity of the iridium complex. They help stabilize the active catalytic species and facilitate the coordination of the substrate, leading to improved reaction rates and higher optical purity.
Q: Can this process be scaled for industrial production of dextromethorphan precursors?
A: Yes, the process is designed for scalability with substrate-to-catalyst molar ratios ranging from 100 to 50,000. The use of robust solvent systems like toluene/methanol mixtures and the ability to operate at moderate pressures makes it highly suitable for commercial-scale manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-(4-methoxy-benzyl)-octahydroisoquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are equipped with stringent purity specifications and rigorous QC labs capable of verifying the enantiomeric excess and chemical purity of complex chiral intermediates to meet the most demanding international pharmacopoeia standards. Our commitment to quality assurance guarantees that every batch of 1-(4-methoxy-benzyl)-octahydroisoquinoline delivered meets the precise requirements necessary for the synthesis of safe and effective medicinal products.
We invite you to collaborate with us to leverage this advanced iridium-catalyzed technology for your supply chain needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this process can optimize your overall manufacturing economics. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us partner with you to drive innovation and efficiency in your pharmaceutical intermediate sourcing strategy.
